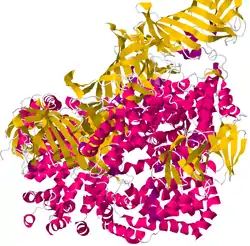
ERAP1
Type 1 tumor necrosis factor receptor shedding aminopeptidase regulator, also known as endoplasmic reticulum aminopeptidase 1 (ARTS-1), is a protein which in humans is encoded by the ARTS-1 gene.[5]
Endoplasmic reticulum amino peptidase 1 is active in the endoplasmic reticulum, which is involved in protein processing and transport. This protein is an aminopeptidase, which is an enzyme that cleaves peptides at the N-terminal into smaller fragments called amino acids.
Nomenclature
ARTS1 is also known as:
Function
ERAP1 has two major functions in the immune system:
- First, ERAP1 cleaves several proteins called cytokine receptors on the surface of cells. Cleaving these receptors reduces their ability to transmit chemical signals into the cell, which affects the process of inflammation.
- Second, ERAP1 trims peptides within the endoplasmic reticulum so that they can be loaded onto major histocompatibility complex (MHC) class I. These peptides are attached to MHC class I in the endoplasmic reticulum and exported to the cell surface, where they are displayed to the immune system. If the immune system recognizes the peptides as foreign (such as viral or bacterial peptides), it responds by triggering the infected cell to self-destruct.[8]
ARTS-1 is a member of the M1 family of zinc metallopeptidases which acts as an aminopeptidase that degrades oligopeptides by cleavage starting at the amino terminus. One of the functions of aminopeptidases is to degrade potentially toxic peptides in the cytosol.[5]
ARTS-1 is a transmembrane protein that is localized to the endoplasmic reticulum. It has been implicated in the following functions:
- Shedding of various cytokine receptors and decoy receptors
- Trimming of antigenic peptides before binding to MHC class I, affecting antigen presentation to cytotoxic T lymphocytes
- Stimulation of phagocytosis upon release by macrophages[9]
Clinical significance
Aminopeptidases play a role in the metabolism of several peptides that may be involved in blood pressure and the pathogenesis of essential hypertension.[5] Mutations in the ARTS-1 have been linked to an increased risk of ankylosing spondylitis but only in HLA-B27 positive patients.[10]
The protein encoded by this gene is an aminopeptidase involved in trimming HLA class I-binding precursors so that they can be presented on MHC class I molecules. The encoded protein acts as a monomer or as a heterodimer with ERAP2. This protein may also be involved in blood pressure regulation by inactivation of angiotensin II. Three transcript variants encoding two different isoforms have been found for this gene.[5]
References
- GRCh38: Ensembl release 89: ENSG00000164307 - Ensembl, May 2017
- GRCm38: Ensembl release 89: ENSMUSG00000021583 - Ensembl, May 2017
- "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- EntrezGene 51752
- "HGNC". HGNC. Retrieved 27 March 2014.
- Serwold T, Gonzalez F, Kim J, Jacob R, Shastri N (2002). "ERAAP customizes peptides for MHC class I molecules in the endoplasmic reticulum". Nature. 419 (6906): 480–3. doi:10.1038/nature01074. PMID 12368856. S2CID 4379291.
- "ERAP1 - endoplasmic reticulum aminopeptidase 1 - Genetics Home Reference".
- Goto Y, Ogawa K, Hattori A, Tsujimoto M (June 2011). "Secretion of endoplasmic reticulum aminopeptidase 1 is involved in the activation of macrophages induced by lipopolysaccharide and interferon-gamma". The Journal of Biological Chemistry. 286 (24): 21906–14. doi:10.1074/jbc.M111.239111. PMC 3122245. PMID 21531727.
- Brionez TF, Reveille JD (2008). "The contribution of genes outside the major histocompatibility complex to susceptibility to ankylosing spondylitis". Current Opinion in Rheumatology. 20 (4): 384–91. doi:10.1097/BOR.0b013e32830460fe. PMID 18525349. S2CID 205485848.
External links
- Human ERAP1 genome location and ERAP1 gene details page in the UCSC Genome Browser.
Further reading
- Nakajima D, Okazaki N, Yamakawa H, Kikuno R, Ohara O, Nagase T (2002). "Construction of Expression-ready cDNA Clones for KIAA Genes: Manual Curation of 330 KIAA cDNA Clones". DNA Research. 9 (3): 99–106. CiteSeerX 10.1.1.500.923. doi:10.1093/dnares/9.3.99. PMID 12168954.
- Tsujimoto M, Hattori A (2005). "The oxytocinase subfamily of M1 aminopeptidases". Biochimica et Biophysica Acta (BBA) - Proteins and Proteomics. 1751 (1): 9–18. doi:10.1016/j.bbapap.2004.09.011. PMID 16054015.
- Maruyama K, Sugano S (1994). "Oligo-capping: a simple method to replace the cap structure of eukaryotic mRNAs with oligoribonucleotides". Gene. 138 (1–2): 171–4. doi:10.1016/0378-1119(94)90802-8. PMID 8125298.
- Suzuki Y, Yoshitomo-Nakagawa K, Maruyama K, Suyama A, Sugano S (1997). "Construction and characterization of a full length-enriched and a 5′-end-enriched cDNA library". Gene. 200 (1–2): 149–56. doi:10.1016/S0378-1119(97)00411-3. PMID 9373149.
- Nagase T, Ishikawa K, Miyajima N, Tanaka A, Kotani H, Nomura N, Ohara O (1998). "Prediction of the Coding Sequences of Unidentified Human Genes. IX. The Complete Sequences of 100 New cDNA Clones from Brain Which Can Code for Large Proteins in vitro". DNA Research. 5 (1): 31–9. doi:10.1093/dnares/5.1.31. PMID 9628581.
- Hattori A, Matsumoto H, Mizutani S, Tsujimoto M (1999). "Molecular cloning of adipocyte-derived leucine aminopeptidase highly related to placental leucine aminopeptidase/oxytocinase". Journal of Biochemistry. 125 (5): 931–8. doi:10.1093/oxfordjournals.jbchem.a022371. PMID 10220586.
- Hattori A, Kitatani K, Matsumoto H, Miyazawa S, Rogi T, Tsuruoka N, Mizutani S, Natori Y, Tsujimoto M (2000). "Characterization of recombinant human adipocyte-derived leucine aminopeptidase expressed in Chinese hamster ovary cells". Journal of Biochemistry. 128 (5): 755–62. doi:10.1093/oxfordjournals.jbchem.a022812. PMID 11056387.
- Hattori A, Matsumoto K, Mizutani S, Tsujimoto M (2001). "Genomic organization of the human adipocyte-derived leucine aminopeptidase gene and its relationship to the placental leucine aminopeptidase/oxytocinase gene". Journal of Biochemistry. 130 (2): 235–41. doi:10.1093/oxfordjournals.jbchem.a002977. PMID 11481040.
- Yamamoto N, Nakayama J, Yamakawa-Kobayashi K, Hamaguchi H, Miyazaki R, Arinami T (2002). "Identification of 33 polymorphisms in the adipocyte-derived leucine aminopeptidase (ALAP) gene and possible association with hypertension". Human Mutation. 19 (3): 251–7. doi:10.1002/humu.10047. PMID 11857741. S2CID 23459237.
- Cui X, Hawari F, Alsaaty S, Lawrence M, Combs CA, Geng W, Rouhani FN, Miskinis D, Levine SJ (2002). "Identification of ARTS-1 as a novel TNFR1-binding protein that promotes TNFR1 ectodomain shedding". Journal of Clinical Investigation. 110 (4): 515–26. doi:10.1172/JCI13847. PMC 150410. PMID 12189246.
- Serwold T, Gonzalez F, Kim J, Jacob R, Shastri N (2002). "ERAAP customizes peptides for MHC class I molecules in the endoplasmic reticulum". Nature. 419 (6906): 480–3. doi:10.1038/nature01074. PMID 12368856. S2CID 4379291.
- Saric T, Chang SC, Hattori A, York IA, Markant S, Rock KL, Tsujimoto M, Goldberg AL (2002). "An IFN-γ–induced aminopeptidase in the ER, ERAP1, trims precursors to MHC class I–presented peptides". Nature Immunology. 3 (12): 1169–76. doi:10.1038/ni859. PMID 12436109. S2CID 21648629.
- York IA, Chang SC, Saric T, Keys JA, Favreau JM, Goldberg AL, Rock KL (2002). "The ER aminopeptidase ERAP1 enhances or limits antigen presentation by trimming epitopes to 8–9 residues" (PDF). Nature Immunology. 3 (12): 1177–84. doi:10.1038/ni860. PMID 12436110. S2CID 21337369.
- Cui X, Rouhani FN, Hawari F, Levine SJ (2003). "An Aminopeptidase, ARTS-1, Is Required for Interleukin-6 Receptor Shedding". Journal of Biological Chemistry. 278 (31): 28677–85. doi:10.1074/jbc.M300456200. PMID 12748171.
- Clark HF, Gurney AL, Abaya E, Baker K, Baldwin D, Brush J, Chen J, Chow B, Chui C, Crowley C, Currell B, Deuel B, Dowd P, Eaton D, Foster J, Grimaldi C, Gu Q, Hass PE, Heldens S, Huang A, Kim HS, Klimowski L, Jin Y, Johnson S, Lee J, Lewis L, Liao D, Mark M, Robbie E, Sanchez C, Schoenfeld J, Seshagiri S, Simmons L, Singh J, Smith V, Stinson J, Vagts A, Vandlen R, Watanabe C, Wieand D, Woods K, Xie MH, Yansura D, Yi S, Yu G, Yuan J, Zhang M, Zhang Z, Goddard A, Wood WI, Godowski P, Gray A (2003). "The Secreted Protein Discovery Initiative (SPDI), a Large-Scale Effort to Identify Novel Human Secreted and Transmembrane Proteins: A Bioinformatics Assessment". Genome Research. 13 (10): 2265–70. doi:10.1101/gr.1293003. PMC 403697. PMID 12975309.
- Cui X, Rouhani FN, Hawari F, Levine SJ (2003). "Shedding of the Type II IL-1 Decoy Receptor Requires a Multifunctional Aminopeptidase, Aminopeptidase Regulator of TNF Receptor Type 1 Shedding". The Journal of Immunology. 171 (12): 6814–9. doi:10.4049/jimmunol.171.12.6814. PMID 14662887.
- Shibata D, Ando H, Iwase A, Nagasaka T, Hattori A, Tsujimoto M, Mizutani S (2004). "Distribution of Adipocyte-derived Leucine Aminopeptidase (A-LAP)/ER-aminopeptidase (ERAP)-1 in Human Uterine Endometrium". Journal of Histochemistry and Cytochemistry. 52 (9): 1169–75. doi:10.1369/jhc.3A6216.2004. PMID 15314084.