2-Ethylhexanoic acid
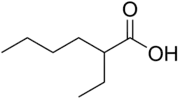
2-Ethylhexanoic acid is the organic compound with the formula CH3(CH2)3CH(C2H5)CO2H. It is a carboxylic acid that is widely used to prepare lipophilic metal derivatives that are soluble in nonpolar organic solvents. 2-Ethylhexanoic acid is a colorless viscous oil. It is supplied as a racemic mixture.
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
2-Ethylhexanoic acid[1] | |
| Identifiers | |
| |
3D model (JSmol) |
|
| 1750468 | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.005.222 |
| EC Number |
|
| MeSH | 2-ethylhexanoic+acid |
PubChem CID |
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C8H16O2 | |
| Molar mass | 144.214 g·mol−1 |
| Appearance | Colorless liquid |
| Density | 903 mg mL−1 |
| Melting point | −59.00 °C; −74.20 °F; 214.15 K |
| Boiling point | 228.1 °C; 442.5 °F; 501.2 K |
| log P | 2.579 |
| Vapor pressure | <1 Pa (at 25 °C) |
| Acidity (pKa) | 4.819 |
| Basicity (pKb) | 9.178 |
Refractive index (nD) |
1.425 |
| Thermochemistry | |
Std enthalpy of formation (ΔfH⦵298) |
−635.1 kJ mol−1 |
Std enthalpy of combustion (ΔcH⦵298) |
-4.8013–4.7979 MJ mol−1 |
| Hazards | |
| GHS labelling: | |
   | |
| Danger | |
| H312, H318, H361 | |
| P280, P305+P351+P338 | |
| Flash point | 114 °C (237 °F; 387 K) |
| 371 °C (700 °F; 644 K) | |
| Explosive limits | 0.9–6.7% |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
|
| Related compounds | |
Related compounds |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Production
2-Ethylhexanoic acid is produced industrially from propylene, which is hydroformylated to give butyraldehyde. Aldol condensation of the aldehyde gives 2-ethylhexenal, which is hydrogenated to 2-ethylhexanal. Oxidation of this aldehyde gives the carboxylic acid.[2]
Metal ethylhexanoates
2.jpg.webp)
2-Ethylhexanoic acid forms compounds with metal cations that have stoichiometry as metal acetates. These ethylhexanoate complexes are used in organic and industrial chemical synthesis. They function as catalysts in polymerizations as well as for oxidation reactions as "oil drying agents."[3] They are highly soluble in nonpolar solvents. These metal complexes are often described as salts. They are, however, not ionic but charge-neutral coordination complexes. Their structures are akin to the corresponding acetates.
Examples of metal ethylhexanoates
- Hydroxyl aluminium bis(2-ethylhexanoate), used as a thickener
- Tin(II) ethylhexanoate (CAS# 301-10-0), a catalyst for polylactide and poly(lactic-co-glycolic acid).[4]
- Cobalt(II) ethylhexanoate (CAS# 136-52-7), a drier for alkyd resins
- Nickel(II) ethylhexanoate (CAS# 4454-16-4)
See also
References
- "2-ethylhexanoic acid - Compound Summary". PubChem Compound. USA: National Center for Biotechnology Information. 26 March 2005. Identification and Related Records. Retrieved 21 February 2012.
- Riemenschneider, Wilhelm (2002). "Carboxylic Acids, Aliphatic". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a05_235.
- Raju, Ravinder; Prasad, Kapa (2012). "Synthetic applications of 2-ethylhexanoic acid derived reagents". Tetrahedron. 68 (5): 1341–1349. doi:10.1016/j.tet.2011.10.078.
- Coulembier, O.; Degee, P.; Hedrick, J. L.; Dubois, P. (2006). "Controlled Ring-Opening Polymerization to Biodegradable Aliphatic Polyester: Especially Poly(Β-Malic Acid) Derivatives". Prog. Polym. Sci. 31: 723–747. doi:10.1016/j.progpolymsci.2006.08.004.