Centralite
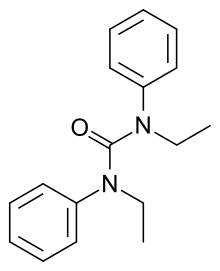
Centralite (empirical formula: C17H20N2O) is a gunshot residue also known as ethyl centralite. Its IUPAC name is 1,3-diethyl-1,3-diphenylurea. Ethyl centralite is insoluble in water, but is soluble in acetone, ethanol and benzene. It is mainly used as a burning rate moderator and stabilizer for smokeless powder, and also a plasticizer for celluloid.
 | |
| Names | |
|---|---|
| Preferred IUPAC name
N,N′-Diethyl-N,N′-Diphenylurea | |
| Other names
Ethyl centralite N,N′-Diethylcarbanilide | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.001.496 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C17H20N2O | |
| Molar mass | 268.360 g·mol−1 |
| Appearance | White to light grey crystalline powder |
| Density | 0.8 g/cm3 |
| Insoluble | |
| Solubility in Acetone, ethanol and benzene | Soluble |
| -134.05·10−6 cm3/mol | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
History
In the 19th century chemists identified that nitrocellulose can destroy itself by action of nitrogen oxides separating from it at storage, and tried to find bases which might capture those oxides. Urea has been used for stabilizing celluloid in the 19th century (and even in early American military powders), but like other water-soluble bases, it also attacks nitrocellulose, so German chemists substituted hydrogen atoms with nonpolar organic radicals to diminish this effect.[2]
Naming
Synonyms: Bis(N-ethyl-N-phenyl)urea; Carbamite; Centralit; Centralite; Centralite1; Centralite 1; Centralite-1; Centralite I; N,N′-Diethylcarbanilide; Diethyldiphenylharnstoff; N,N′-Diethyl-N′-diphenyl-L-harnstoff; 1,3-Diethyl-1,3-diphenylharnstoff; Diethyldiphenylurea; 1,3-Diethyldiphenylurea; 1,3-Diethyl-1,3-diphenylurea; s-Diethyldiphenylurea; sym-Diethyldiphenylurea; N,N′-Diphenyl-N,N′-diethylharnstoff; N,N′-Diphenyl-N,N′-diethylurea; Ethylcentralit; Ethylcentralite; Ethyl centralite; USAF EK-1047; Zentralit.
The term "Centralite" was originally applied to dimethyldiphenylurea developed about 1906 at the German Central War Laboratory Zentralstelle für wissenschaftlich-technische Untersuchungen in Neubabelsberg as a deterrent coating for smokeless powder in military rifle cartridges. Thereafter, all hydrocarbon-substituted symmetrical diphenyl urea compounds used as smokeless powder deterrents (or moderants) were called centralites after the laboratory. The preferred ethyl centralite became known as Centralite No. 1 and the original methyl centralite was identified as Centralite No. 2. Butyl centralite was also used as a celluloid plasticizer.[3][4]
Comparison with analogs
Its reaction history is considerably more complicated than that of diphenylurea. Ending up with nitrated anilines, the methyl analog centralite-2 or sym-dimethyldiphenylurea is also known and is used somewhat abroad. The centralite are considered to be somewhat less effective as stabilizers than 2-nitrodiphenylamine, but they are also quite good plasticizers. When found in propellants they are frequently used at higher fractions than the diphenylamines to take advantage of their plasticizing properties.
References
- CID 6828 from PubChem
- https://archive.org/details/explosives02mars/page/641/mode/1up
- Davis, Tenney L. (1943). The Chemistry of Powder & Explosives (Angriff Press [1992] ed.). John Wiley & Sons Inc. pp. 317–320. ISBN 0-913022-00-4.
- Davis, William C., Jr. (1981). Handloading. National Rifle Association of America. p. 130. ISBN 0-935998-34-9.
{{cite book}}: CS1 maint: multiple names: authors list (link)