2,2'-Dipyrromethene
2,2'-Dipyrromethene, often called just dipyrromethene or dipyrrin, is a chemical compound with formula C
9H
8N
2 whose skeleton can be described as two pyrrole rings C
5N connected by a methyne bridge =CH– through their nitrogen-adjacent (position-2) carbons; the remaining bonds being satisfied by hydrogen atoms. It is an unstable compound that is readily attacked by nucleophilic compounds above −40 °C.[1]
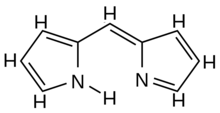 Schematic formula of the dipyrromethene molecule. | |
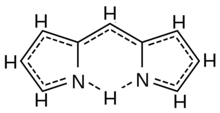 Schematic formula of dipyrromethene (fractional bonding view). | |
| Names | |
|---|---|
| Preferred IUPAC name
2-[(2H-Pyrrol-2-ylidene)methyl]-1H-pyrrole | |
| Other names
dipyrrin, pyrromethene, dipyrromethene | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
PubChem CID |
|
| |
| |
| Properties | |
| C9H8N2 | |
| Molar mass | 144.177 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
2,2'-Dipyrromethene and its more stable and easily prepared derivatives—formally obtained by replacing one or more hydrogen atoms by other functional groups—are important precursors for the family of BODIPY fluorescent dies.[1] The derivatives include salts of the dipyrrinato anion C
9H
7N–
2 and of the cation C
9H
9N+
2.
Preparation
2,2'-Dipyrromethene and its derivatives can be obtained from suitable pyrrole derivatives by several methods.[2]
The unsubstituted compound can be prepared by oxidation of 2,2'-dipyrrolemethane with 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ) at −78 °C in dry dichloromethane solution.[1] An alternative synthesis that avoids the oxidation step is the condensation of 2-formyl pyrrole and pyrrole catalyzed by trifluoroacetic acid, followed by deprotonation with N,N-diisopropylethylamine.[3]
More generally, one starts with a pyrrole with suitable substituents at positions 3, 4, or 5 (but not 2). Condensation of two such molecules at their 2 positions with a bridging compound gives the corresponding 2,2'-dipyrromethane. The condensation may use, for example, the Knorr pyrrole synthesis, with an aromatic aldehyde in the presence of TFA. The dipyrromethane core is then oxidized to dipyrromethene using a quinone oxidant such as DDQ or p-chloranil.
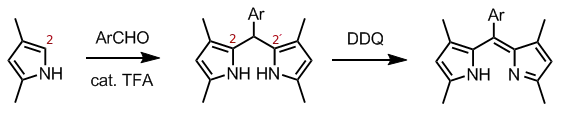
Alternatively, one may use an activated carboxylic acid derivative, usually an acyl chloride. As another possibility, one may condense a substituted pyrroles with a 2-acylpyrrole; this route allows the synthesis of unsymmetrical dipyrromethenes.
Reactions
Dipyrrin is unstable above −40 °C. However, its acts as a base, and its chloride [C
9H
9N+
2] [Cl−
] is sufficiently stable in solution.[4]
The so-called BODIPY dyes can be obtained by reacting 2,2'-dipyrromethene or its derivatives with boron trifluoride-diethyl ether complex (BF
3·(C
2H
5)
2O) in the presence of triethylamine or 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU).[1]
Dipyrrin and its derivatives for coordination complexes with transition metals. For example, the derivative anion 5-phenyl dipirrinato (pdp) forms the neutral iron(III) complex Fe(pdp)
3 (dark green monoclinic crystals, soluble in benzene, orange solution in dichloromethane), where the Fe3+
ion is coordinated to six nitrogen atoms of the dipyrrin cores in distorted octahedral geometry.[5] A similar cobalt(III) complex has also been reported, as well as a complex with copper(II) Cu(pdp)
2[6]
References
- =K. Tram, H. Yan, H. A. Jenkins, S. Vassiliev, and D. Bruce (2009): "The synthesis and crystal structure of unsubstituted 4,4-difluoro-4-bora-3a,4a-diaza-s-indacene (BODIPY)". Dyes and Pigments, volume 82, issue 3, pages = 392–395. doi:10.1016/j.dyepig.2009.03.001
- Burgess, Kevin (October 2007). "BODIPY Dyes and Their Derivatives: Syntheses and Spectroscopic Properties". Chemical Reviews. 107 (11): 4891–4932. doi:10.1021/cr078381n. PMID 17924696.
- A. Schmitt, B. Hinkeldey, M. Wild , and G. Jung (): "Synthesis of the core compound of the BODIPY dye class: 4,4′-difluoro-4-bora-(3a,4a)-diaza-s-indacene". Journal of Fluorescence, volume = 19, issue = 4, pages = 755–759 doi:10.1007/s10895-008-0446-7 PMID 19067126
- Brandon R. Groves, Sarah M. Crawford,a Travis Lundrigan, Chérif F. Matta, Shahin Sowlati-Hashjin, and Alison Thompson (2013): "Synthesis and characterisation of the unsubstituted dipyrrin and 4,4-dichloro-4-bora-3a,4a-diaza-s-indacene: improved synthesis and functionalisation of the simplest BODIPY framework." Chemical Communications, volume 49, pages 816-818. doi:10.1039/c2cc37480c
- Seth M Cohen and Sara R Halper (2002): "Dipyrromethene complexes of iron". Inorganica Chimica Acta, volume 341, pages 12-16.
- Sara R. Halper, Mitchell R. Malachowski, Heather M. Delaney, and Seth M. Cohen (2004): "Heteroleptic copperdipyrromethene complexes: synthesis, structure,and coordination polymers". Inorganic Chemistry, volume 43, pages 1242−1249