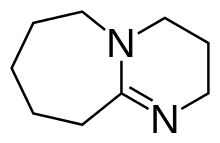
1,8-Diazabicyclo(5.4.0)undec-7-ene
1,8-Diazabicyclo[5.4.0]undec-7-ene, or more commonly DBU, is a chemical compound and belongs to the class of amidine compounds. It is used in organic synthesis as a catalyst, a complexing ligand, and a non-nucleophilic base.[3]
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
2,3,4,6,7,8,9,10-Octahydropyrimido[1,2-a]azepine | |
| Other names
DBU, Diazabicycloundecene | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.027.013 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C9H16N2 | |
| Molar mass | 152.241 g·mol−1 |
| Appearance | Colorless liquid |
| Density | 1.018 g/mL liquid |
| Melting point | −70 °C (−94 °F; 203 K) |
| Boiling point | 80 to 83 °C (176 to 181 °F; 353 to 356 K) (0.6 mmHg); 261 °C (1 atm) |
| Acidity (pKa) | 13.5±1.5[1] (pKa of conjugate acid in water); 24.34[2] (pKa of conjugate acid in acetonitrile) |
| Hazards | |
| GHS labelling: | |
   | |
| Danger | |
| H301, H302, H312, H314, H412 | |
| P260, P264, P270, P273, P280, P301+P310, P301+P312, P301+P330+P331, P302+P352, P303+P361+P353, P304+P340, P305+P351+P338, P310, P312, P321, P322, P330, P363, P405, P501 | |
| Flash point | 119.9 °C (247.8 °F; 393.0 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Occurrence
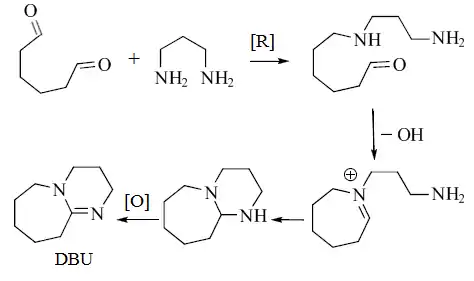
Although all commercially available DBU is produced synthetically, it may also be isolated from the sea sponge Niphates digitalis.[4] The biosynthesis of DBU has been proposed to begin with 1,6-hexanedial and 1,3-diaminopropane.
Uses
As a reagent in organic chemistry, DBU is used as a catalyst, a complexing ligand, and a non-nucleophilic base. It is also used as a curing agent for epoxy resins.
It is used in the separation of fullerenes in conjunction with trimethylbenzene. It reacts with C70 and higher fullerenes, but not with to C60
It is also used as a catalyst in the production of polyurethanes. It also exhibited its dual character (base and nucleophile) in the synthesis of aryl- and styryl-terminal acetylenes.
See also
References
- Kaupmees, K.; Trummal, A.; Leito, I. (2014). "Basicities of Strong Bases in Water: A Computational Study". Croat. Chem. Acta. 87 (4): 385–395. doi:10.5562/cca2472.
- Kaljurand, I.; Kütt, A.; Sooväli, L.; Rodima, T.; Mäemets, V.; Leito, I.; Koppel, I. A. (2005). "Extension of the Self-Consistent Spectrophotometric Basicity Scale in Acetonitrile to a Full Span of 28 pKa Units: Unification of Different Basicity Scales". J. Org. Chem. 70 (3): 1019–1028. doi:10.1021/jo048252w. PMID 15675863.
- Ghosh, Nandita (2004). "DBU (1,8-diazabicyclo[5.4.0]undec-7-ene) - A Nucleophillic Base". Synlett (3): 574–575. doi:10.1055/s-2004-815436.
- E. L. Regalado; Judith Mendiola; Abilio Laguna; Clara Nogueiras; Olivier P Thomas (2010). "Polar alkaloids from the Caribbean marine sponge Niphates digitalis". Nat. Prod. Commun. 5 (8): 1187–1190. PMID 20839615.