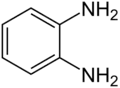
o-Phenylenediamine
 | |||
| |||
 | |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Benzene-1,2-diamine | |||
| Other names
o-Phenylene diamine 1,2-Diaminobenzene 1,2-Phenylenediamine | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.002.210 | ||
| EC Number |
| ||
PubChem CID |
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 1673 | ||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| C6H8N2 | |||
| Molar mass | 108.144 g·mol−1 | ||
| Density | 1.031 g/cm3 | ||
| Melting point | 102 to 104 °C (216 to 219 °F; 375 to 377 K) | ||
| Boiling point | 252 °C (486 °F; 525 K) Other sources: 256 to 258 °C (493 to 496 °F; 529 to 531 K) | ||
| soluble in hot water | |||
| Acidity (pKa) |
| ||
| -71.98·10−6 cm3/mol | |||
| Hazards | |||
| GHS labelling: | |||
    | |||
| Danger | |||
| H301, H312, H317, H319, H332, H341, H351, H410 | |||
| P201, P202, P261, P264, P270, P271, P272, P273, P280, P281, P301+P310, P302+P352, P304+P312, P304+P340, P305+P351+P338, P308+P313, P312, P321, P322, P330, P333+P313, P337+P313, P363, P391, P405, P501 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | 156 °C (313 °F; 429 K) | ||
| Safety data sheet (SDS) | Oxford MSDS | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |||
o-Phenylenediamine (OPD) is an organic compound with the formula C6H4(NH2)2. This aromatic diamine is an important precursor to many heterocyclic compounds. It is isomeric with m-phenylenediamine and p-phenylenediamine.
Preparation
Commonly, 2-nitrochlorobenzene is treated with ammonia and the resulting 2-nitroaniline, whose nitro group is then reduced:[4]
- ClC6H4NO2 + 2 NH3 → H2NC6H4NO2 + NH4Cl
- H2NC6H4NO2 + 3 H2 → H2NC6H4NH2 + 2 H2O
In the laboratory, the reduction of the nitroaniline is effected with zinc powder in ethanol, followed by purification of the diamine as the hydrochloride salt.[5] This compound darkens in air; impurities may be removed by treating a hot aqueous solution with sodium dithionite (reducing agent) and activated carbon, then allowing the product to cool and crystallize.
Reactions and uses
o-Phenylenediamine condenses with ketones and aldehydes to give rise to a variety of useful products. Its reactions with carboxylic acids and their derivatives produce benzimidazoles. The herbicides benomyl and fuberidazole are made in this manner. Thiophanate-methyl is another herbicide produced from o-phenylenediamine.[4]
Quinoxalinedione may be prepared by condensation of o-phenylenediamine with dimethyl oxalate. Condensation with xanthate esters affords mercaptoimidazoles, which are used as antioxidants in rubber products. Treatment with nitrous acid gives benzotriazole, a corrosion inhibitor. Condensation of substituted o-phenylenediamine with various diketones is used in the preparation of a variety of pharmaceuticals.[6]
In coordination chemistry, phenylenediamine is an important ligand precursor. Schiff base derivatives, such as those derived from salicylaldehyde, are excellent chelating ligands. Oxidation of its metal-phenylenediamine complexes affords the diimine derivatives, which are intensely colored and often exist in multiple stable oxidation states.[7]
Safety
With an LD50 of 44 mg/L (in water), o-phenylenediamine is about 1000 times less toxic than the para-isomer. Anilines are typically handled as if they are carcinogenic. For many applications, OPD has been replaced by safer alternatives such as 3,3',5,5'-tetramethylbenzidine.[8]
References
- "DuPont Specialty Intermediates: o-Phenylenediamine (OPD)". Archived from the original on June 22, 2008. Retrieved April 25, 2006.
- Haynes, William M., ed. (2016). CRC Handbook of Chemistry and Physics (97th ed.). CRC Press. p. 5–89. ISBN 978-1498754286.
- "Hazardous Substances Data Bank (HSDB) : 2893 - PubChem". PubChem.
- Smiley, Robert A. (2000). "Phenylene- and Toluenediamines". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a19_405.
- E. L. Martin (1939). "o-Phenylenediamine". Organic Syntheses. 19: 70. doi:10.15227/orgsyn.019.0070. Archived from the original on March 2, 2022.
- See for example, Renault, J.; et al. (1981). "Heterocyclic quinones. Quinoxaline-5,6 and 5,8 diones, potential antitumoral agents". Eur. J. Med. Chem. 16: 545–550.
- Warren, L. F. (1977). "Synthesis of [M'-N4] and [M'-N6] Complexes Based on o-Benzoquinone Diimine with Cobalt, Iron, and Ruthenium". Inorg. Chem. 16 (11): 2814–2819. doi:10.1021/ic50177a028.
- Deshpande SS (1996). Enzyme Immunoassays: From Concept to Product Development. New York: Chapman & Hall. p. 169. ISBN 978-0-412-05601-7.