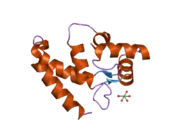Allograft inflammatory factor 1
Allograft inflammatory factor 1 (AIF-1) also known as ionized calcium-binding adapter molecule 1 (IBA1) is a protein that in humans is encoded by the AIF1 gene.[5][6]
Gene
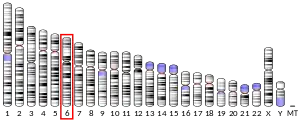
The AIF1 gene is located within a segment of the major histocompatibility complex class III region. It has been shown that this gene is highly expressed in testis, spleen, and brain but weakly expressed in lung, and kidney. Among brain cells, the Iba1 gene is strongly and specifically expressed in microglia. Circulating macrophages also express Iba1.
Function
AIF1 is a protein that exists in the cytoplasm, and it is highly evolutionarily conserved. It is also possibly identical to three other proteins, Iba-2, MRF-1 (microglia response factor) and daintain. However complete functional profiles of all three proteins and how they overlap is unknown.[7] IBA1 is a 17-kDa EF hand protein that is specifically expressed in macrophages / microglia and is upregulated during the activation of these cells. Iba1 expression is up-regulated in microglia following nerve injury,[8] central nervous system ischemia, and several other brain diseases.
AIF1 was originally discovered in atherosclerotic lesions in a rat model of chronic allograft cardiac rejection. It was found to be upregulated in macrophages and neutrophils in response to the cytokine IFN-γ.[9] AIF1 expression has been seen to increase in vascular tissue in response to arterial injury, specifically it is found in activated vascular smooth muscle cells in response to IFN-γ, IL-1β, and T-cell conditioned media.[10] In vascular smooth muscle cells, activation is responsible for arterial thickening in allografts through over proliferation. AIF1 has been found to enhance growth and promote proliferation in vascular smooth muscle cells through deregulation of the cell cycle. It does this by shortening the cell cycle and altering the expression of cyclins.[11] Though histologically different, AIF1 has also been shown to promote the proliferation and activation of endothelial cells (EC). EC activation, leads to proliferation and migration of cells, which is involved in multiple normal vascular processes, such as atherosclerosis, angiogenesis, and wound healing. It is currently theorized that AIF1 works to control endothelial cell proliferation and migration through action in signal transduction pathways.[12] It has features of a cytoplasmic signaling protein, including several domains that allow for binding to multiprotein complexes, called PDZ domains.[13] In endothelial cells, AIF1 has been specifically shown to regulate vasculogenesis, including the formation of aortic sprouting and tube-like formations. AIF1 been shown to interact with kinase p44/42 and PAK1, two previously known signal transduction molecules, in regulating these processes. AIF1 also shows distinct differences in the pathways by which it regulates endothelial cells, macrophages, and vascular smooth muscle cells.[12] Upregulation of AIF-1 is connected with increased migration of mononuclear peripheral blood cell. In the CD14 positive cells, AIF-1 support secretion of IL-6 and various chemokines.[14] AIF-1 may also play a role in the T-cell response. It has been shown that AIF-1 increases expression of IL-2 and IFN-γ in T-cells, while the expression of IL-4 and TGF-β is decreased. The presence of AIF-1 also inhibits polarization into regulatory T cells.[15]
Clinical significance
Allograft Inflammatory Factor 1 is found in activated macrophages. Activated macrophages are found in tissues with inflammation. AIF1 levels in healthy humans have been found to positively correlate with metabolic indicators, such as body mass index, triglycerides, and fasting plasma glucose levels. The excess of adipose tissue found in obese patients is found to cause chronic inflammation with an increase in the number of activated macrophages. Subsequently, AIF1 may be an accurate indicator of macrophage activation in the body.[16] There is also evidence that AIF1 could be a marker for diabetic nephropathy when detected in serum.[17] Since diabetic nephropathy is a consequence of long-term type 1 and type 2 diabetes, this consistent with evidence that AIF1 may be associated with other aspects of diabetes. It is found in activated macrophages in the pancreatic islets, and has been shown to decrease insulin secretion, while simultaneously impairing glucose elimination.[18]
Role in cancer progression
In recent years, the possibility of a role for AIF-1 in cancer development has also been considered. Significantly higher levels of AIF-1 expression were found in hepatocarcinoma cell lines and in tissue compared to healthy samples. One option in which AIF-1 may contribute to the development of pathology is involvement in the proliferation and migration of tumor cells.[19] It was also shown that AIF-1 promote cell proliferation in the brest cancer cells line. This effect was dependent on time and level of AIF-1 protein.[20] Upregulation of AIF-1 enhanced activity of NF-ΚB and increased expression of cyclin D1. Cyclin D1 contribute to cell proliferation and mutation in this gene has been connected with variety of tumors. It has also been shown that AIF-1 expression can contribute to progression of cancer by inhibition of apoptosis in cells.[21][22]
Role in Rheumatoid arthritis
The role of up-regulation of expression of AIF-1 was demonstrated in rheumatoid arthritis. Presence of AIF-1 was confirmed in synovial tissue of patient with this pathology. AIF-1 was strongly expressed in several cell types of synovial tissue, such as fibroblast and synovial cells, but also in infiltrated immune cells. It was also shown that upregulation of AIF-1 contribute to induction of enhances the production of IL-6 . Another factor by which the expression of AIF-1 affects the course of the disease is increased proliferation of synovial cells.[23]
Role in kidney diseases
AIF-1 was also considered as a player in the diseases connected with fibrosis. For example, in kidney diseases overexpression of AIF-1 in the macrophages contributes to signaling through AKT and mTOR. Another way in which AIF-1 contribute to the pathology of kidney is upregulation of enzyme NADPH oxidase 2. This upregulation leads to oxidative stress in the cells and progression of renal injury.[24] Expression of AIF-1 is considered to be linked with calcification in hemodyalisis patients. Higher presence of AIF-1/NF-κB/MCP-1/CCR-2-pathway was detected in calcifaied vascular smooth muscle cells. AIF-1 was also detected as a potential factor which contribue to apoptosis and inflammation.[25]
Role in retinal diseases
Since the immune response in the retina is tightly regulated under physiological conditions, microglia may play a role in retinal diseases.[26] Retinitis pigmentosa is an inherited disease in which photoreceptors are gradually degenerated. That condition gradually leads to reduced of dark vision and eventually complete blindness. In an experimental model RCS (The Royal College of Surgeons) rats with progression loss of photoreceptors, level of AIF-1 was elevated in retina in contrast to wild type[27]
References
- ENSG00000235985, ENSG00000235588, ENSG00000234836, ENSG00000237727, ENSG00000204472 GRCh38: Ensembl release 89: ENSG00000206428, ENSG00000235985, ENSG00000235588, ENSG00000234836, ENSG00000237727, ENSG00000204472 - Ensembl, May 2017
- GRCm38: Ensembl release 89: ENSMUSG00000024397 - Ensembl, May 2017
- "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- Autieri MV (November 1996). "cDNA cloning of human allograft inflammatory factor-1: tissue distribution, cytokine induction, and mRNA expression in injured rat carotid arteries". Biochemical and Biophysical Research Communications. 228 (1): 29–37. doi:10.1006/bbrc.1996.1612. PMID 8912632.
- "Entrez Gene: AIF1 allograft inflammatory factor 1".
- Deininger MH, Meyermann R, Schluesener HJ (March 2002). "The allograft inflammatory factor-1 family of proteins". FEBS Letters. 514 (2–3): 115–21. doi:10.1016/S0014-5793(02)02430-4. PMID 11943136. S2CID 34086133.
- Ito D, Imai Y, Ohsawa K, Nakajima K, Fukuuchi Y, Kohsaka S (June 1998). "Microglia-specific localisation of a novel calcium binding protein, Iba1". Brain Research. Molecular Brain Research. 57 (1): 1–9. doi:10.1016/s0169-328x(98)00040-0. PMID 9630473.
- Utans U, Arceci RJ, Yamashita Y, Russell ME (June 1995). "Cloning and characterization of allograft inflammatory factor-1: a novel macrophage factor identified in rat cardiac allografts with chronic rejection". The Journal of Clinical Investigation. 95 (6): 2954–62. doi:10.1172/JCI118003. PMC 295984. PMID 7769138.
- Autieri MV, Carbone C, Mu A (July 2000). "Expression of allograft inflammatory factor-1 is a marker of activated human vascular smooth muscle cells and arterial injury". Arteriosclerosis, Thrombosis, and Vascular Biology. 20 (7): 1737–44. doi:10.1161/01.ATV.20.7.1737. PMID 10894811.
- Autieri MV, Carbone CM (September 2001). "Overexpression of allograft inflammatory factor-1 promotes proliferation of vascular smooth muscle cells by cell cycle deregulation". Arteriosclerosis, Thrombosis, and Vascular Biology. 21 (9): 1421–6. doi:10.1161/hq0901.095566. PMID 11557666.
- Tian Y, Jain S, Kelemen SE, Autieri MV (February 2009). "AIF-1 expression regulates endothelial cell activation, signal transduction, and vasculogenesis". American Journal of Physiology. Cell Physiology. 296 (2): C256-66. doi:10.1152/ajpcell.00325.2008. PMC 2643850. PMID 18787073.
- Hung AY, Sheng M (February 2002). "PDZ domains: structural modules for protein complex assembly". The Journal of Biological Chemistry. 277 (8): 5699–702. doi:10.1074/jbc.R100065200. PMID 11741967.
- Kadoya M, Yamamoto A, Hamaguchi M, Obayashi H, Mizushima K, Ohta M, et al. (June 2014). "Allograft inflammatory factor-1 stimulates chemokine production and induces chemotaxis in human peripheral blood mononuclear cells". Biochemical and Biophysical Research Communications. 448 (3): 287–91. doi:10.1016/j.bbrc.2014.04.106. PMID 24796669.
- Cano-Martínez D, Monserrat J, Hernández-Breijo B, Sanmartín Salinas P, Álvarez-Mon M, Val Toledo-Lobo M, Guijarro LG (February 2020). "Extracellular allograft inflammatory factor-1 (AIF-1) potentiates Th1 cell differentiation and inhibits Treg response in human peripheral blood mononuclear cells from normal subjects". Human Immunology. 81 (2–3): 91–100. doi:10.1016/j.humimm.2020.01.011. PMID 32057519. S2CID 211112950.
- Fukui M, Tanaka M, Toda H, Asano M, Yamazaki M, Hasegawa G, et al. (July 2012). "The serum concentration of allograft inflammatory factor-1 is correlated with metabolic parameters in healthy subjects". Metabolism. 61 (7): 1021–5. doi:10.1016/j.metabol.2011.12.001. PMID 22225958.
- Fukui M, Tanaka M, Asano M, Yamazaki M, Hasegawa G, Imai S, et al. (July 2012). "Serum allograft inflammatory factor-1 is a novel marker for diabetic nephropathy". Diabetes Research and Clinical Practice. 97 (1): 146–50. doi:10.1016/j.diabres.2012.04.009. PMID 22560794.
- Chen ZW, Ahren B, Ostenson CG, Cintra A, Bergman T, Möller C, et al. (December 1997). "Identification, isolation, and characterization of daintain (allograft inflammatory factor 1), a macrophage polypeptide with effects on insulin secretion and abundantly present in the pancreas of prediabetic BB rats". Proceedings of the National Academy of Sciences of the United States of America. 94 (25): 13879–84. Bibcode:1997PNAS...9413879C. doi:10.1073/pnas.94.25.13879. PMC 28401. PMID 9391121.
- Zhang Q, Sun S, Zhu C, Xie F, Cai Q, Sun H, et al. (September 2018). "Expression of Allograft Inflammatory Factor-1 (AIF-1) in Hepatocellular Carcinoma". Medical Science Monitor. 24: 6218–6228. doi:10.12659/MSM.908510. PMC 6139115. PMID 30188879.
- Yu Z, Song YB, Cui Y, Fu AQ (July 2019). "Effects of AIF-1 inflammatory factors on the regulation of proliferation of breast cancer cells". Journal of Biological Regulators and Homeostatic Agents. 33 (4): 1085–1095. PMID 31389223.
- Jia S, Chaibou MA, Chen Z (2012-12-23). "Daintain/AIF-1 reinforces the resistance of breast cancer cells to cisplatin". Bioscience, Biotechnology, and Biochemistry. 76 (12): 2338–41. doi:10.1271/bbb.120577. PMID 23221708. S2CID 883243.
- Liu S, Tan WY, Chen QR, Chen XP, Fu K, Zhao YY, Chen ZW (May 2008). "Daintain/AIF-1 promotes breast cancer proliferation via activation of the NF-kappaB/cyclin D1 pathway and facilitates tumor growth". Cancer Science. 99 (5): 952–7. doi:10.1111/j.1349-7006.2008.00787.x. PMID 18341653.
- Kimura M, Kawahito Y, Obayashi H, Ohta M, Hara H, Adachi T, et al. (March 2007). "A critical role for allograft inflammatory factor-1 in the pathogenesis of rheumatoid arthritis". Journal of Immunology. 178 (5): 3316–22. doi:10.4049/jimmunol.178.5.3316. PMID 17312183.
- Yuan X, Wang X, Li Y, Li X, Zhang S, Hao L (November 2019). "Aldosterone promotes renal interstitial fibrosis via the AIF‑1/AKT/mTOR signaling pathway". Molecular Medicine Reports. 20 (5): 4033–4044. doi:10.3892/mmr.2019.10680. PMC 6797939. PMID 31545432.
- Hao J, Tang J, Zhang L, Li X, Hao L (2020-12-04). Muntean D (ed.). "The Crosstalk between Calcium Ions and Aldosterone Contributes to Inflammation, Apoptosis, and Calcification of VSMC via the AIF-1/NF-κB Pathway in Uremia". Oxidative Medicine and Cellular Longevity. 2020: 3431597. doi:10.1155/2020/3431597. PMC 7732390. PMID 33343805.
- Okunuki Y, Mukai R, Nakao T, Tabor SJ, Butovsky O, Dana R, et al. (May 2019). "Retinal microglia initiate neuroinflammation in ocular autoimmunity". Proceedings of the National Academy of Sciences of the United States of America. 116 (20): 9989–9998. Bibcode:2019PNAS..116.9989O. doi:10.1073/pnas.1820387116. PMC 6525481. PMID 31023885.
- Lew DS, Mazzoni F, Finnemann SC (2020). "Microglia Inhibition Delays Retinal Degeneration Due to MerTK Phagocytosis Receptor Deficiency". Frontiers in Immunology. 11: 1463. doi:10.3389/fimmu.2020.01463. PMC 7381113. PMID 32765507.
Further reading
- Deininger MH, Meyermann R, Schluesener HJ (March 2002). "The allograft inflammatory factor-1 family of proteins". FEBS Letters. 514 (2–3): 115–21. doi:10.1016/S0014-5793(02)02430-4. PMID 11943136. S2CID 34086133.
- Utans U, Arceci RJ, Yamashita Y, Russell ME (June 1995). "Cloning and characterization of allograft inflammatory factor-1: a novel macrophage factor identified in rat cardiac allografts with chronic rejection". The Journal of Clinical Investigation. 95 (6): 2954–62. doi:10.1172/JCI118003. PMC 295984. PMID 7769138.
- Iris FJ, Bougueleret L, Prieur S, Caterina D, Primas G, Perrot V, et al. (February 1993). "Dense Alu clustering and a potential new member of the NF kappa B family within a 90 kilobase HLA class III segment". Nature Genetics. 3 (2): 137–45. doi:10.1038/ng0293-137. PMID 8499947. S2CID 33041319.
- Utans U, Quist WC, McManus BM, Wilson JE, Arceci RJ, Wallace AF, Russell ME (May 1996). "Allograft inflammatory factory-1. A cytokine-responsive macrophage molecule expressed in transplanted human hearts". Transplantation. 61 (9): 1387–92. doi:10.1097/00007890-199605150-00018. PMID 8629302.
- Autieri MV, Agrawal N (June 1998). "IRT-1, a novel interferon-gamma-responsive transcript encoding a growth-suppressing basic leucine zipper protein". The Journal of Biological Chemistry. 273 (24): 14731–7. doi:10.1074/jbc.273.24.14731. PMID 9614071.
- Neville MJ, Campbell RD (April 1999). "A new member of the Ig superfamily and a V-ATPase G subunit are among the predicted products of novel genes close to the TNF locus in the human MHC". Journal of Immunology. 162 (8): 4745–54. PMID 10202016.
- Sasaki Y, Ohsawa K, Kanazawa H, Kohsaka S, Imai Y (August 2001). "Iba1 is an actin-cross-linking protein in macrophages/microglia". Biochemical and Biophysical Research Communications. 286 (2): 292–7. doi:10.1006/bbrc.2001.5388. PMID 11500035.
- Autieri MV, Kelemen SE, Wendt KW (May 2003). "AIF-1 is an actin-polymerizing and Rac1-activating protein that promotes vascular smooth muscle cell migration". Circulation Research. 92 (10): 1107–14. doi:10.1161/01.RES.0000074000.03562.CC. PMID 12714565.
- Deininger MH, Weinschenk T, Meyermann R, Schluesener HJ (August 2003). "The allograft inflammatory factor-1 in Creutzfeldt-Jakob disease brains". Neuropathology and Applied Neurobiology. 29 (4): 389–99. doi:10.1046/j.1365-2990.2003.00476.x. PMID 12887599. S2CID 21401126.
- Xie T, Rowen L, Aguado B, Ahearn ME, Madan A, Qin S, et al. (December 2003). "Analysis of the gene-dense major histocompatibility complex class III region and its comparison to mouse". Genome Research. 13 (12): 2621–36. doi:10.1101/gr.1736803. PMC 403804. PMID 14656967.
- Ohsawa K, Imai Y, Sasaki Y, Kohsaka S (February 2004). "Microglia/macrophage-specific protein Iba1 binds to fimbrin and enhances its actin-bundling activity". Journal of Neurochemistry. 88 (4): 844–56. doi:10.1046/j.1471-4159.2003.02213.x. PMID 14756805. S2CID 25092822.
- Chen X, Kelemen SE, Autieri MV (July 2004). "AIF-1 expression modulates proliferation of human vascular smooth muscle cells by autocrine expression of G-CSF". Arteriosclerosis, Thrombosis, and Vascular Biology. 24 (7): 1217–22. doi:10.1161/01.ATV.0000130024.50058.de. PMID 15117732.
- Arvanitis DA, Flouris GA, Spandidos DA (2005). "Genomic rearrangements on VCAM1, SELE, APEG1and AIF1 loci in atherosclerosis". Journal of Cellular and Molecular Medicine. 9 (1): 153–9. doi:10.1111/j.1582-4934.2005.tb00345.x. PMC 6741330. PMID 15784173.
- Autieri MV, Chen X (July 2005). "The ability of AIF-1 to activate human vascular smooth muscle cells is lost by mutations in the EF-hand calcium-binding region". Experimental Cell Research. 307 (1): 204–11. doi:10.1016/j.yexcr.2005.03.002. PMID 15922740.
External links
- Human AIF1 genome location and AIF1 gene details page in the UCSC Genome Browser.
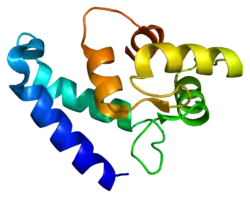
- PDBe-KB provides an overview of all the structure information available in the PDB for Human Allograft inflammatory factor 1