Fat interesterification
In the food industry and biochemistry, interesterification (IE) is a process that rearranges the fatty acids of a fat product, typically a mixture of triglyceride. The process implies breaking and reforming the ester bonds C–O–C that connect the fatty acid chains to the glycerol hubs of the fat molecules. These reactions are performed by inorganic catalysts, yielding what is called chemical interesterification (CIE) in the industry; or by enzymes, in the so-called enzymatic interesterification (EIE).[1]
This process is typically used to adjust the physical characteristics of the fat, such as melting point and plasticity, for specific uses. It can be used, for instance, to turn oils into solid or semisolid products by combining them with other solid fats. It can also be used to prevent separation of solid fractions in palm oil and lauric fats,[2] slow rancidification, or create oils more suitable for deep frying.
Compared to other processes that are used for the same purpose, such as hydrogenation, interesterification generally preserves the original distribution of fatty acids in the product, and hence is expected to preserve its nutritional and health attributes. However, those other techniques may still be applied to the starting fats or to the products of IE, and the latter may be blended with other fats. Also, some of the new triglycerides produced by IE may be fractionated (separated) through controlled crystallization.[3]
Interesterified fats are used in many industrial food products, including cookies, crackers, biscuits, cakes and icings, dairy fat replacers, pie crust, popcorn, flatbread and tortillas.[4]
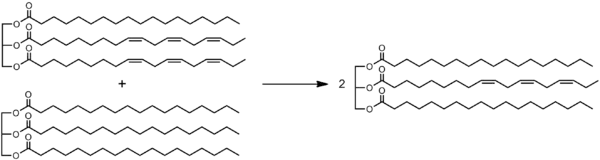
Feedstock
Typically the feedstock (starting product) is a mixture of two or more oils.
In particular, an unsaturated vegetable oil can be interesterified with a fully hydrogenated version thereof. This procedure yield a less unsaturated fat without creating the trans fatty acids that would be produced by partial hydrogenation.[3]
Process
Chemistry
In principle, when interesterification applied to two pure triglycerides, each with three identical fatty acids (AAA and BBB), the result could contain six different triglycerides (AAA, AAB, ABA, ABB, BAB, and BBB). The number is 6, rather than 23 = 8, because of the symmetry of the glycerol backbone.[5] The number is much greater if the feedstock has three or more distinct fatty acids.
"Chemical" interesterification
The so-called "chemical" interesterification, the catalyst is an inorganic compound such as sodium methoxide. The reaction is carried out at high temperatures and creates three by-products — sodium soaps, fatty methyl esters, and monoglycerides) in addition to the interesterified fats.[6]
Enzymatic interesterification

Enzymatic interesterification (IE) uses an enzyme to break and reform the ester bonds. Enzymes most suitable for this process are esterase; lipase; acylase; those enzymes that facilitate acidolysis reactions, transesterification reactions, ester synthesis or ester interchange reactions; enzymes having phospholipase or protease activity, including thermostable and thermotolerant hydrolase activity; and polynucleotides.[7][8][9][10][11]
Some enzymes will break and reform ester bonds only at positions 1 and 3 (sp1 and sp3) of the glycerol hub, leaving the acids in position 2 (sp2) fixed.[5]
The most common industrial EIE process forces the liquid fat feedstock through a fixed-bed reactor, that typically contains an oil purification bed followed by an enzyme bed. The latter has the enzyme fixed on some inert granular substrate. The first bed removes impurities from the oil blend that could inactivate the enzyme or affect its performance. The enzyme activity decreases over time, so flow must be carefully monitored and adjusted over time to maintain conversion. [5]
Two or more reactors maybe used in tandem, where the first reactor has the lowest enzyme activity and absorbs most of the impurities and harmful compounds. This sequencing protects the most active enzymes, which are in the last reactors.[12][13]
EIE has been replacing CIE because it has fewer processing steps, can be carried out at lower temperatures, produces no by-products and has lower production costs.[14][15] [12]
Advantages
Compared to simple blends, interesterified fats have a wider plasticity range, meaning that they retain their physical properties over a wider temperature range, without separation of their components.[16] IE can also use a wider variety of feedstocks, such as soybean oil, they provide a better risk management profile than globally produced palm oil.
History
The earliest record of enzymatic Interesterification was in 1844, when Théophile-Jules Pelouze published a study on the synthesis of a triglyceride through the esterification of glycerol by butyric acid.[17] In 1920, Wilhelm Norman, who also patented the catalytic hydrogenation of fatty acids, was granted a patent for the chemical interesterification of edible lipids.[18] This process became a viable option for the food industry as it improved the spreadability and baking properties of the common shortening lard.
Enzymatic interesterification was developed in the 1970s by the team at the Unilever Research Center at Colworth House in England. Their work proved that the use of a specific enzyme predictably rearranged the fatty acids on the glycerol backbone of a triglyceride at positions 1 and 3. This provided an expanded range of available triglyceride types.[19]
Still, EIE remained largely confined to research laboratories due to high enzyme prices. It was only in the 2000s that general concerns about the health effects of trans acids drove the industry to adopt interesterification as a replacement for partial hydrogenation (which has been the oil hardening method of choice, due to its lower cost).[20] Adoption was greatly facilitated by the development of enzymes bound to inert solid substrates like silica, by Novozymes and other companies.[15]
See also
- Hydrogenation
- Trans fat
- Triglyceride
References
- ↑ Institute of Shortenings and Edible oils (2006). "Food Fats and oils" (PDF). Archived from the original (PDF) on 2007-03-26. Retrieved 2009-02-19.
- ↑ Costales-Rodriquez, R.; Gibon, V.; Verhe, R.; De Greyt, W. (2009), "Chemical and Enzymatic Interesterification of a Blend of Palm Stearin: Soybean Oil for Low Trans-Margarine Formulation.", J Am Oil Chem Soc, 86 (7): 681–697, doi:10.1007/s11746-009-1395-2
- 1 2 Kellens, Marc (2000). "Interesterification Process Conditions". Retrieved 2007-01-29.
- ↑ Hui, Y.H. (2006), Handbook of Food Science, Technology, and Engineering, Vol.1, Boca Raton: Taylor & Francis, ISBN 1-57444-551-0
- 1 2 3 “Chemical vs. Enzymatic Interesterification.” De Greyt, Wim. IUPAC-AOCS Workshop on Fats, Oils & Oilseeds Analyses & Production, 6 Dec. 2004. Retrieved October 20, 2010
- ↑ Rousseau, D. (2002): "The Effects of Interesterification on the Physical Properties of Fats". Chapter 13 of Physical Properties of Lipids. CRC Press.
- ↑ US 2001/0004462 Sugeria, et al.
- ↑ US 5,773,266 Bosley, et al.
- ↑ US 5,658,768 Quinlan.
- ↑ US 5,451,170 Miymoto, et al.
- ↑ US 5,219,733 Myojo, et al.
- 1 2 US application 0138867 Dayton
- ↑ US application 0317902 Dayton
- ↑ W. Hamm and R. Hamilton, editors (2000): Edible Oil Processing. ISBN 1-84127-038-5
- 1 2 T. L. Husum, L. S. Pedersen, P. M. Nielsen, M. W. Christensen, D. Kristensen, and H. C. Holm (2003): "Enzymatic interesterification: Process advantages and product benefits." Archived 2006-01-04 at the Wayback Machine . Palm Oil Information Online Service. Retrieved 2010-10-20.
- ↑ Osório, N. M.; Dubreucq, E.; Da Fonseca, MM R.; Ferreira-Dias, S. (2009), "Operational Stability of Immobilized Lipase/acyltransferase during Interesterification of Fat Blends.", Eur J Lipid Sci Technol, 111 (4): 358–367, doi:10.1002/ejlt.200800194, hdl:10400.5/9067
- ↑ Chim Phys 10 (1844). Pelouze, J. Ann. Page 434.
- ↑ DE 417,215 Norman
- ↑ Wisdom, R. A.; Dunnill, P.; Lilly, M. D.; Macrae, A. (1984), "Enzymic Interesterification of Fats: Factors Influencing the Choice of Support for Immobilized Lipase.", Enzyme and Microbial Technology, 6 (10): 443–446, doi:10.1016/0141-0229(84)90093-0
- ↑ "Legal and Policy Resources on Public Health 'Winnable Battles'" Archived 2011-01-31 at the Wayback Machine www.cdc.gov. Retrieved October 20, 2010.