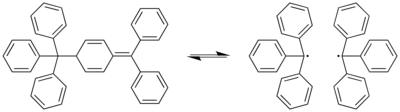
Gomberg's dimer
Gomberg's dimer is the organic compound with the formula Ph2C=C6H5-CPh3, where Ph = C6H5. It is a yellow solid that is air-stable for hours at room temperature and soluble in organic solvents.[1] The compound achieved fame as the dimer of triphenylmethyl radical, which was prepared by Moses Gomberg in his quest for hexaphenylethane.
 | |
.jpg.webp) | |
| Names | |
|---|---|
| Preferred IUPAC name
1,1′,1′′-{[4-(Diphenylmethylidene)cyclohexa-2,5-dien-1-yl]methanetriyl}tribenzene | |
| Other names
3-triphenylmethyl-6-diphenylmethylidene-1,4-cyclohexadiene | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C38H30 | |
| Molar mass | 486.658 g·mol−1 |
| Appearance | Yellow solid |
| Density | 1.16 g/cm3 |
| Melting point | 140–144 °C; 284–291 °F; 413–417 K |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Its quinoid structure has been determined by X-ray crystallography. The C-C bond that reversibly breaks is rather long at 159.7 picometers.[1]
Synthesis and reactions
Gomberg's dimer can be prepared quantitatively by treating trityl bromide with powdered copper or silver:[2]
- 2 Ph3CBr + 2 Cu → Ph2C=C6H5-CPh3 + 2 CuBr
Gomberg's dimer reversibly dissociates to the triphenylmethyl radical in organic solvents:[3]
See also
References
- Bochkarev, L. N.; Molosnova, N. E.; Zakharov, L. N.; Fukin, G. K.; Yanovsky, A. I.; Struchkov, Y. T. (1995). "1-Diphenylmethylene-4-(triphenylmethyl)cyclohexa-2,5-diene Benzene Solvate". Acta Crystallographica Section C Crystal Structure Communications. 51 (3): 489–491. doi:10.1107/S0108270194009005.
- Eisenberg, David C.; Lawrie, Christophe J. C.; Moody, Anne E.; Norton, Jack R. (1991). "Relative Rates of Hydrogen Atom (H.) Transfer from Transition-Metal Hydrides to Trityl Radicals". Journal of the American Chemical Society. 113 (13): 4888–4895. doi:10.1021/ja00013a026.
- Pangia, Thomas M.; Davies, Casey G.; Prendergast, Joshua R.; Gordon, Jesse B.; Siegler, Maxime A.; Jameson, Guy N. L.; Goldberg, David P. (28 March 2018). "Observation of Radical Rebound in a Mononuclear Nonheme Iron Model Complex". Journal of the American Chemical Society. 140 (12): 4191–4194. doi:10.1021/jacs.7b12707. PMC 6047074. PMID 29537258.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.