1-Aminoethanol
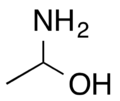
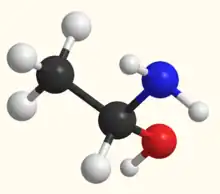
1-Aminoethanol is an organic compound with the formula CH3CH(NH2)OH. It is classified as an alkanolamine. Specifically, it is a structural isomer of 2-aminoethanol (ethanolamine). These two compounds differ in the position of the amino group. Since the central carbon atom in 1-aminoethanol has four different substituents, the compound has two stereoisomers. Unlike 2-aminoethanol, which is of considerable importance in commerce, 1-aminoethanol is not encountered as a pure material and is mainly of theoretical interest.
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
1-Aminoethan-1-ol | |
| Other names
1-Aminoethanol Acetaldehyde ammonia | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.000.790 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C2H7NO | |
| Molar mass | 61.084 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
1-Aminoethanol exists in a solution of acetaldehyde and aqueous ammonia.[1]
1-Aminoethanol is suggested as intermediate in Strecker reaction of alanine synthesis.[2]
1-Aminoethanol was first prepared in 1833 by the German chemist Johann Wolfgang Döbereiner; its empirical formula was first determined by the German chemist Justus von Liebig in 1835.[3] The structure of 1-aminoethanol remained unproven until 1877, when the German-Italian chemist Robert Schiff showed that the structure was CH3CH(OH)NH2.[4]
References
- Smith, Michael B.; March, Jerry (2007), Advanced Organic Chemistry: Reactions, Mechanisms, and Structure (6th ed.), New York: Wiley-Interscience, ISBN 978-0-471-72091-1
- Shinichi Yamabe; Guixiang Zeng; Wei Guan; Shigeyoshi Sakaki (2014). "Proton transfers in the Strecker reaction revealed by DFT calculations". Beilstein Journal of Organic Chemistry. 10: 1765–1774. doi:10.3762/bjoc.10.184. PMC 4143099. PMID 25161735.
- Liebig, Justus (1835) "Ueber die Producte der Oxydation des Alkohols" (On the products of oxidation of alcohol [i.e., ethanol]), Annalen der Chemie, 14 : 133–167. In 1821, Döbereiner had prepared what he called Saüerstoffäther (literally: ether oxide), which, as Liebig later showed, contained acetaldehyde. On p. 134, Liebig states that Döbereiner first produced 1-amino-ethanol by perfusing acetaldehyde (Saüerstoffäther or Aldehyd, as Liebig called it) with ammonia gas (Ammoniak) : "Der berühmte Entdecker des Acetals theilte mir damals gleichzeitig mit, dass sein Saüerstoffäther mit Ammoniakgas eine weisse krystallinische Verbingdung bilde; ... " (The famous discoverer of acetal [viz, Döbereiner] simultaneously communicated to me at that time that his ether oxide forms a white crystalline compound with ammonia gas; ... ) On p. 146, Liebig presented his empirical formula for 1-aminoethanol: C4H14N2O2. If the subscripts are divided by 2, the resulting formula is correct.
- Although it had long been assumed that the structure of 1-aminoethanol was CH3CH(OH)NH2, it had not been proven that 1-aminoethanol contained a hydroxyl (-OH) group until Schiff's investigation in 1877. See:
- Schiff, Robert (1877) "Zur Constitution des Chloralammoniaks und des Aldehydammoniaks," Berichte der Deutschen Chemischen Gesellschaft, 10 : 165–171.
- Aschan, Ossian (1915) "Neues über Aldehyd-ammoniak und Chloral-ammoniak." Berichte der Deutschen Chemischen Gesellschaft, 48 (1) : 874–891.