Passive electrolocation in fish

Passive electrolocation is a process where certain species of fish or aquatic amphibians can detect electric fields using specialized electroreceptors to detect and to locate the source of an external electric field in its environment creating the electric field. These external electric fields can be produced by any bioelectrical process in an organism, especially by actions of the nerves or muscles of fish, or indeed by the specially developed electric organs of fish. Other fields are induced by movement of a conducting organism through the earth's magnetic field, or from atmospheric electricity.[1][2][3]
Electrolocating fish use this ability to detect prey, locate other fish, avoid predators, and perhaps to navigate by the Earth's magnetic field. Electroreceptors probably evolved once or twice early in vertebrate evolution, but the sense was apparently lost in amniotes, and in a large number of the Actinopterygii (ray finned fishes) only to reappear independently in two teleost clades.[1] In fish, the ampullary receptor is a specialized receptor that it uses to sense these electric fields and allows the fish to follow electric field lines to their source. Sharks primarily use specialized receptors, called Ampullae of Lorenzini, to detect their prey's low frequency DC fields and may also use their receptors in navigation by the Earth's magnetic field. Weakly electric fish use their ampullary receptors and tuberous receptors to detect the weakly electric fields produced by other fish, as well as for possible predator avoidance.[3] Passive electrolocation contrasts with active electrolocation, in which the animal emits its own weak self generated electric field and detects nearby objects by detecting the distortion of its produced electric field. In active electrolocation the animal senses its own electromotor discharge or reafference instead of some externally generated electric field or discharge.[4]
Evolution of electroreception and electrolocation
Electroreception occurred early in evolutionary history with the evolution of an ampullary sensory system that included receptors able to detect weak electric signals in the environment (less than 1 µV/cm or 50 Hz).[2] Sense organs specialized for electroreception have only been found among vertebrates, and around 8.600 species are known to be electroreceptive.[4] The majority of teleosts and amniotes do not have an electroreceptive system, but the distribution of electroreception in terms of evolution involves different classes of fish.[4] First, there is an origin of a common ancestor of current existent vertebrates (close to lampreys and gnathostomes). This ancestor evolved a lateral line that is important in processing sensory information and present in today's hagfishes. Ampullary receptors are ancestral to jawed fish, for lampreys and agnathans were found to have ampullary receptors 400 million years earlier.[2] Second, there is a loss of electroreception in amniotes. This could be due to the fact that air is a poor medium to effectively conduct the electric fields, unlike water. Third, there is also a loss of electroreception in gars, bowfin, and teleosts (neopterygian fishes). Ampullary receptors are present in all surviving cartilaginous fishes and bony fishes except a few species present in Neopterygii (which include gars, bowfins, and teleosts)[2] Fourth, monotremes and a least three groups of fresh-water teleosts re-evolved electroreception. Fifth, a group of teleosts, Xenomystinae, acquired passive electrolocation with low frequency sensitive ampullary receptors. Sixth, another group of teleosts, Mormyroids, evolved passive electrolocation and active location, as well as a pulse type EOD in active electrolocation. Seventh, Silurifiormes and Gymnoformes evolved passive electrolocation, and Gymnoformes also evolved active electrolocation with a pulse type or wave type EODs.[4]
Detecting electric fields
Electric fields found in aquatic environments range from local fields to large-scale uniform fields produced by ocean currents.[2] For example, uniform electric fields might provide information on the orientation of field intensity and can give information about the voltage of the electric field through the velocity and direction of the ocean current. Electric fields produced locally by an animate source represent more of an electric field produced by a dipole, which can be represented by two point charges, a positive and a negative. The electric field in water generates an electric current which flows from positive to negative, along lines resembling a dipole field. A field produced by a fish changes according to distance. In a field far away, a dipole produced by a fish looks more like a dipole source. Higher magnitude order multipole fields may dominate closer to the fish, where the current does not converge on one point for the anterior pole but makes a perpendicular gradient over the length of the fish.[1][3] A dipole contribution is more accurate in fields farther from the source because in three dimensions the dipole contribution to the electric field decreases due to the inverse third power of distance. In contrast, the contributions from quadrupole sources decrease due to the source decreasing by an inverse fourth power of distance.[1] When fish use their electroreceptive sense to detect the electric fields, it is different from other senses such as vision, auditory, and vibration senses. This is due to the fact that stimuli such as light and sound travel in vectors that are associated from the propagation of the wave, and the velocity vector points to the source of the signal.[1] The signal produced by fish used in electroreception exist rather as an electrostatic field rather than propagating as waves due to the fact the frequencies of the fields are very low and only reach up to several kilohertz.[1] Propagating electromagnetic waves would have a wavelength measured in kilometers at this low frequency. The electric fields produced by biological organisms like fish attenuate rapidly in aquatic environments and have a field magnitude around zero at ranges of a few meters at which fish detect these fields. Unlike sounds waves in hearing, there is no velocity vector to describe propagation, shadow cast by the receiver's body, or temporal delay associated with propagation.[1] The non-propagating electric field only gives curved paths about signal direction, but no information about delay times or a direct path to the source.[5] Instead of using precise coding such as in hearing, it is important that the receptors are able to detect the low frequency electric fields by relying on spatial information of intensity, orientation, and polarity of the fields.[5]
Another type of electric field detected in electroreception is DC field potentials. All aquatic animals show DC fields, and it is the strongest in the head and gill region in species of fish.[2] Teleosts had a range of about 500 µV potential around the gills, while elasmobrachs and crustaceans were 10 times weaker; but if a crustacean was wounded, its DC potential increased to 1 µV. A study by Peters and Bretschneider measured the bioelectric potential across catfish and found the strongest negative potential around the head and gills of the fish, measured 1 mm from the skin.[2] Yet the sources of DC potentials are still not well understood, for potentials still persist even if the animal is anesthetized, so muscle contractions do not modulate DC potentials.[2] Respiratory movements can module DC potentials, but are not caused by the contraction of muscles, since passive moments of the gill covers cause similar modulation in anesthetized or awake animals and muscle contractions produce high frequency signals.[2] When a fish is hurt or distressed, it also emits a stronger DC field potential, which makes it more susceptible to predation.
In order to detect low frequency fields and DC fields, fish have specialized receptors to read and process the incoming information. The ampullary electrosensory systems of fish and amphibians are modified hair cell sensors with similarities to the lateral line system and audition.[5] Ampullary receptors pores on the skins of the animal, and each pore opens to a water or jelly filled tube which leads to the terminal swelling.[3] Outer faces contact the ampullary lumen via the aquatic environment while the inner faces synapse with nerve fibers.[3] Flattened cells line the ampullary duct and create a layer of high electrical resistance. The length of the tube varies in length depending on the environment. A marine environment has longer tubes in the receptor when compared to freshwater environments because of cable theory. This theory states when a potential difference is applied between the outside pore and to loose connective tissues to the location of the ampulla proper, no appreciable voltage drops occurs along the canal and the voltage difference occurs across the ampullary wall near the pore of the skin.[3] The tube is well insulated and signals voltage down the sensory epithelium to the electroreceptors. In freshwater, the resistance of the medium is higher than in saltwater, so the tube must channel voltage down a shorter path in order to have the same voltage reach the electroreceptor.[3]

In sharks, the Ampullae of Lorenzini is the ampullary organ used to detect electrical stimuli. These cells are distributed along the dorsal and ventral head region of the sharks, but are also found in the pectoral region in skates and rays. The receptor is located in clusters, with a higher density of receptors on the ventral side of the head than the dorsal side of the head, but density and canal length varies by individual and species.[6][7] The species of teleosts with electroreception (Gymnotiformes, Mormyridae, and Silurids) are freshwater fish and therefore have smaller ampullary canals than marine species. Mormyrids have ampullary ducts distributed on the head as well as along the top and bottom of their body.[7] Silurids have most ampullary organs in the head skin, but ampullary receptors are also found along the body side. Gymnotids have ampullary organs distributed on the head as well as scattered along the side of the body.[7] Besides having ampullary receptors, teleost fish possess tuberous receptors, which come in two types: pulse marker receptors that code for the amplitude of the signal, and burst duration coders that code for the timing of the signal.[8] Both of these receptors are directional in their responses to the electrical fields. The receptors are scattered across the head and body of the fish, directionally selective, and the best response is when the current is perpendicular to the skin.[9]
Localization of external sources (electric fields)
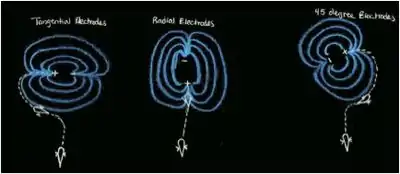

Fish use electroreception to detect the electrical fields produced by other fish, but they also need a way to navigate to the source of the electric field. The use of electroreception to locate the source of the electric field is known as electroreception. Local bioelectric fields produced do not remain strong, and get proportionally weaker to the cube of the distance.[5] These low frequency bioelectric fields are produced from a dipole like orientation in the body of the fish when detected from a farther distance. The current will flow from negative to positive. Using electrodes, Hopkins made different orientations of electric fields to test how fish navigate. He aligned the field radially, tangentially, and at a 45-degree geometry orientation. In other cases, the dipoles could rotate and thus rotate the electric field. The field flows from a positive electrode to the negative electrode.[9]
The ampullary receptors of non-teleost fish are not homologous with those of teleosts. Non-teleosts are sensitive to cathodal stimulation, (positive electrode to negative electrode); teleosts are sensitive to anodal stimulation (negative to positive electrode).[10] The tuberous cells in teleosts play a role in detecting the electric fields and helping the fish to proper orient.
A paper by Hopkins proposes a method for which the fish may navigate. The fish is trying to reduce its alignment "error", meaning it is trying to stay as parallel as it can with the electric field and reduce the angular difference between the electric field and the angle of its body. The equation to describe this orientation is ξh= ϕE-ϕF. ϕE represents the angle of the electric field. ϕF represents the angle of the fish, taken from three body segments as the fish bends. ξh represents the error angle, which is how far off the fish is from being parallel with the field(. When the fish is parallel with the field, ξh =0.[9] Since the electric field does not stay parallel (the fish follows a curve path from positive to negative changes), the fish with stray off the path and have an alignment error ξh. The fish will then adjust β, its bend angle, to turn toward the electric field and reorient itself parallel with the field.[9] A negative β means the fish turns to the right, and a positive β means a fish turns toward the left.
How does the fish know which way to turn? The directionality of the tuberous receptors seems to be controlled by the current flow through the fish's skin. The burst duration coders encode the amplitude by the number of spikes while the Pulse marker receptors encode timing.[6] When the electric field is lined up at the best axis to the receptor, it will produce the strongest signal. This is the case if the electric field is lined up perpendicularly with the receptor.[6] If the fish is lined up with the field, both sides of the fish are stimulated equally in amplitude and the direction of the signals. When the electric field turns to the fish's left, receptors on the left side will be more strongly stimulated with perpendicular signals on the fish's left side while the receptors on the right side would receive a weaker stimulus. The fish will turn to the side of the stronger stimulus. Using the receptors, the fish can detect field and orient itself to the electric field to follow the electric field and locate its source.
Behavior
Sharks
Sharks can detect weak electric fields in their environment using ampullary receptors in to find their prey. A study by Kalmijn et al. 1971 observed this action.[11] Kalmijn used a type of flatfish, called a plaice as a living source of food along with the electrodes and the whiting pieces. He saw that sharks could detect plaice buried under the sand, and would smoothly change direction to where the plaice lay hidden and attack it. To determine if sharks were using electric fields or smell to detect their prey, he devised an experiment. He used the shark species Scyliorhinus canicula and the ray species Raja clavata. He created an agar chamber, which would hold a plaice (small fish), and an opening tube downstream to separate any smells. The agar chamber would enclose the plaice on all sides. The agar does not allow odors to pass, but it does allow electric field to pass through it. Kalmijn would place the plaice in the agar and bury it underneath the sand to eliminate any visual cues. When sharks were in feeding mode, they would locate the agar with the plaice underneath it and attack it near the head region (where bioelectric fields are thought to originate). If chopped up bits of whiting were put in the agar chamber, the shark would try to locate the prey via smell coming out of the outlet tube. Also, if the plaice was put in an agar chamber and covered with polyethylene film, the sharks could no longer detect the plaice.[11] The polyethylene layer inhibits the bioelectric fields and prevents the shark from using passive electrolocation to hunt its prey.[11] One additional behavior observed in sharks is their orientation to the Earth's magnetic field. The Earth's magnetic field acts as a uniform field, B. When the shark swim through the Earth's magnetic field with its own velocity v, the shark induces its own electric current. Sharks swimming though the Earth's magnetic field create a voltage gradient that could act as a physical basis for compass orientation.[3]
Weakly electric fish
Mormyrids and Gymnotiforms are weakly electric fish that also use electric fields to detect and locate other fish. A Gymnotiform fish, Gymnotus carapo, is a very territorial fish, and responds to an intruder by approaching the intruder and making threatening displays that include short discharges and lunging at the intruder.[1] To find the intruder, G. carapo detects the EOD of the intruder, but the electric field emitted by the intruder fish does not point to the source. The fish instead follows the electric field lines to the source or the fish. Hopkins et al. 1997 replicated this behavior by creating a large pool with a number of electrodes on each end.[9] Based on the orientation of the electrodes (radial, tangential, or in 45 degree angles) the fish would swim until it sensed an electric field, and then would reorient itself parallel to an electric field line and swim to the source. Most often their head was facing opposite the flow of the current. Hopkins then took this lab further and rotated the electrodes so the electric field would orient with the rotating electrodes. When the electrode was rotated clockwise, the fish would try to reorient itself and often swim in counterclockwise loops.[9] This behavior is cause by the fish trying to reduce error angle ξ by turning its bend angle β to come in contact with the electric field lines produced by the dipole in another fish. Similar results were also found using a Mormyrid fish, Brienmyrus brachyistius.[8] Catfish in the order Siluriformes have also shown behavioral responses to electric fields or DC poles. They can detect electric fields as low as .75 uV/cm. An early study by Parker and Hersen involving catfish used electrodes helped to determine the fish's response to current. When the electrodes released a current of 1 uA or more, the catfish would avoid the electrodes, and approach the electrode if the current was less than 1 uA and nibble at the area between the electrodes.[3]
See also
- Electric fish
- Electroreception
Notes
- 1 2 3 4 5 6 7 8 Hopkins, C. D. (2005). Passive electrolocation and the sensory guidance of oriented behavior. In T. H. Bullock, C. D. Hopkins, A. R. Popper, & R. R. Fay (Eds.), Electroreception (pp. 264-289). New York: Springer.
- 1 2 3 4 5 6 7 8 9 Wilkens, L. A., & Hofmann, M. H. (2005). Behavior of animals with passive, low-frequency electrosensory Systems. In T. H. Bullock, C. D. Hopkins, A. R. Popper, & R. R. Fay (Eds.), Electroreception (pp. 229-263). New York: Springer.
- 1 2 3 4 5 6 7 8 9 Kalmijn, A. J. (1974). The detection of electric fields from inanimate and animate sources other than electric organs. In A. Fessard (Ed.), Electroreceptors and Other Specialized Receptors in Lower Vertebrates (pp. 148-194). Heidelberg: Springer-Verlag Berlin.
- 1 2 3 4 Albert, J. & Crampton, W. (2006). Electroreception and electrogenesis. In Evans, D.H. & Claiborne, J.B. (Eds) The Physiology of Fishes, 3rd Edition. (pp 431-470). CPC Press. Taylor and Francis Group, Boca Raton, Florida. ISBN 978-0849320224.
- 1 2 3 4 Bodznick, D., & Montgomery, J. C. (2005). The physiology of low-frequency electrosensory systems. In T. H. Bullock, C. D. Hopkins, A. N. Popper, & R. R. Fay (Eds.), Electroreception (pp. 132-153). New York: Springer.
- 1 2 3 Collin, S. P., & Whitehead, D. (2004). The functional roles of passive electroreception in non-electric fishes. Animal Biology, 54(1), 1-25. doi:10.1163/157075604323010024
- 1 2 3 Jorgensen, J. M. (2005). Morphology of electroreceptive sensory organs. In T. H. Bullock, C. D. Hopkins, A. R. Popper, & R. R. Fay (Eds.), Electroreception (pp. 47-67). New York: Springer.
- 1 2 Hopkins, C.D., McKibben, J.R.,& Yager, D.D. (1993). Directional sensitivity of tuberous electroreceptors: polarity preferences and frequency tuning. J. Comp. Physiol. A. 173:415-424
- 1 2 3 4 5 6 7 Hopkins, C. D. (1997). A quantitative analysis of passive electrolocation behavior in electric fish. Brain, 1997(suppl l), 32-59.
- ↑ Crampton, William G. R. (2019). "Electroreception, electrogenesis and electric signal evolution". Journal of Fish Biology. 95 (1): 92–134. doi:10.1111/jfb.13922. ISSN 1095-8649.
- 1 2 3 Kalmijn AJ (1971). "The electric sense of sharks and rays". The Journal of Experimental Biology. 55 (2): 371–83. PMID 5114029.
References
- Albert, J. & Crampton, W.. Electroreception and Electrogenesis (2006). In: The Physiology of Fishes, 3rd Edition. In Evans, D.H. & Claiborne, J.B CPC Press. Taylor and Francis Group, Boca Raton, Florida pp 431–470.
- Bodznick, D., & Montgomery, J. C. (2005). The Physiology of Low-Frequency Electrosensory Systems. In T. H. Bullock, C. D. Hopkins, A. N. Popper, & R. R. Fay (Eds.), Electroreception (pp. 132–153). New York: Springer.
- Collin, S. P., & Whitehead, D. (2004). The functional roles of passive electroreception in non-electric fishes. Animal Biology, 54(1), 1-25. doi:10.1163/157075604323010024
- Hopkins, C. D., McKibben, J.R.,& Yager, D.D. (1993). Directional sensitivity of tuberous electroreceptors: polarity preferences and frequency tuning. J Comp Physiol A. 173:415-424.
- Hopkins, C. D. (1997). A quantitative analysis of passive electrolocation behavior in electric fish. Brain, 1997(suppl l), 32-59.
- Hopkins, C. D. (2005). Passive Electrolocation and the Sensory Guidance of Oriented Behavior. In T. H. Bullock, C. D. Hopkins, A. R. Popper, & R. R. Fay (Eds.), Electroreception (pp. 264–289). New York: Springer.
- Jorgensen, J. M. (2005). Morphology of Electroreceptive Sensory Organs. In T. H. Bullock, C. D. Hopkins, A. R. Popper, & R. R. Fay (Eds.), Electroreception (pp. 47–67). New York: Springer.
- Kalmijn, A. J. (1971). "The electric sense of sharks and rays". The Journal of Experimental Biology. 55 (2): 371–83. PMID 5114029.
- Kalmijn, A. J. (1974). The Detection of Electric Fields from Inanimate and Animate Sources Other than Electric Organs. In A. Fessard (Ed.), Electroreceptors and Other Specialized Receptors in Lower Vertrebrates (pp. 148–194). Heidelberg: Springer-Verlag Berlin.
- Kawasaki, M. (2005). Physiology of Tuberous Electrosensory Systems. In T. H. Bullock, C. D. Hopkins, A. R. Popper, & R. R. Fay (Eds.), Electroreception (pp. 154–194). New York: Springer.
- Schluger, J. H., & Hopkins, C. D. (1987). Electric fish approach stationary signal sources by following electric current lines. Journal of Experimental Biology, 130, 359–367.
- Tricas, T. C.; New, J. G. (1998). "Sensitivity and response dynamics of elasmobranch electrosensory primary afferent neurons to near threshold fields". Journal of Comparative Physiology A. 182 (1): 89–101. doi:10.1007/s003590050161. PMID 9447716.
- Wilkens, L. A., & Hofmann, M. H. (2005). Behavior of Animals with Passive, Low-Frequency Electrosensory Systems. In T. H. Bullock, C. D. Hopkins, A. R. Popper, & R. R. Fay (Eds.), Electroreception (pp. 229–263). New York: Springer.
- Zupanc, G. K. H., & Bullock, T. H. (2005). From Electrogenesis to Electroreception: An Overview. Electroreception (pp. 5–46). New York: Springer Handbook of Auditory Research.
