Apoplast
The apoplast is the extracellular space outside of plant cell membranes, especially the fluid-filled cell walls of adjacent cells where water and dissolved material can flow and diffuse freely. Fluid and material flows occurring in any extracellular space are called apoplastic flow or apoplastic transport. The apoplastic pathway is one route by which water and solutes are transported and distributed to different places through tissues and organs, contrasting with the symplastic pathway.
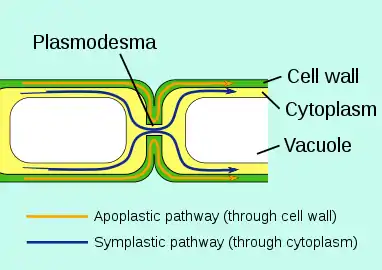
To prevent uncontrolled leakage to unwanted places, in certain areas there are barriers to the apoplastic flow: in roots the Casparian strip has this function Outside the plant epidermis of aerial plant parts is a protective waxy film called plant cuticle that protects against drying out, but also waterproofs the plant against external water.
The apoplast is important for all the plant's interaction with its environment: The main carbon source (carbon dioxide) needs to be solubilized, which happens in the apoplast, before it diffuses through the cell wall and across the plasma membrane, into the cell's inner content, the cytoplasm, where it diffuses in the symplast to the chloroplasts for photosynthesis. In the roots, ions diffuse into the apoplast of the epidermis before diffusing into the symplast, or in some cases being taken up by specific ion channels, and being pulled by the plant's transpiration stream, which also occurs completely within the boundaries of the apoplast. Similarly, all gaseous molecules emitted and received by plants such as oxygen must pass through the apoplast.
In nitrate poor soils, acidification of the apoplast increases cell wall extensibility and root growth rate. This is believed to be caused by a decrease in nitrate uptake (due to deficit in the soil medium) and supplanted with an increase in chloride uptake. H+ATPase increases the efflux of H+, thus acidifying the apoplast. [1]
The apoplast is a site for cell-to-cell communication. During local oxidative stress, hydrogen peroxide and superoxide anions can diffuse through the apoplast and transport a warning signal to neighbouring cells. In addition, a local alkalinization of the apoplast due to such stress can travel within minutes to the rest of the plant body via the xylem and trigger systemic acquired resistance.[2]
The apoplast also plays an important role in resistance to aluminium toxicity.
In addition to resistance to chemicals, the apoplast provides the rich environment for microorganisms endophytes which arises[??] the abiotic resistance of plants. [3] Exclusion of aluminium ions in the apoplast prevent toxic levels which inhibit shoot growth, reducing[?] crop yields.[4]
History
The term apoplast was coined in 1930 by Münch in order to separate the "living" symplast from the "dead" apoplast.[5][6]
Apoplastic transport
The apoplastic pathway is one of the two main pathways for water transport in plants, the other being symplastic pathway. In the root via the apoplast water and minerals flow in an upward direction to the xylem.[7]
The concentration of solutes transported through the apoplast in aboveground organs is established through a combination of import from the xylem, absorption by cells, and export by the phloem.[8]
Transport velocity is higher (transport is faster) in the apoplast than in the symplast.[9] This method of transport also accounts for a higher proportion of water transport in plant tissues than does symplastic transport.[10]
The apoplastic pathway is also involved in passive exclusion. Some of the ions that enter through the roots do not make it to the xylem. The ions are excluded by the cell walls (plasma membranes) of the endodermal cells.[11]
Apoplastic colonization
It is well known that the apoplast in plants’ tissues contains rich mineral nutrients, and it becomes the main factor for microorganisms to thrive at the apoplast. Even though there are apoplastic immunity systems, but there are pathogens that have effectors that can modulate the host immunity or suppress the immunity responses as known as effector-triggered susceptibility.[12] Another factor that pathogens colonize the apoplastic space so frequent is because when they enter the plants from leaves, the first place they come across is the apoplastic space.[13] Therefore, the apoplast is a popular biotic interface and also a reservoir for microbes. One of the common apoplastic disease appear in plants without restricted habitat or climate is black rot, caused by the gram-negative bacteria Xanthomonas campestris.
Entophytic bacteria can cause severe problems in agriculture in a way of inhibiting plant growth by alkalizing the apoplast with their volatiles. In especially, the rhizobacteria has been found that its major component of the volatiles are phytotoxic, it is identified as 2-phenylethanol. 2-phenylethanol can influence the regulation of WRKY18 which is a transcription factor that engages in multiple plant hormones, one of them is abscisic acid (ABA) hormone.[14] 2-phyenlethanol modulates the sensitivity of ABA through WRKY18 and WRKY40, but WRKY18 is the central mediator of the pathway of triggering cell death and modulation of ABA sensitivity influenced by 2-phyenlethanol.[15] Therefore, it results in the inhibition of root growth, and the plants have no capacity to grow without having the roots absorb nutrients in soils.
However, the microbial colonization in the apoplast is not always harmful to the plants, indeed, it can be beneficial to establish a symbiotic relationship with the host. One of the examples is the endophytic and phyllosphere microbes can indirectly promote plant growth and protect the plant from other pathogens by inducing salicylic acid (SA)and jasmonic acid (JA) signaling pathways, and they are both parts of the pathogen associated molecular patterns triggered immunity (PTI). The productions of SA and JA hormones also modulate the ABA signaling to be the components on the defense gene expression, and there are a lot more responses with the involvement of other hormones to respond to different biotic and abiotic stress. In the experiment performed by Romero et al., they inoculated the known entophytic bacteria, Xanthomonas into Canola, a plant that grows in multiple habitats, and it is found its apoplastic fluids that are 99% identity to another bacteria, Pseudomonas viridiflava, by performing 16S rRNA sequences with the Genebank and reference strains. They further used the markers on the SA-responsive transcriptional factor and other specific genes such as lipoxygenase 3 as marker genes for JA signaling and ABA signaling to perform quantitative reverse-transcription PCR. It has shown Xanthomonas only activates the related gene of SA pathway, in comparison, Pseudomonas viridiflava is able to trigger the genes of both SA and JA pathway, which suggest Pseudomonas viridiflava originally in Canola can stimulate PTI by the accumulation of both signaling pathway to inhibit the growth of Xanthomonas[16]. In conclusion, the apoplast acts as a crucial role in plants, involving in all kinds of regulations of hormone and transportation of nutrients, so once it has been colonized, the effect it brings cannot be neglected.
Notes
- Apoplast was previously defined as "everything but the symplast, consisting of cell walls and spaces between cells in which water and solutes can move freely". However, since solutes can neither freely move through the air spaces between plant cells nor through the cuticle, this definition has been changed. When referring to "everything outside the plasma membrane", the term "extracellular space" is in use.
- The word apoplasm is also in use with similar meaning as apoplast, although less common.
References
- Skobelev, O (July 2010). "Accelerated root growth induced by nitrate deficit is related to apoplast acidification". Russian Journal of Plant Physiology. 57 (4): 489. ISSN 1021-4437.
- H. H. Felle; A. Herrmann; R. Hückelhoven; K.-H. Kogel (December 2005). "Root-to-shoot signalling: apoplastic alkalinization, a general stress response and defence factor in barley (Hordeum vulgare)". Protoplasma. 227 (1): 17–24. doi:10.1007/s00709-005-0131-5. PMID 16389490. S2CID 5017915.
- Sattelmacher, Burkhard (February 2001). "The apoplast and its significance for plant mineral nutrition". New Phytologist. 149 (2): 167–192. doi:10.1046/j.1469-8137.2001.00034.x. ISSN 0028-646X. PMID 33874640. S2CID 86799768.
- Horst, Walter J. (1995). "The role of the apoplast in aluminium toxicity and resistance of higher plants: A review". Zeitschrift für Pflanzenernährung und Bodenkunde. 158 (5): 419–428. doi:10.1002/jpln.19951580503.
- Münch, E (1930). Die Stoffbewegungen in der Pflanze. Verlag von Gustav Fischer, Jena.
- Sattelmacher, Burkhard (2001). "The apoplast and its significance for plant mineral nutrition". New Phytologist. 149 (2): 167–192. doi:10.1046/j.1469-8137.2001.00034.x. PMID 33874640. S2CID 86799768.
- Ross, Merrill A.; Lembi, Carole A. (2008). Applied Weed Science: Including the Ecology and Management of Invasive Plants. Prentice Hall. p. 79. ISBN 978-0-13-502814-8.
- Grignon, C.; Sentenac, H. (1991-01-01). "pH and Ionic Conditions in the Apoplast". Annual Review of Plant Physiology and Plant Molecular Biology. 42 (1): 103–128. doi:10.1146/annurev.pp.42.060191.000535.
- "Transport in Plants". Department of Biological Sciences, University of Illinois at Chicago. Archived from the original on 12 December 2015. Retrieved 10 December 2015.
- Thomas N. Buckley; Grace P. John; Christine Scoffoni & Lawren Sack (17 June 2015). "How Does Leaf Anatomy Influence Water Transport outside the Xylem?". Plant Physiology. 168 (4): 1616–1635. doi:10.1104/pp.15.00731. PMC 4528767. PMID 26084922.
- Freeman, Scott (2011). Biological Science. San Francisco, California: Pearson Benjamin Cummings. p. 747. ISBN 978-0-321-59820-2.
- Doehlemann, Gunther; Hemetsberger, Christoph (2013). "Apoplastic immunity and its suppression by filamentous plant pathogens". New Phytologist. 198 (4): 1001–1016. doi:10.1111/nph.12277. ISSN 1469-8137. PMID 23594392.
- Sattelmacher, Burkhard (2001). "The apoplast and its significance for plant mineral nutrition". New Phytologist. 149 (2): 167–192. doi:10.1046/j.1469-8137.2001.00034.x. ISSN 1469-8137. PMID 33874640. S2CID 86799768.
- H, Chen; Z, Lai; J, Shi; Y, Xiao; Z, Chen; X, Xu (2010-12-19). "Roles of Arabidopsis WRKY18, WRKY40 and WRKY60 Transcription Factors in Plant Responses to Abscisic Acid and Abiotic Stress". BMC Plant Biology. 10: 281. doi:10.1186/1471-2229-10-281. PMC 3023790. PMID 21167067.
- Wenke, Katrin; Wanke, Dierk; Kilian, Joachim; Berendzen, Kenneth; Harter, Klaus; Piechulla, Birgit (2012). "Volatiles of two growth-inhibiting rhizobacteria commonly engage AtWRKY18 function". The Plant Journal. 70 (3): 445–459. doi:10.1111/j.1365-313X.2011.04891.x. ISSN 1365-313X. PMID 22188129.
- Romero, Fernando M.; Rossi, Franco R.; Gárriz, Andrés; Carrasco, Pedro; Ruíz, Oscar A. (2018-08-29). "A Bacterial Endophyte from Apoplast Fluids Protects Canola Plants from Different Phytopathogens via Antibiosis and Induction of Host Resistance". Phytopathology. 109 (3): 375–383. doi:10.1094/PHYTO-07-18-0262-R. ISSN 0031-949X. PMID 30156501.
Footnotes
- Salibury F; Ross C (1991). Plant Physiology. Brooks Cole. p. 682. ISBN 978-0-534-15162-1..