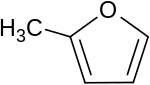
2-Methylfuran
2-Methylfuran, also known with the older name of sylvane, is a flammable, water-insoluble liquid[3] with a chocolate odor, found naturally in Myrtle and Dutch Lavender[4] used as a FEMA GRAS flavoring substance,[5] with the potential for use in alternative fuels.
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
2-Methylfuran | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.007.814 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C5H6O | |
| Molar mass | 82.10 g/mol |
| Appearance | Colorless to pale yellow/green liquid |
| Density | 0.91546 g/mL (20 °C) [1] |
| Boiling point | 64[2][1] °C (147 °F; 337 K) |
| 3000 mg/L (20 °C) | |
| Solubility in ethanol | Soluble |
Refractive index (nD) |
1.4332 (20 °C) [1] |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
Very flammable, harmful |
| NFPA 704 (fire diamond) | |
| Flash point | −22 °C; −8 °F; 251 K |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Manufacture
2-Methylfuran is an article of commerce (chemical intermediate) and is normally manufactured by catalytic hydrogenolysis of furfural alcohol or via a hydrogenation-hydrogenolysis sequence from furfural in the vapor phase.[6]
See also
References
- Baird, Zachariah Steven; Uusi-Kyyny, Petri; Pokki, Juha-Pekka; Pedegert, Emilie; Alopaeus, Ville (6 Nov 2019). "Vapor Pressures, Densities, and PC-SAFT Parameters for 11 Bio-compounds". International Journal of Thermophysics. 40 (11): 102. Bibcode:2019IJT....40..102B. doi:10.1007/s10765-019-2570-9.
- NIST Chemistry WebBook. http://webbook.nist.gov
- Kenneth Barbalace. "Chemical Database - 2-Methylfuran. EnvironmentalChemistry.com. 1995 - 2008. Accessed on-line: 8/26/2008". Retrieved 2008-08-26.
- Jim Duke. "Dr. Duke's Phytochemical and Ethnobotanical Databases. [Online Database] 26 August 2008. 2-METHYL-FURAN". Archived from the original on 2016-11-18. Retrieved 2008-08-26.
- "2-methyl furan". The Good Scents Company. Retrieved 2008-08-26.
- L. W. Burnette, et al., "Production of 2-Methylfuran by Vapor Phase Hydrogenation of Furfural" Industrial and engineering Chemistry, V40, P502-505 (1948).
External links
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.
