Mesenchymal–epithelial transition
A mesenchymal–epithelial transition (MET) is a reversible biological process that involves the transition from motile, multipolar or spindle-shaped mesenchymal cells to planar arrays of polarized cells called epithelia. MET is the reverse process of epithelial–mesenchymal transition (EMT) and it has been shown to occur in normal development, induced pluripotent stem cell reprogramming,[1] cancer metastasis[2] and wound healing.[3]
Introduction
Unlike epithelial cells – which are stationary and characterized by an apico-basal polarity with binding by a basal lamina, tight junctions, gap junctions, adherent junctions and expression of cell-cell adhesion markers such as E-cadherin,[4] mesenchymal cells do not make mature cell-cell contacts, can invade through the extracellular matrix, and express markers such as vimentin, fibronectin, N-cadherin, Twist, and Snail.[4] MET plays also a critical role in metabolic switching and epigenetic modifications. In general epithelium-associated genes are upregulated and mesenchyme-associated genes are downregulated in the process of MET.[5]
In development
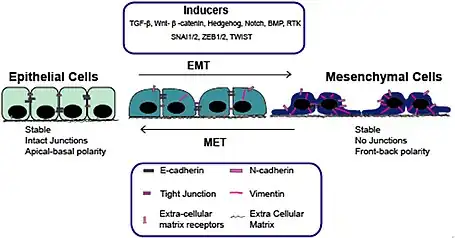
During embryogenesis and early development, cells switch back and forth between different cellular phenotypes via MET and its reverse process, epithelial–mesenchymal transition (EMT). Developmental METs have been studied most extensively in embryogenesis during somitogenesis[6] and nephrogenesis[7] and carcinogenesis during metastasis,[8] but it also occurs in cardiogenesis[9] or foregut development.[10] MET is an essential process in embryogenesis to gather mesenchymal-like cells into cohesive structures.[1] Although the mechanism of MET during various organs morphogenesis is quite similar, each process has a unique signaling pathway to induce changes in gene expression profiles.
Nephrogenesis
One example of this, the most well described of the developmental METs, is kidney ontogenesis. The mammalian kidney is primarily formed by two early structures: the ureteric bud and the nephrogenic mesenchyme, which form the collecting duct and nephrons respectively (see kidney development for more details). During kidney ontogenesis, a reciprocal induction of the ureteric bud epithelium and nephrogenic mesenchyme occurs. As the ureteric bud grows out of the Wolffian duct, the nephrogenic mesenchyme induces the ureteric bud to branch. Concurrently, the ureteric bud induces the nephrogenic mesenchyme to condense around the bud and undergo MET to form the renal epithelium, which ultimately forms the nephron.[7] Growth factors, integrins, cell adhesion molecules, and protooncogenes, such as c-ret, c-ros, and c-met, mediate the reciprocal induction in metanephrons and consequent MET.[11]
Somitogenesis
Another example of developmental MET occurs during somitogenesis. Vertebrate somites, the precursors of axial bones and trunk skeletal muscles, are formed by the maturation of the presomitic mesoderm (PSM). The PSM, which is composed of mesenchymal cells, undergoes segmentation by delineating somite boundaries (see somitogenesis for more details). Each somite is encapsulated by an epithelium, formerly mesenchymal cells that had undergone MET. Two Rho family GTPases – Cdc42 and Rac1 – as well as the transcription factor Paraxis are required for chick somitic MET.[12]
Cardiogenesis
Development of heart is involved in several rounds of EMT and MET. While development splanchnopleure undergo EMT and produce endothelial progenitors, these then form the endocardium through MET. Pericardium is formed by sinus venosus mesenchymal cells that undergo MET.[1] Quite similar processes occur also while regeneration in the injured heart. Injured pericardium undergoes EMT and is transformed into adipocytes or myofibroblasts which induce arrhythmia and scars. MET than leads to the formation of vascular and epithelial progenitors that can differentiate into vasculogenic cells which lead to regeneration of heart injury.[9]
Hepatogenesis
In cancer
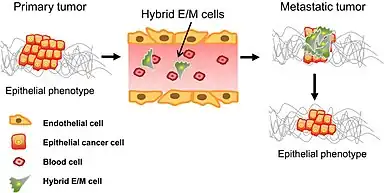
While relatively little is known about the role MET plays in cancer when compared to the extensive studies of EMT in tumor metastasis, MET is believed to participate in the establishment and stabilization of distant metastases by allowing cancerous cells to regain epithelial properties and integrate into distant organs. Between these two states, cells occur in 'intermediate‐state', or so‐called partial EMT.[8]
In recent years, researchers have begun to investigate MET as one of many potential therapeutic targets in the prevention of metastases.[14] This approach to preventing metastasis is known as differentiation-based therapy or differentiation therapy and it can be used for development of new anti-cancer therapeutic strategies.[1]
In iPS cell reprogramming
A number of different cellular processes must take place in order for somatic cells to undergo reprogramming into induced pluripotent stem cells (iPS cells). iPS cell reprogramming, also known as somatic cell reprogramming, can be achieved by ectopic expression of Oct4, Klf4, Sox2, and c-Myc (OKSM).[15] Upon induction, mouse fibroblasts must undergo MET to successfully begin the initiation phase of reprogramming. Epithelial-associated genes such as E-cadherin/Cdh1, Cldns −3, −4, −7, −11, Occludin (Ocln), Epithelial cell adhesion molecule (Epcam), and Crumbs homolog 3 (Crb3), were all upregulated before Nanog, a key transcription factor in maintaining pluripotency, was turned on. Additionally, mesenchymal-associated genes such as Snail, Slug, Zeb −1, −2, and N-cadherin were downregulated within the first 5 days post-OKSM induction.[16] Addition of exogenous TGF-β1, which blocks MET, decreased iPS reprogramming efficiency significantly.[17] These findings are all consistent with previous observations that embryonic stem cells resemble epithelial cells and express E-cadherin.[18]
Recent studies have suggested that ectopic expression of Klf4 in iPS cell reprogramming may be specifically responsible for inducing E-cadherin expression by binding to promoter regions and the first intron of CDH1 (the gene encoding for E-cadherin).[17]
See also
References
- 1 2 3 4 Pei D, Shu X, Gassama-Diagne A, Thiery JP (January 2019). "Mesenchymal-epithelial transition in development and reprogramming". Nature Cell Biology. 21 (1): 44–53. doi:10.1038/s41556-018-0195-z. PMID 30602762. S2CID 57373557.
- ↑ Pastushenko I, Brisebarre A, Sifrim A, Fioramonti M, Revenco T, Boumahdi S, et al. (April 2018). "Identification of the tumour transition states occurring during EMT". Nature. 556 (7702): 463–468. Bibcode:2018Natur.556..463P. doi:10.1038/s41586-018-0040-3. PMID 29670281. S2CID 4933657.
- ↑ Kalluri R (June 2009). "EMT: when epithelial cells decide to become mesenchymal-like cells". The Journal of Clinical Investigation. 119 (6): 1417–9. doi:10.1172/JCI39675. PMC 2689122. PMID 19487817.
- 1 2 Das V, Bhattacharya S, Chikkaputtaiah C, Hazra S, Pal M (February 2019). "The basics of epithelial-mesenchymal transition (EMT): A study from a structure, dynamics, and functional perspective". Journal of Cellular Physiology. 234 (9): 14535–14555. doi:10.1002/jcp.28160. PMID 30723913. S2CID 73448308.
- ↑ Owusu-Akyaw A, Krishnamoorthy K, Goldsmith LT, Morelli SS (January 2019). "The role of mesenchymal-epithelial transition in endometrial function". Human Reproduction Update. 25 (1): 114–133. doi:10.1093/humupd/dmy035. PMID 30407544.
- ↑ Hamidi S, Nakaya Y, Nagai H, Alev C, Shibata T, Sheng G (April 2019). "Biomechanical regulation of EMT and epithelial morphogenesis in amniote epiblast". Physical Biology. 16 (4): 041002. Bibcode:2019PhBio..16d1002H. doi:10.1088/1478-3975/ab1048. PMID 30875695. S2CID 80627655.
- 1 2 Holmquist Mengelbier L, Lindell-Munther S, Yasui H, Jansson C, Esfandyari J, Karlsson J, et al. (January 2019). "The Iroquois homeobox proteins IRX3 and IRX5 have distinct roles in Wilms tumour development and human nephrogenesis". The Journal of Pathology. 247 (1): 86–98. doi:10.1002/path.5171. PMC 6588170. PMID 30246301.
- 1 2 Liao TT, Yang MH (July 2017). "Revisiting epithelial-mesenchymal transition in cancer metastasis: the connection between epithelial plasticity and stemness". Molecular Oncology. 11 (7): 792–804. doi:10.1002/1878-0261.12096. PMC 5496497. PMID 28649800.
- 1 2 Nebigil CG, Désaubry L (May 2019). "The role of GPCR signaling in cardiac Epithelial to Mesenchymal Transformation (EMT)". Trends in Cardiovascular Medicine. 29 (4): 200–204. doi:10.1016/j.tcm.2018.08.007. PMID 30172578.
- ↑ Mu T, Xu L, Zhong Y, Liu X, Zhao Z, Huang C, et al. (30 July 2019). "Characterizing the Emergence of Liver and Gallbladder from the Embryonic Endoderm through Single-Cell RNA-Seq". doi:10.1101/718775.
{{cite journal}}: Cite journal requires|journal=(help) - ↑ Horster MF, Braun GS, Huber SM (October 1999). "Embryonic renal epithelia: induction, nephrogenesis, and cell differentiation". Physiological Reviews. 79 (4): 1157–91. doi:10.1152/physrev.1999.79.4.1157. PMID 10508232.
- ↑ Nakaya Y, Kuroda S, Katagiri YT, Kaibuchi K, Takahashi Y (September 2004). "Mesenchymal-epithelial transition during somitic segmentation is regulated by differential roles of Cdc42 and Rac1". Developmental Cell. 7 (3): 425–38. doi:10.1016/j.devcel.2004.08.003. PMID 15363416.
- ↑ Li B, Zheng YW, Sano Y, Taniguchi H (February 2011). Abdelhay E (ed.). "Evidence for mesenchymal-epithelial transition associated with mouse hepatic stem cell differentiation". PLOS ONE. 6 (2): e17092. Bibcode:2011PLoSO...617092L. doi:10.1371/journal.pone.0017092. PMC 3037942. PMID 21347296.
- ↑ Pattabiraman DR, Bierie B, Kober KI, Thiru P, Krall JA, Zill C, et al. (March 2016). "Activation of PKA leads to mesenchymal-to-epithelial transition and loss of tumor-initiating ability". Science. 351 (6277): aad3680. doi:10.1126/science.aad3680. PMC 5131720. PMID 26941323.
- ↑ Takahashi K, Yamanaka S (August 2006). "Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors". Cell. 126 (4): 663–76. doi:10.1016/j.cell.2006.07.024. hdl:2433/159777. PMID 16904174. S2CID 1565219.
- ↑ Samavarchi-Tehrani P, Golipour A, David L, Sung HK, Beyer TA, Datti A, et al. (July 2010). "Functional genomics reveals a BMP-driven mesenchymal-to-epithelial transition in the initiation of somatic cell reprogramming". Cell Stem Cell. 7 (1): 64–77. doi:10.1016/j.stem.2010.04.015. PMID 20621051.
- 1 2 Li R, Liang J, Ni S, Zhou T, Qing X, Li H, et al. (July 2010). "A mesenchymal-to-epithelial transition initiates and is required for the nuclear reprogramming of mouse fibroblasts". Cell Stem Cell. 7 (1): 51–63. doi:10.1016/j.stem.2010.04.014. PMID 20621050.
- ↑ Baum B, Settleman J, Quinlan MP (June 2008). "Transitions between epithelial and mesenchymal states in development and disease". Seminars in Cell & Developmental Biology. 19 (3): 294–308. doi:10.1016/j.semcdb.2008.02.001. PMID 18343170.