Thiazoline
Thiazolines (or dihydrothiazoles) are a group of isomeric 5-membered heterocyclic compounds containing both sulfur and nitrogen in the ring. Although unsubstituted thiazolines are rarely encountered themselves, their derivatives are more common and some are bioactive. For example, in a common post-translational modification, cysteine residues are converted into thiazolines.[1]
 | |
 | |
 | |
| Names | |
|---|---|
| IUPAC names
2,3-Dihydrothiazole 2,5-Dihydrothiazole 4,5-Dihydrothiazole | |
| Other names
2,3-Dihydro-1,3-thiazole or 4-thiazoline 2,5-Dihydro-1,3-thiazole or 3-thiazoline 4,5-Dihydro-1,3-thiazole or 2-thiazoline | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C3H5NS | |
| Molar mass | 87.14 g·mol−1 |
| Appearance | Colorless liquids |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
The name thiazoline originates from the Hantzsch–Widman nomenclature.
Isomers
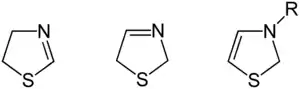
Three structural isomers of thiazoline exist depending on the position of the double bond. These forms do not readily interconvert and hence are not tautomers. Of these 2-thiazoline is the most common.
A fourth structure exists in which the N and S atoms are adjacent; this known as isothiazoline.
Synthesis
Thiazolines were first prepared by dialkylation of thioamides by Richard Willstatter in 1909.[2] 2-Thiazolines are commonly prepared from 2-aminoethanethiols (e.g. cysteamine).[3] They may also be synthesized via the Asinger reaction.
Applications
Many molecules contain thiazoline rings, one example being Firefly luciferin, the light-emitting molecule in fireflies. The amino acid cysteine is produced industrially from substituted thiazole.[3] 2-Aminothiazoline-4-carboxylic acid is an intermediate in the industrial synthesis of L-cysteine.[4]
See also
- Thiazole - an analogue with 2 double bonds
- Thiazolidine - an analogue with no double bonds
- Oxazoline - an analogue with O in place of S
References
- Walsh, Christopher T.; Nolan, Elizabeth M. (2008). "Morphing peptide backbones into heterocycles". Proceedings of the National Academy of Sciences USA. 105 (15): 5655–5656. Bibcode:2008PNAS..105.5655W. doi:10.1073/pnas.0802300105. PMC 2311349. PMID 18398003.
- Willstätter, Richard; Wirth, Theodor (1909). "Über Thioformamid". Chem. Ber. 42 (2): 1908–1922. doi:10.1002/cber.19090420267.
- Gaumont, Annie-Claude; Gulea, Mihaela; Levillain, Jocelyne (11 March 2009). "Overview of the Chemistry of 2-Thiazolines". Chemical Reviews. 109 (3): 1371–1401. doi:10.1021/cr800189z. PMID 19154153.
- Karlheinz Drauz, Ian Grayson, Axel Kleemann, Hans-Peter Krimmer, Wolfgang Leuchtenberger, Christoph Weckbecker (2006). Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a02_057.pub2.
{{cite encyclopedia}}: CS1 maint: multiple names: authors list (link)