2-Bromobutane
2-Bromobutane is an isomer of 1-bromobutane. Both compounds share the molecular formula C4H9Br. 2-Bromobutane is also known as sec-butyl bromide or methylethylbromomethane. Because it contains bromine, a halogen, it is part of a larger class of compounds known as alkyl halides. It is a colorless liquid with a pleasant odor. Because the carbon atom connected to the bromine is connected to two other carbons the molecule is referred to as a secondary alkyl halide. 2-Bromobutane is chiral and thus can be obtained as either of two enantiomers designated as (R)-(−)-2-bromobutane and (S)-(+)-2-bromobutane.
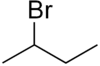 | |
| Names | |
|---|---|
| Preferred IUPAC name
2-Bromobutane[1] | |
| Other names
sec-Butylbromide | |
| Identifiers | |
3D model (JSmol) |
|
| 505949 | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.001.037 |
| EC Number |
|
| MeSH | 2-bromobutane |
PubChem CID |
|
| RTECS number |
|
| UNII | |
| UN number | 2339 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C4H9Br | |
| Molar mass | 137.020 g·mol−1 |
| Appearance | Colourless liquid |
| Density | 1.255 g mL−1 |
| Melting point | −112.65 °C; −170.77 °F; 160.50 K |
| Boiling point | 91 °C; 196 °F; 364 K |
| log P | 2.672 |
Refractive index (nD) |
1.437 |
| Thermochemistry | |
Std enthalpy of formation (ΔfH⦵298) |
−156 kJ mol−1 |
Std enthalpy of combustion (ΔcH⦵298) |
−2.706–−2.704 MJ mol−1 |
| Hazards | |
| GHS labelling: | |
 | |
| Danger | |
| H225 | |
| P210 | |
| Flash point | 21 °C (70 °F; 294 K) |
| Related compounds | |
Related alkanes |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
2-Bromobutane is relatively stable, but is toxic and flammable. When treated with a strong base, it is prone to undergo an E2 reaction, which is a bimolecular elimination reaction, resulting in (predominantly) 2-butene, an alkene (double bond). 2-Bromobutane is an irritant, and harmful if ingested. It can irritate and burn skin and eyes.
References
- "2-bromobutane - Compound Summary". PubChem Compound. USA: National Center for Biotechnology Information. 26 March 2005. Retrieved 17 June 2012.