Human vestigiality
In the context of human evolution, human vestigiality involves those traits occurring in humans that have lost all or most of their original function through evolution. Although structures called vestigial often appear functionless, a vestigial structure may retain lesser functions or develop minor new ones. In some cases, structures once identified as vestigial simply had an unrecognized function. Vestigial organs are sometimes called rudimentary organs.[1] Many human characteristics are also vestigial in other primates and related animals.

History
Charles Darwin listed a number of putative human vestigial features, which he termed rudimentary, in The Descent of Man (1871). These included the muscles of the ear; wisdom teeth; the appendix; the tail bone; body hair; and the semilunar fold in the corner of the eye. Darwin also commented on the sporadic nature of many vestigial features, particularly musculature. Making reference to the work of the anatomist William Turner, Darwin highlighted a number of sporadic muscles which he identified as vestigial remnants of the panniculus carnosus, particularly the sternalis muscle.[2][3]
In 1893, Robert Wiedersheim published The Structure of Man, a book on human anatomy and its relevance to man's evolutionary history. This book contained a list of 86 human organs that he considered vestigial, or as Wiedersheim himself explained: "Organs having become wholly or in part functionless, some appearing in the Embryo alone, others present during Life constantly or inconstantly. For the greater part Organs which may be rightly termed Vestigial."[4] His list of supposedly vestigial organs included many of the examples on this page as well as others then mistakenly believed to be purely vestigial, such as the pineal gland, the thymus gland, and the pituitary gland. Some of these organs that had lost their obvious, original functions later turned out to have retained functions that had gone unrecognized before the discovery of hormones or many of the functions and tissues of the immune system.[5][6] Examples included:
- the role of the pineal in the regulation of the circadian rhythm (neither the function nor even the existence of melatonin was yet known);
- discovery of the role of the thymus in the immune system lay many decades in the future; it remained a mystery organ until after the mid-20th century;
- the pituitary and hypothalamus with their many and varied hormones were far from understood, let alone the complexity of their interrelationships.
Historically, there was a trend not only to dismiss the vermiform appendix as being uselessly vestigial, but an anatomical hazard, a liability to dangerous inflammation. As late as the mid-20th century, many reputable authorities conceded it no beneficial function.[7] This was a view supported, or perhaps inspired, by Darwin himself in the 1874 edition of his book The Descent of Man, and Selection in Relation to Sex. The organ's patent liability to appendicitis and its poorly understood role left the appendix open to blame for a number of possibly unrelated conditions. For example, in 1916, a surgeon claimed that removal of the appendix had cured several cases of trifacial neuralgia and other nerve pain about the head and face, even though he stated that the evidence for appendicitis in those patients was inconclusive.[8] The discovery of hormones and hormonal principles, notably by Bayliss and Starling, argued against these views, but in the early twentieth century, there remained a great deal of fundamental research to be done on the functions of large parts of the digestive tract. In 1916, an author found it necessary to argue against the idea that the colon had no important function and that "the ultimate disappearance of the appendix is a coordinate action and not necessarily associated with such frequent inflammations as we are witnessing in the human".[9]
There had been a long history of doubt about such dismissive views. Around 1920, the prominent surgeon Kenelm Hutchinson Digby documented previous observations, going back more than thirty years, that suggested lymphatic tissues, such as the tonsils and appendix, may have substantial immunological functions.
Anatomical
Appendix
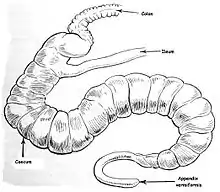

In modern humans, the appendix is sometimes believed to be a vestige of a redundant organ that in ancestral species had digestive functions, much as it still does in extant species in which intestinal flora hydrolyze cellulose and similar indigestible plant materials.[10] This view has changed over the past decades,[11] with research suggesting that the appendix may serve an important purpose. In particular, it may serve as a reservoir for beneficial gut bacteria.
Some herbivorous animals, such as rabbits, have a terminal vermiform appendix and cecum that apparently bear patches of tissue with immune functions and may also be important in maintaining the composition of intestinal flora. It does not however seem to have much digestive function, if any, and is not present in all herbivores, even those with large caeca.[12] As shown in the accompanying pictures however, the human appendix typically is about comparable to that of the rabbit's in size, though the caecum is reduced to a single bulge where the ileum empties into the colon.[7] Some carnivorous animals may have appendices too, but seldom have more than vestigial caeca.[13] In line with the possibility of vestigial organs developing new functions, some research suggests that the appendix may guard against the loss of symbiotic bacteria that aid in digestion, though that is unlikely to be a novel function, given the presence of vermiform appendices in many herbivores.[14][15] Intestinal bacterial populations entrenched in the appendix may support quick re-establishment of the flora of the large intestine after an illness, poisoning, or after an antibiotic treatment depletes or otherwise causes harmful changes to the bacterial population of the colon.[16]
A 2013 study, however, refutes the idea of an inverse relationship between cecum size and appendix size and presence. It is widely present in euarchontoglires (a superorder of mammals that includes rodents, lagomorphs and primates) and has also evolved independently in the diprotodont marsupials, monotremes, and is highly diverse in size and shape which could suggest it is not vestigial. Researchers deduce that the appendix has the ability to protect good bacteria in the gut. That way, when the gut is affected by a bout of diarrhea or other illness that cleans out the intestines, the good bacteria in the appendix can repopulate the digestive system and keep the person healthy.[17]
Coccyx
The coccyx, or tailbone, is the remnant of a lost tail.[18] All mammals have a tail at some point in their development; in humans, it is present for a period of 4 weeks, during stages 14 to 22 of human embryogenesis.[19] This tail is most prominent in human embryos 31–35 days old.[20] The tailbone, located at the end of the spine, has lost its original function in assisting balance and mobility, though it still serves some secondary functions, such as being an attachment point for muscles, which explains why it has not degraded further.
In rare cases, congenital defect results in a short tail-like structure being present at birth. Twenty-three cases of human babies born with such a structure have been reported in the medical literature since 1884.[21][22] In rare cases such as these, the spine and skull were determined to be entirely normal. The only abnormality was that of a tail approximately twelve centimeters long. These tails, though of no deleterious effect, were almost always surgically removed.[23]
Wisdom teeth
Wisdom teeth are vestigial third molars that human ancestors used to help in grinding down plant tissue. The common postulation is that the skulls of human ancestors had larger jaws with more teeth, which were possibly used to help chew down foliage to compensate for a lack of ability to efficiently digest the cellulose that makes up a plant cell wall. As human diets changed, smaller jaws were naturally selected, yet the third molars, or "wisdom teeth", still commonly develop in human mouths.[24]
Agenesis (failure to develop) of wisdom teeth in human populations ranges from zero in Tasmanian Aboriginals to nearly 100% in indigenous Mexicans.[25] The difference is related to the PAX9 gene (and perhaps other genes).[26]
Vomeronasal organ
In some animals, the vomeronasal organ (VNO) is part of a second, completely separate sense of smell, known as the accessory olfactory system. Many studies have been performed to find if there is an actual presence of a VNO in adult human beings. Trotier et al.[27] estimated that around 92% of their subjects who had not had septal surgery had at least one intact VNO. Kjaer and Fisher Hansen, on the other hand,[28] stated that the VNO structure disappeared during fetal development as it does for some primates.[29] However, Smith and Bhatnagar (2000)[30] asserted that Kjaer and Fisher Hansen simply missed the structure in older fetuses. Won (2000) found evidence of a VNO in 13 of his 22 cadavers (59.1%) and in 22 of his 78 living patients (28.2%).[31] Given these findings, some scientists have argued that there is a VNO in adult human beings.[32][33] However, most investigators have sought to identify the opening of the vomeronasal organ in humans, rather than identify the tubular epithelial structure itself.[34] Thus it has been argued that such studies, employing macroscopic observational methods, have sometimes missed or even misidentified the vomeronasal organ.[35]
Among studies that use microanatomical methods, there is no reported evidence that human beings have active sensory neurons like those in working vomeronasal systems of other animals.[35][36] Furthermore, there is no evidence to date that suggests there are nerve and axon connections between any existing sensory receptor cells that may be in the adult human VNO and the brain.[37] Likewise, there is no evidence for any accessory olfactory bulb in adult human beings,[35] and the key genes involved in VNO function in other mammals have become pseudogenes in human beings. Therefore, while the presence of a structure in adult human beings is debated, a review of the scientific literature by Tristram Wyatt concluded, "most in the field ... are sceptical about the likelihood of a functional VNO in adult human beings on current evidence."[38]
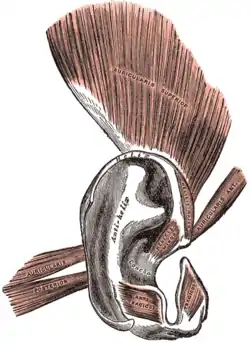
Ear
The ears of a macaque monkey and most other monkeys have far more developed muscles than those of humans, and therefore have the capability to move their ears to better hear potential threats.[39] Humans and other primates such as the orangutan and chimpanzee however have ear muscles that are minimally developed and non-functional, yet still large enough to be identifiable.[10] A muscle attached to the ear that cannot move the ear, for whatever reason, can no longer be said to have any biological function. In humans there is variability in these muscles, such that some people are able to move their ears in various directions, and it can be possible for others to gain such movement by repeated trials.[10][40] In such primates, the inability to move the ear is compensated mainly by the ability to turn the head on a horizontal plane, an ability which is not common to most monkeys—a function once provided by one structure is now replaced by another.[41]
The outer structure of the ear also shows some vestigial features, such as the node or point on the helix of the ear known as Darwin's tubercle which is found in around 10% of the population.
Eye
The plica semilunaris is a small fold of tissue on the inside corner of the eye. It is the vestigial remnant of the nictitating membrane, i.e., third eyelid, an organ that is fully functional in some other species of mammals.[42] Its associated muscles are also vestigial.[10] Only one species of primate, the Calabar angwantibo, is known to have a functioning nictitating membrane.[43]
The orbitalis muscle is a vestigial or rudimentary nonstriated muscle (smooth muscle) of the eye that crosses from the infraorbital groove and sphenomaxillary fissure and is intimately united with the periosteum of the orbit. It was described by Johannes Peter Müller and is often called Müller's muscle. The muscle forms an important part of the lateral orbital wall in some animals, but in humans it is not known to have any significant function.[44][45]
Reproductive system
Genitalia
In the internal genitalia of each human sex, there are some residual organs of mesonephric and paramesonephric ducts during embryonic development:
Human vestigial structures also include leftover embryological remnants that once served a function during development, such as the belly button, and analogous structures between biological sexes. For example, men are also born with two nipples, which are not known to serve a function compared to women.[46] In regards to genitourinary development, both internal and external genitalia of male and female fetuses have the ability to fully or partially form their analogous phenotype of the opposite biological sex if exposed to a lack/overabundance of androgens or the SRY gene during fetal development.[47][48] Examples of vestigial remnants of genitourinary development include the hymen, which is a membrane that surrounds or partially covers the external vaginal opening that derives from the sinus tubercle during fetal development and is homologous to the male seminal colliculus.[49] Some researchers have hypothesized that the persistence of the hymen may be to provide temporary protection from infection, as it separates the vaginal lumen from the urogenital sinus cavity during development.[50] Other examples include the glans penis and the clitoris, the labia minora and the ventral penis, and the ovarian follicles and the seminiferous tubules.[49]
In modern times, there is controversy regarding whether the foreskin is a vital or vestigial structure.[51] In 1949, British physician Douglas Gairdner noted that the foreskin plays an important protective role in newborns. He wrote, "It is often stated that the prepuce is a vestigial structure devoid of function ... However, it seems to be no accident that during the years when the child is incontinent the glans is completely clothed by the prepuce, for, deprived of this protection, the glans becomes susceptible to injury from contact with sodden clothes or napkin."[51] During the physical act of sex, the foreskin reduces friction, which can reduce the need for additional sources of lubrication.[51] "Some medical researchers, however, claim circumcised men enjoy sex just fine and that, in view of recent research on HIV transmission, the foreskin causes more trouble than it's worth."[51] The area of the outer foreskin measures between 7 and 100 cm2,[52] and the inner foreskin measures between 18 and 68 cm2,[53] which is a wide range. Regarding vestigial structures, Charles Darwin wrote, "An organ, when rendered useless, may well be variable, for its variations cannot be checked by natural selection."[54]
Musculature
A number of muscles in the human body are thought to be vestigial, either by virtue of being greatly reduced in size compared to homologous muscles in other species, by having become principally tendonous, or by being highly variable in their frequency within or between populations.
Head
The occipitalis minor is a muscle in the back of the head which normally joins to the auricular muscles of the ear. This muscle is very sporadic in frequency—always present in Malays, present in 56% of Africans, 50% of Japanese, and 36% of Europeans, and nonexistent in the Khoikhoi people of southwestern Africa and in Melanesians.[55] Other small muscles in the head associated with the occipital region and the post-auricular muscle complex are often variable in their frequency.[56]
The platysma, a quadrangular (four sides) muscle in a sheet-like configuration, is a vestigial remnant of the panniculous carnosus of animals. In horses, it is the muscle that allows it to flick a fly off its back.
Face
In many lower animals, the upper lip and sinus area is associated with whiskers or vibrissae which serve a sensory function. In humans, these whiskers do not exist but there are still sporadic cases where elements of the associated vibrissal capsular muscles or sinus hair muscles can be found. Based on histological studies of the upper lips of 20 cadavers, Tamatsu et al. found that structures resembling such muscles were present in 35% (7/20) of their specimens.[57]
Arm
The palmaris longus muscle is seen as a small tendon between the flexor carpi radialis and the flexor carpi ulnaris, although it is not always present. The muscle is absent in about 14% of the population, however this varies greatly with ethnicity. It is believed that this muscle actively participated in the arboreal locomotion of primates, but currently has no function, because it does not provide more grip strength.[58] One study has shown the prevalence of palmaris longus agenesis in 500 Indian patients to be 17.2% (8% bilateral and 9.2% unilateral).[59] The palmaris is a popular source of tendon material for grafts and this has prompted studies which have shown the absence of the palmaris does not have any appreciable effect on grip strength.[60]
The levator claviculae muscle in the posterior triangle of the neck is a supernumerary muscle present in only 2–3% of all people[61] but nearly always present in most mammalian species, including gibbons and orangutans.[62]
Torso
The pyramidalis muscle of the abdomen is a small and triangular muscle, anterior to the rectus abdominis, and contained in the rectus sheath. It is absent in 20% of humans and when absent, the lower end of the rectus then becomes proportionately increased in size. Anatomical studies suggest that the forces generated by the pyramidalis muscles are relatively small.[63]
The latissimus dorsi muscle of the back has several sporadic variations. One particular variant is the existence of the dorsoepitrochlearis or latissimocondyloideus muscle which is a muscle passing from the tendon of the latissimus dorsi to the long head of the triceps brachii. It is notable due to its well developed character in other apes and monkeys, where it is an important climbing muscle, namely the dorsoepitrochlearis brachii.[64][65] This muscle is found in ≈5% of humans.[66]
Leg
The plantaris muscle is composed of a thin muscle belly and a long thin tendon. The muscle belly is approximately 5–10 centimetres (2–4 inches) long, and is absent in 7–10% of the human population. It has some weak functionality in moving the knee and ankle but is generally considered redundant and is often used as a source of tendon for grafts. The long, thin tendon of the plantaris is humorously called "the freshman's nerve", as it is often mistaken for a nerve by new medical students.
Tongue
Another example of human vestigiality occurs in the tongue, specifically the chondroglossus muscle. In a morphological study of 100 Japanese cadavers, it was found that 86% of fibers identified were solid and bundled in the appropriate way to facilitate speech and mastication. The other 14% of fibers were short, thin and sparse – nearly useless, and thus concluded to be of vestigial origin.[67]
Breasts
Extra nipples or breasts sometimes appear along the mammary lines of humans, appearing as a remnant of mammalian ancestors who possessed more than two nipples or breasts.[68][69] One 2021 report demonstrated that all healthy young men and women who participated in an anatomic study of the front surface of the body exhibited 8 pairs of focal fat mounds running along the embryological mammary ridges from axillae to the upper inner thighs. These were always located in the same relative anatomic sites – analogous to the loci of breasts in other placental mammals – and often had nipple-like moles or extra hairs located atop the mounds. Therefore, focal fatty prominences on the fronts of human torsos likely represent chains of vestigial breasts composed of primordial breast fat.[70]
Behavioral
Humans also bear some vestigial behaviors and reflexes.[71]
Goose bumps

The formation of goose bumps in humans under stress is a vestigial reflex; a possible function in the distant evolutionary ancestors of humanity was to raise the body's hair, making the ancestor appear larger and scaring off predators.[72][71] Raising the hair is also used to trap an extra layer of air, keeping an animal warm.[71] Due to the diminished amount of hair in humans, the reflex formation of goose bumps when cold is also vestigial.[71]
Palmar grasp reflex
The palmar grasp reflex is thought to be a vestigial behavior in human infants. When placing a finger or object to the palm of an infant, it will securely grasp it. This grasp is found to be rather strong.[73] Some infants—37% according to a 1932 study—are able to support their own weight from a rod,[74] although there is no way they can cling to their mother. The grasp is also evident in the feet. When a baby is sitting down, its prehensile feet assume a curled-in posture, similar to that observed in an adult chimp.[75][76] An ancestral primate would have had sufficient body hair to which an infant could cling, unlike modern humans, thus allowing its mother to escape from danger, such as climbing up a tree in the presence of a predator without having to occupy her hands holding her baby.
Hiccup
It has been proposed that the hiccup is an evolutionary remnant of earlier amphibian respiration.[77] Amphibians such as tadpoles gulp air and water across their gills via a rather simple motor reflex akin to mammalian hiccuping. The motor pathways that enable hiccuping form early during fetal development, before the motor pathways that enable normal lung ventilation form. Additionally, hiccups and amphibian gulping are inhibited by elevated CO2 and may be stopped by GABAB receptor agonists, illustrating a possible shared physiology and evolutionary heritage. These proposals may explain why premature infants spend 2.5% of their time hiccuping, possibly gulping like amphibians, as their lungs are not yet fully formed. Fetal intrauterine hiccups are of two types. The physiological type occurs before 28 weeks after conception and tend to last five to ten minutes. These hiccups are part of fetal development and are associated with the myelination of the phrenic nerve, which primarily controls the thoracic diaphragm. The phylogeny hypothesis explains how the hiccup reflex might have evolved, and if there is not an explanation, it may explain hiccups as an evolutionary remnant, held-over from our amphibious ancestors. This hypothesis has been questioned because of the existence of the afferent loop of the reflex, the fact that it does not explain the reason for glottic closure, and because the very short contraction of the hiccup is unlikely to have a significant strengthening effect on the slow-twitch muscles of respiration.
Pseudogenes
There are many pseudogenes present in the human genome. One example of this is L-gulonolactone oxidase, a gene that is functional in most other mammals and produces an enzyme that synthesizes vitamin C.[78] In humans and other members of the suborder Haplorrhini, a mutation disabled the gene and made it unable to produce the enzyme. However, the remains of the gene are still present in the human genome.[79]
See also
References
- "Difference between rudimentary and vestigial organ - Biology - Evolution - 11741123 | Meritnation.com". www.meritnation.com. Retrieved 16 February 2021.
- Darwin C, The Descent of Man, and Selection in Relation to Sex, London: John Murray, 1890, p.13.
- Turner W, On the musculus sternalis, Proc. Royal Soc. Edinburgh session 1866–1867, p.65.
- Wiedersheim, R. (1893) The Structure of Man: An Index to His Past History. Second Edition. Translated by H. and M. Bernard. London: Macmillan and Co. 1895.
- Muller, G. B. (2002). "Vestigial Organs and Structures". In Pagel, Mark (ed.). Encyclopedia of Evolution. New York: Oxford University Press. pp. 1131–1133.
- Koerth-Baker, Maggie (30 July 2009). "Vestigial Organs Not So Useless After All". National Geographic. Retrieved 27 July 2013.
- Wells, H. G.; Huxley, J.; Wells, G. P. (1929). The Science of Life. Cassells.
- Rosenthal, M. I.: Journal of the American Medical Association, Volume 67, Issues 15–26, 1916. p. 1326
- W. Colin MacKenzie. "A Contribution to the Biology of the Vermiform Appendix". Medical Record, Volume 89, p. 342, 1916
- Darwin, Charles (1871). The Descent of Man, and Selection in Relation to Sex. John Murray: London.
- Kooij IA, Sahami S, Meijer SL, Buskens CJ, Te Velde AA (October 2016). "The immunology of the vermiform appendix: a review of the literature". Clinical and Experimental Immunology. 186 (1): 1–9. doi:10.1111/cei.12821. PMC 5011360. PMID 27271818.
- Stevens, C. Edward; Hume, Ian (2004). Comparative Physiology of the Vertebrate Digestive System. Cambridge: Cambridge University Press. ISBN 978-0-521-61714-7.
- Peter Robert Cheeke, Ellen S. Dierenfeld, Comparative Animal Nutrition and Metabolism. Publisher: CABI; 2010 ISBN 978-1-84593-631-0
- "Appendix may be useful after all – Health – Health care – More health news – NBC News". NBC News.
- Randal Bollinger, R.; Barbas, Andrew S.; Bush, Errol L.; Lin, Shu S.; Parker, William (2007). "Biofilms in the large bowel suggest an apparent function of the human vermiform appendix". Journal of Theoretical Biology. 249 (4): 826–31. Bibcode:2007JThBi.249..826R. doi:10.1016/j.jtbi.2007.08.032. PMID 17936308.
- Charles Q. Choi, "The Appendix: Useful and in Fact Promising", Live Science, 2009, Appendix has useful function
- Smith, H. F.; Fisher, R. E.; Everett, M. L.; Thomas, A. D.; Randal Bollinger, R.; Parker, W. (2009). "Comparative anatomy and phylogenetic distribution of the mammalian cecal appendix". Journal of Evolutionary Biology. 22 (10): 1984–99. doi:10.1111/j.1420-9101.2009.01809.x. PMID 19678866.
- Biswanath Mukhopadhyay, Ram M. Shukla, Madhumita Mukhopadhyay, Kartik C. Mandal, Pankaj Haldar, & Abhijit Benare (2012), "Spectrum of human tails: A report of six cases", Journal of Indian Association of Pediatric Surgeons, 17 (1): 23–25, doi:10.4103/0971-9261.91082, PMC 3263034, PMID 22279360
{{citation}}: CS1 maint: multiple names: authors list (link) - Saraga-Babić, Mirna; Lehtonen, Eero; Švajger, Anton; Wartiovaara, Jorma (1994). "Morphological and immunohistochemical characteristics of axial structures in the transitory human tail". Annals of Anatomy - Anatomischer Anzeiger. 176 (3): 277–86. doi:10.1016/S0940-9602(11)80496-6. PMID 8059973.
- Fallon, John F.; Simandl, B. Kay (1978). "Evidence of a role for cell death in the disappearance of the embryonic human tail". American Journal of Anatomy. 152 (1): 111–29. doi:10.1002/aja.1001520108. PMID 677043.
- Dao, Anh H.; Netsky, Martin G. (1984). "Human tails and pseudotails". Human Pathology. 15 (5): 449–53. doi:10.1016/S0046-8177(84)80079-9. PMID 6373560.
- Dubrow, Terry J.; Wackym, Phillip Ashley; Lesavoy, Malcolm A. (1988). "Detailing the Human Tail". Annals of Plastic Surgery. 20 (4): 340–4. doi:10.1097/00000637-198804000-00009. PMID 3284435.
- Spiegelmann, Roberto; Schinder, Edgardo; Mintz, Mordejai; Blakstein, Alexander (1985). "The human tail: A benign stigma". Journal of Neurosurgery. 63 (3): 461–2. doi:10.3171/jns.1985.63.3.0461. PMID 3894599.
- Johnson, Dr. George B. "Evidence for Evolution" Archived 10 March 2008 at the Wayback Machine. (Page 12) Txtwriter Inc. 8 June 2006.
- Rozkovcová, E; Marková, M; Dolejsí, J (1999). "Studies on agenesis of third molars amongst populations of different origin". Sbornik Lekarsky. 100 (2): 71–84. PMID 11220165.
- Pereira, T. V.; Salzano, F. M.; Mostowska, A.; Trzeciak, W. H.; Ruiz-Linares, A.; Chies, J. A. B.; Saavedra, C.; Nagamachi, C.; Hurtado, A. M.; Hill, K.; Castro-De-Guerra, D.; Silva-Junior, W. A.; Bortolini, M.-C. (2006). "Natural selection and molecular evolution in primate PAX9 gene, a major determinant of tooth development". Proceedings of the National Academy of Sciences. 103 (15): 5676–81. Bibcode:2006PNAS..103.5676P. doi:10.1073/pnas.0509562103. PMC 1458632. PMID 16585527.
- Trotier, D.; Eloit, C; Wassef, M; Talmain, G; Bensimon, J. L.; Døving, K. B.; Ferrand, J (2000). "The Vomeronasal Cavity in Adult Humans". Chemical Senses. 25 (4): 369–80. doi:10.1093/chemse/25.4.369. PMID 10944499.
- Kjær, Inger; Hansen, Birgit Fischer (1996). "The human vomeronasal organ: Prenatal developmental stages and distribution of luteinizing hormone-releasing hormone". European Journal of Oral Sciences. 104 (1): 34–40. doi:10.1111/j.1600-0722.1996.tb00043.x. PMID 8653495.
- Smith, Timothy D.; Siegel, Michael I.; Bhatnagar, Kunwar P. (2001). "Reappraisal of the vomeronasal system of catarrhine primates: Ontogeny, morphology, functionality, and persisting questions". The Anatomical Record. 265 (4): 176–192. doi:10.1002/ar.1152. PMID 11519019.
- Smith, Timothy D.; Bhatnagar, Kunwar P. (2000). "The human vomeronasal organ. Part II: Prenatal development". Journal of Anatomy. 197 (3): 421–436. doi:10.1046/j.1469-7580.2000.19730421.x. PMC 1468143. PMID 11117628.
- Won, J; Mair, E. A.; Bolger, W. E.; Conran, R. M. (2000). "The vomeronasal organ: an objective anatomic analysis of its prevalence". Ear, Nose, & Throat Journal. 79 (8): 600–605. doi:10.1177/014556130007900814. PMID 10969469.
- Johnson, A.; Josephson, R.; Hawke, M. (1985). "Clinical and histological evidence for the presence of the vomeronasal (Jacobson's) organ in adult humans". The Journal of Otolaryngology. 14 (2): 71–79. PMID 4068105.
- Foltán, René; Šedý, Jiří (2009). "Behavioral changes of patients after orthognathic surgery develop on the basis of the loss of vomeronasal organ: a hypothesis". Head & Face Medicine. 5: 5. doi:10.1186/1746-160X-5-5. PMC 2653472. PMID 19161592.
- Bhatnagar, Kunwar P.; Smith, Timothy D. (2001). "The human vomeronasal organ. III. Postnatal development from infancy to the ninth decade". Journal of Anatomy. 199 (Pt 3): 289–302. doi:10.1046/j.1469-7580.2001.19930289.x. PMC 1468331. PMID 11554506.
- Bhatnagar, Kunwar P.; Kennedy, Ray C.; Baron, Georg; Greenberg, Richard A. (1987). "Number of mitral cells and the bulb volume in the aging human olfactory bulb: A quantitative morphological study". The Anatomical Record. 218 (1): 73–87. doi:10.1002/ar.1092180112. PMID 3605663. S2CID 25630359.
- Witt, M.; Hummel, T. (2006). Vomeronasal Versus Olfactory Epithelium: Is There a Cellular Basis for Human Vomeronasal Perception?. International Review of Cytology. Vol. 248. pp. 209–259. doi:10.1016/S0074-7696(06)48004-9. ISBN 978-0-12-364652-1. PMID 16487792.
- Wysocki CJ, Preti G (November 2004). "Facts, fallacies, fears, and frustrations with human pheromones". The Anatomical Record Part A: Discoveries in Molecular, Cellular, and Evolutionary Biology. 281 (1): 1201–1211. doi:10.1002/ar.a.20125. PMID 15470677.
- Wyatt, Tristram D. (2003). Pheromones and Animal Behaviour: Communication by Smell and Taste. Cambridge: Cambridge University Press. p. 295. ISBN 978-0-521-48526-5.
- Prof. A. Macalister, Annals and Magazine of Natural History, vol. vii., 1871, p. 342.
- Bair, J. H. (1901). "Development of voluntary control". Psychological Review. 8 (5): 474–510. doi:10.1037/h0074157. hdl:2027/mdp.39015070189314.
- Mr. St. George Mivart, Elementary Anatomy, 1873, p. 396.
- Owen, R. 1866–1868. Comparative Anatomy and Physiology of Vertebrates. London.
- Montagna, W.; Machida, H.; Perkins, E. M. (1966). "The skin of primates. XXXIII. The skin of the angwantibo (Arctocebus calabarensis)". American Journal of Physical Anthropology. 25 (3): 277–90. doi:10.1002/ajpa.1330250307. PMID 5971502.
- Toerien, M. J.; Gous, A. E. (1978). "The orbital muscle of Müller". South African Medical Journal. 53 (4): 139–41. PMID 653491.
- Dutton, J.J., Atlas of Clinical and Surgical Orbital Anatomy, 2nd Edition, Elsevier, 2011. p.116-117.
- "Breast Anatomy and Embryology". Essentials of Plastic Surgery (2015): 355–361
- Hadjiathanasiou, C.G.; Brauner, R.; Lortat-Jacob, S.; Nivot, S.; Jaubert, F.; Fellous, M.; Nihoul-Fékété, C.; Rappaport, R. (1994). "True hermaphroditism: Genetic variants and clinical management". The Journal of Pediatrics. 125 (5): 738–744. doi:10.1016/S0022-3476(06)80172-1. PMID 7965425.
- Eren, Erdal; Edgünlü, Tuba; Asut, Emre; Karakaş Çelik, Sevim (2016). "Homozygous Ala65Pro Mutation with V89L Polymorphism in SRD5A2 Deficiency". Journal of Clinical Research in Pediatric Endocrinology. 8 (2): 218–223. doi:10.4274/jcrpe.2495. PMC 5096479. PMID 26761946.
- Healey, Andrew (2010). "Embryology of the Female Reproductive Tract". Imaging of Gynecological Disorders in Infants and Children. Medical Radiology. pp. 21–30. doi:10.1007/174_2010_128. ISBN 978-3-540-85601-6.
- Basaran, Mustafa; Usal, Deniz; Aydemir, Cumhur (2009). "Hymen Sparing Surgery for Imperforate Hymen: Case Reports and Review of Literature". Journal of Pediatric and Adolescent Gynecology. 22 (4): e61–64. doi:10.1016/j.jpag.2008.03.009. PMID 19646660.
- Collier, Roger (22 November 2011). "Vital or vestigial? The foreskin has its fans and foes". CMAJ. 183 (17): 1963–1964. doi:10.1503/cmaj.109-4014. ISSN 0820-3946. PMC 3225416. PMID 22025652.
- Kigozi G, Wawer M, Ssettuba A, et al. "Foreskin surface area and HIV acquisition in Rakai, Uganda (size matters)". AIDS. 2009; 23(16):2209–2213. 10.1097/QAD.0b013e328330eda8.
- Werker PMN, Terng ASC, Kon M. "The prepuce free flap: dissection feasibility study and clinical application of a super-thin new flap". Plastic Reconstructive Surgery. 1998; 102(4):1075–1082. 10.1097/00006534-199809020-00024.
- Darwin C. The Origin of Species by Means of Natural Selection. London, UK: John Murray; 1859.
- Macalister, Alexander (1875). "Additional Observations on Muscular Anomalies in Human Anatomy. (Third Series) With a Catalogue of the Principal Muscular Variations Hitherto Published". The Transactions of the Royal Irish Academy. 25: 1–134. JSTOR 30079154.
- Guerra, Aldo Benjamin; Metzinger, Stephen Eric; Metzinger, Rebecca Crawford; Xie, Chen; Xie, Yue; Rigby, Peter Lister; Naugle, Thomas (2004). "Variability of the Postauricular Muscle Complex". Archives of Facial Plastic Surgery. 6 (5): 342–7. doi:10.1001/archfaci.6.5.342. PMID 15381582.
- Tamatsu, Yuichi; Tsukahara, Kazue; Hotta, Mitsuyuki; Shimada, Kazuyuki (2007). "Vestiges of vibrissal capsular muscles exist in the human upper lip". Clinical Anatomy. 20 (6): 628–31. doi:10.1002/ca.20497. PMID 17458869. S2CID 21055062.
- Aversi-Ferreira, Roqueline A. G. M. F.; Bretas, Rafael Vieira; Maior, Rafael Souto; Davaasuren, Munkhzul; Paraguassú-Chaves, Carlos Alberto; Nishijo, Hisao; Aversi-Ferreira, Tales Alexandre (2014). "Morphometric and Statistical Analysis of the Palmaris Longus Muscle in Human and Non-Human Primates". BioMed Research International. 2014: 1–6. doi:10.1155/2014/178906. PMC 4016873. PMID 24860810.
- Kapoor, Sudhir K.; Tiwari, Akshay; Kumar, Abhishek; Bhatia, Rajesh; Tantuway, Vinay; Kapoor, Saurabh (2008). "Clinical relevance of palmaris longus agenesis: Common anatomical aberration". Anatomical Science International. 83 (1): 45–8. doi:10.1111/j.1447-073X.2007.00199.x. PMID 18402087. S2CID 25324691.
- Sebastin, S; Lim, A; Bee, W; Wong, T; Methil, B (2005). "Does the absence of the palmaris longus affect grip and pinch strength?". The Journal of Hand Surgery: Journal of the British Society for Surgery of the Hand. 30 (4): 406–8. doi:10.1016/j.jhsb.2005.03.011. PMID 15935531. S2CID 35394120.
- Rubinstein, David; Escott, Edward J.; Hendrick, Laura L. (April 1999). "The prevalence and CT appearance of the levator claviculae muscle: a normal variant not to be mistaken for an abnormality" (PDF). AJNR Am J Neuroradiol. 20 (4): 583–6. PMC 7056035. PMID 10319965.
- Loukas, M.; Sullivan, A.; Tubbs, R.S.; Shoja, M.M. (2008). "Levator claviculae: a case report and review of the literature". Folia Morphol. 67 (4): 307–310. PMID 19085875.
- Lovering, Richard M.; Anderson, Larry D. (2008). "Architecture and fiber type of the pyramidalis muscle". Anatomical Science International. 83 (4): 294–7. doi:10.1111/j.1447-073X.2007.00226.x. PMC 3531545. PMID 19159363.
- P., Haninec; R., Tomáš; R., Kaiser; R., Čihák (2009). "Development and clinical significance of the musculus dorsoepitrochlearis in men". Clinical Anatomy. 22 (4): 481–8. doi:10.1002/ca.20799. PMID 19373904. S2CID 221547558.
- Edwards, William E., The Musculoskeletal Anatomy of the Thorax and Brachium of an Adult Female Chimpanzee,6571st Aeromedical Research Laboratory, New Mexico, 1965. http://www.dtic.mil/dtic/tr/fulltext/u2/462433.pdf
- "Anatomy Atlases: Illustrated Encyclopedia of Human Anatomic Variation: Opus I: Muscular System: Alphabetical Listing of Muscles: L:Latissimus Dorsi".
- Ogata, Shigemitsu; Mine, Kazuharu; Tamatsu, Yuichi; Shimada, Kazuyuki (2002). "Morphological study of the human chondroglossus muscle in Japanese". Annals of Anatomy - Anatomischer Anzeiger. 184 (5): 493–9. doi:10.1016/S0940-9602(02)80087-5. PMID 12392330.
- Kajava, Y (1915). "The proportions of supernumerary nipples in the Finnish population". Duodecim. 1: 143–70.
- Goyal, Tarang; Bakshi, SK; Varshney, Anupam (2012). "Seven nipples in a male: World′s second case report". Indian Journal of Human Genetics. 18 (3): 373–5. doi:10.4103/0971-6866.108051. PMC 3656534. PMID 23716953.
- Teplica, David; Kovich, Grant; Srock, Jamey; Whitaker, Robert; Jeffers, Eileen; Wagstaff, David A. (14 October 2021). "Newly Identified Gross Human Anatomy: Eight Paired Vestigial Breast Mounds Run along the Embryological Mammary Ridges in Lean Adults". Plastic and Reconstructive Surgery - Global Open. 9 (10): e3863. doi:10.1097/GOX.0000000000003863. ISSN 2169-7574. PMC 8517303. PMID 34667697.
- Laura Spinney (2008), "Remnants of evolution", New Scientist, 198 (2656): 42–45, doi:10.1016/S0262-4079(08)61231-2
- Darwin, Charles. (1872) The Expression of the Emotions in Man and Animals John Murray, London.
- Peter Gray (2007). Psychology (fifth ed.). Worth Publishers. p. 66. ISBN 978-0-7167-0617-5.
- Behavior Development in Infants (via Google Books) by Evelyn Dewey, citing a study "Reflexes and other motor activities in newborn infants: a report of 125 cases as a preliminary study of infant behavior" published in the Bull. Neurol. Inst. New York, 1932, Vol. 2, pp. 1–56.
- Jerry Coyne (2009). Why Evolution is True. Penguin Group. pp. 85–86. ISBN 978-0-670-02053-9.
- Anthony Stevens (1982). Archetype: A Natural History of the Self. Routledge & Kegan Paul. p. 87. ISBN 978-0-7100-0980-7.
- Straus, C.; Vasilakos, K.; Wilson, R. J. A.; Oshima, T.; Zelter, M.; Derenne, J-Ph.; Similowski, T.; Whitelaw, W. A. (2003). "A phylogenetic hypothesis for the origin of hiccough". BioEssays. 25 (2): 182–8. doi:10.1002/bies.10224. PMID 12539245.
- Ohta, Yuriko; Nishikimi, Morimitsu (1999). "Random nucleotide substitutions in primate nonfunctional gene for l-gulono-γ-lactone oxidase, the missing enzyme in l-ascorbic acid biosynthesis". Biochimica et Biophysica Acta (BBA) - General Subjects. 1472 (1–2): 408–11. doi:10.1016/S0304-4165(99)00123-3. PMID 10572964.
- Nishikimi M, Fukuyama R, Minoshima S, Shimizu N, Yagi K (6 May 1994). "Cloning and chromosomal mapping of the human nonfunctional gene for L-gulono-gamma-lactone oxidase, the enzyme for L-ascorbic acid biosynthesis missing in man". J. Biol. Chem. 269 (18): 13685–8. doi:10.1016/S0021-9258(17)36884-9. PMID 8175804.
Further reading
- Shubin, Neil (2009). Your Inner Fish: A Journey into the 3.5-Billion-Year History of the Human Body. New York: Vintage Books. ISBN 978-0-307-27745-9.