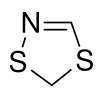
1,4,2-Dithiazole
1,4,2-Dithiazole is a heterocyclic compound consisting of an unsaturated five-membered ring containing two carbon atoms, one nitrogen atom, and two sulfur atoms. 1,4,2-Dithiazole compounds may be formed by the reaction of nitrile sulfide (formed by the thermolysis of oxathiazolone) with various reactive species;[1] for instance thiocarbonyls via a 1,3-dipolar cycloaddition reaction.[2] These compounds may be protonated by strong acids to give synthetically useful aromatic cations.[3]
 | |
| Names | |
|---|---|
| Preferred IUPAC name
5H-1,4,2-Dithiazole | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
| UNII | |
| |
| |
| Properties | |
| C2H3NS2 | |
| Molar mass | 105.17 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
References
- Argyropoulos, Nikolaos G. (1996). "1,4-Oxa/thia-2-azoles". Comprehensive Heterocyclic Chemistry II: 491–543. doi:10.1016/B978-008096518-5.00092-7. ISBN 978-0-08-096518-5.
- Wai, Kwok-Fai; Sammes, Michael P. (1991). "Dithiazoles and related compounds. Part 3. Preparation of 5H-1,4,2-dithiazoles via 1,3-dipolar cycloadditions between nitrile sulphides and thiocarbonyl compounds, and some conversions into 3,5-diaryl-1,4,2-dithiazolium salts". Journal of the Chemical Society, Perkin Transactions 1 (1): 183. doi:10.1039/p19910000183. ISSN 0300-922X.
- Wai, Kwok-Fai; Sammes, Michael P. (1992). "Dithiazoles and related compounds. Part. 4. Preparation of 1,4,2-dithiazolium salts unsubstituted at C-5 including the parent heterocycle, NMR spectroscopic evidence for aromaticity, and some novel reactions". Journal of the Chemical Society, Perkin Transactions 1 (16): 2065. doi:10.1039/p19920002065. ISSN 0300-922X.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.