(Cymene)ruthenium dichloride dimer
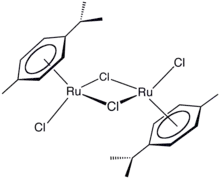
(Cymene)ruthenium dichloride dimer is the organometallic compound with the formula [(cymene)RuCl2]2. This red-coloured, diamagnetic solid is a reagent in organometallic chemistry and homogeneous catalysis. The complex is structurally similar to (benzene)ruthenium dichloride dimer.
 | |
RuCl2_dimer.jpg.webp) | |
| Names | |
|---|---|
| Other names
Dichloro(p-cymene)ruthenium(II) dimer | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.103.371 |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C20H28Cl4Ru2 | |
| Molar mass | 612.38 g·mol−1 |
| Appearance | Red solid |
| Melting point | 247 to 250 °C (477 to 482 °F; 520 to 523 K) (decomposes) |
| Slightly, with hydrolysis | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Preparation and reactions
The dimer is prepared by the reaction of the phellandrene with hydrated ruthenium trichloride.[1] At high temperatures, [(cymene)RuCl2]2 exchanges with other arenes:
- [(cymene)RuCl2]2 + 2 C6Me6 → [(C6Me6)RuCl2]2 + 2 cymene
(Cymene)ruthenium dichloride dimer reacts with Lewis bases to give monometallic adducts:
- [(cymene)RuCl2]2 + 2 PPh3 → 2 (cymene)RuCl2(PPh3)
Such monomers adopt pseudo-octahedral piano-stool structures.
Precursor to catalysts
Treatment of [(cymene)RuCl2]2 with the chelating ligand TsDPENH gives (cymene)Ru(TsDPEN-H), a catalyst for asymmetric transfer hydrogenation.[2]
[(cymene)RuCl2]2 is also used to prepare catalysts (by monomerization with dppf) used in borrowing hydrogen catalysis,[3] a catalytic reaction that is based on the activation of alcohols towards nucleophilic attack.
It can also used to prepare other ruthenium—arene complexes.[4]
References
- Bennett, M. A.; Huang, T. N.; Matheson, T. W.; Smith, A. K. (1982). "16. (η6 -Hexamethylbenzene)Ruthenium Complexes". (η6-Hexamethylbenzene)ruthenium Complexes. Inorganic Syntheses. Vol. 21. pp. 74–78. doi:10.1002/9780470132524.ch16. ISBN 9780470132524.
- Takao Ikariya; Shohei Hashiguchi; Kunihiko Murata; Ryōji Noyori (2005). "Preparation of Optically Active (R,R)-Hydrobenzoin from Benzoin or Benzil". Organic Syntheses: 10.
- Hamid et al.; Advanced Synthesis & Catalysis Volume 349, Issue 10, pages 1555–1575, July 2, 2007; doi:10.1002/adsc.200600638
- Lautens, M., ed. (2001). Category 1, Organometallics: Compounds with Transition Metal—Carbon π-Bonds and Compounds of Groups 10–8 (Ni, Pd, Pt, Co, Rh, Ir, Fe, Ru, Os). Stuttgart: Georg Thieme Verlag. doi:10.1055/sos-sd-001-00899. ISBN 978-3-13-112131-8.