Green chemistry
Green chemistry, similar to sustainable chemistry or circular chemistry,[1] is an area of chemistry and chemical engineering focused on the design of products and processes that minimize or eliminate the use and generation of hazardous substances.[2] While environmental chemistry focuses on the effects of polluting chemicals on nature, green chemistry focuses on the environmental impact of chemistry, including lowering consumption of nonrenewable resources and technological approaches for preventing pollution.[3][4][5][6][7][8]
The overarching goals of green chemistry—namely, more resource-efficient and inherently safer design of molecules, materials, products, and processes—can be pursued in a wide range of contexts.
Green chemistry (sustainable chemistry): Design of chemical products and processes that minimize or eliminate the use or generation of substances hazardous to humans, animals, plants, and the environment. Note 1: Modified from ref.[9] to be more general.
Note 2: Green chemistry discusses the engineering concept of pollution prevention and zero waste both at laboratory and industrial scales. It encourages the use of economical and ecocompatible techniques that not only improve the yield but also bring down the cost of disposal of wastes at the end of a chemical process.[10]
History
Green chemistry emerged from a variety of existing ideas and research efforts (such as atom economy and catalysis) in the period leading up to the 1990s, in the context of increasing attention to problems of chemical pollution and resource depletion. The development of green chemistry in Europe and the United States was linked to a shift in environmental problem-solving strategies: a movement from command and control regulation and mandated lowering of industrial emissions at the "end of the pipe," toward the active prevention of pollution through the innovative design of production technologies themselves. The set of concepts now recognized as green chemistry coalesced in the mid- to late-1990s, along with broader adoption of the term (which prevailed over competing terms such as "clean" and "sustainable" chemistry).[11][12]
In the United States, the Environmental Protection Agency played a significant early role in fostering green chemistry through its pollution prevention programs, funding, and professional coordination. At the same time in the United Kingdom, researchers at the University of York contributed to the establishment of the Green Chemistry Network within the Royal Society of Chemistry, and the launch of the journal Green Chemistry.[12]
Principles
In 1998, Paul Anastas (who then directed the Green Chemistry Program at the US EPA) and John C. Warner (then of Polaroid Corporation) published a set of principles to guide the practice of green chemistry.[13] The twelve principles address a range of ways to lower the environmental and health impacts of chemical production, and also indicate research priorities for the development of green chemistry technologies.
The principles cover such concepts as:
- the design of processes to maximize the amount of raw material that ends up in the product;
- the use of renewable material feedstocks and energy sources;
- the use of safe, environmentally benign substances, including solvents, whenever possible;
- the design of energy efficient processes;
- avoiding the production of waste, which is viewed as the ideal form of waste management.
The twelve principles of green chemistry are:[14]
- Prevention: Preventing waste is better than treating or cleaning up waste after it is created.
- Atom economy: Synthetic methods should try to maximize the incorporation of all materials used in the process into the final product. This means that less waste will be generated as a result.
- Less hazardous chemical syntheses: Synthetic methods should avoid using or generating substances toxic to humans and/or the environment.
- Designing safer chemicals: Chemical products should be designed to achieve their desired function while being as non-toxic as possible.
- Safer solvents and auxiliaries: Auxiliary substances should be avoided wherever possible, and as non-hazardous as possible when they must be used.
- Design for energy efficiency: Energy requirements should be minimized, and processes should be conducted at ambient temperature and pressure whenever possible.
- Use of renewable feedstocks: Whenever it is practical to do so, renewable feedstocks or raw materials are preferable to non-renewable ones.
- Reduce derivatives: Unnecessary generation of derivatives—such as the use of protecting groups—should be minimized or avoided if possible; such steps require additional reagents and may generate additional waste.
- Catalysis: Catalytic reagents that can be used in small quantities to repeat a reaction are superior to stoichiometric reagents (ones that are consumed in a reaction).
- Design for degradation: Chemical products should be designed so that they do not pollute the environment; when their function is complete, they should break down into non-harmful products.
- Real-time analysis for pollution prevention: Analytical methodologies need to be further developed to permit real-time, in-process monitoring and control before hazardous substances form.
- Inherently safer chemistry for accident prevention: Whenever possible, the substances in a process, and the forms of those substances, should be chosen to minimize risks such as explosions, fires, and accidental releases.
Trends
Attempts are being made not only to quantify the greenness of a chemical process but also to factor in other variables such as chemical yield, the price of reaction components, safety in handling chemicals, hardware demands, energy profile and ease of product workup and purification. In one quantitative study,[15] the reduction of nitrobenzene to aniline receives 64 points out of 100 marking it as an acceptable synthesis overall whereas a synthesis of an amide using HMDS is only described as adequate with a combined 32 points.
Green chemistry is increasingly seen as a powerful tool that researchers must use to evaluate the environmental impact of nanotechnology.[16] As nano materials are developed, the environmental and human health impacts of both the products themselves and the processes to make them must be considered to ensure their long-term economic viability. There is a trend of nano material technology in the practice, however, people ignored the potential nanotoxicity. Therefore, people need to address further consideration on legal, ethical, safety, and regulatory issues associated with nanomaterials, [17]
Examples
Green solvents
The major application of solvents in human activities is in paints and coatings (46% of usage). Smaller volume applications include cleaning, de-greasing, adhesives, and in chemical synthesis.[18] Traditional solvents are often toxic or are chlorinated. Green solvents, on the other hand, are generally less harmful to health and the environment and preferably more sustainable. Ideally, solvents would be derived from renewable resources and biodegrade to innocuous, often a naturally occurring product.[19][20] However, the manufacture of solvents from biomass can be more harmful to the environment than making the same solvents from fossil fuels.[21] Thus the environmental impact of solvent manufacture must be considered when a solvent is being selected for a product or process.[22] Another factor to consider is the fate of the solvent after use. If the solvent is being used in an enclosed situation where solvent collection and recycling is feasible, then the energy cost and environmental harm associated with recycling should be considered; in such a situation water, which is energy-intensive to purify, may not be the greenest choice. On the other hand, a solvent contained in a consumer product is likely to be released into the environment upon use, and therefore the environmental impact of the solvent itself is more important than the energy cost and impact of solvent recycling; in such a case water is very likely to be a green choice. In short, the impact of the entire lifetime of the solvent, from cradle to grave (or cradle to cradle if recycled) must be considered. Thus the most comprehensive definition of a green solvent is the following: "a green solvent is the solvent that makes a product or process have the least environmental impact over its entire life cycle."[23]
By definition, then, a solvent might be green for one application (because it results in less environmental harm than any other solvent that could be used for that application) and yet not be a green solvent for a different application. A classic example is water, which is a very green solvent for consumer products such as toilet bowl cleaner but is not a green solvent for the manufacture of polytetrafluoroethylene. For the production of that polymer, the use of water as solvent requires the addition of perfluorinated surfactants which are highly persistent. Instead, supercritical carbon dioxide seems to be the greenest solvent for that application because it performs well without any surfactant.[23] In summary, no solvent can be declared to be a "green solvent" unless the declaration is limited to a specific application.
Synthetic techniques
Novel or enhanced synthetic techniques can often provide improved environmental performance or enable better adherence to the principles of green chemistry. For example, the 2005 Nobel Prize for Chemistry was awarded to Yves Chauvin, Robert H. Grubbs and Richard R. Schrock, for the development of the metathesis method in organic synthesis, with explicit reference to its contribution to green chemistry and "smarter production."[24] A 2005 review identified three key developments in green chemistry in the field of organic synthesis: use of supercritical carbon dioxide as green solvent, aqueous hydrogen peroxide for clean oxidations and the use of hydrogen in asymmetric synthesis.[25] Some further examples of applied green chemistry are supercritical water oxidation, on water reactions, and dry media reactions.
Bioengineering is also seen as a promising technique for achieving green chemistry goals. A number of important process chemicals can be synthesized in engineered organisms, such as shikimate, a Tamiflu precursor which is fermented by Roche in bacteria. Click chemistry is often cited as a style of chemical synthesis that is consistent with the goals of green chemistry. The concept of 'green pharmacy' has recently been articulated based on similar principles.[26]
Carbon dioxide as blowing agent
In 1996, Dow Chemical won the 1996 Greener Reaction Conditions award for their 100% carbon dioxide blowing agent for polystyrene foam production. Polystyrene foam is a common material used in packing and food transportation. Seven hundred million pounds are produced each year in the United States alone. Traditionally, CFC and other ozone-depleting chemicals were used in the production process of the foam sheets, presenting a serious environmental hazard. Flammable, explosive, and, in some cases toxic hydrocarbons have also been used as CFC replacements, but they present their own problems. Dow Chemical discovered that supercritical carbon dioxide works equally as well as a blowing agent, without the need for hazardous substances, allowing the polystyrene to be more easily recycled. The CO2 used in the process is reused from other industries, so the net carbon released from the process is zero.
Hydrazine
Addressing principle #2 is the peroxide process for producing hydrazine without cogenerating salt. Hydrazine is traditionally produced by the Olin Raschig process from sodium hypochlorite (the active ingredient in many bleaches) and ammonia. The net reaction produces one equivalent of sodium chloride for every equivalent of the targeted product hydrazine:[27]
- NaOCl + 2 NH3 → H2N-NH2 + NaCl + H2O
In the greener peroxide process hydrogen peroxide is employed as the oxidant and the side product is water. The net conversion follows:
- 2 NH3 + H2O2 → H2N-NH2 + 2 H2O
Addressing principle #4, this process does not require auxiliary extracting solvents. Methyl ethyl ketone is used as a carrier for the hydrazine, the intermediate ketazine phase separates from the reaction mixture, facilitating workup without the need of an extracting solvent.
1,3-Propanediol
Addressing principle #7 is a green route to 1,3-propanediol, which is traditionally generated from petrochemical precursors. It can be produced from renewable precursors via the bioseparation of 1,3-propanediol using a genetically modified strain of E. coli.[28] This diol is used to make new polyesters for the manufacture of carpets.
Lactide
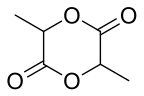
In 2002, Cargill Dow (now NatureWorks) won the Greener Reaction Conditions Award for their improved method for polymerization of polylactic acid . Unfortunately, lactide-base polymers do not perform well and the project was discontinued by Dow soon after the award. Lactic acid is produced by fermenting corn and converted to lactide, the cyclic dimer ester of lactic acid using an efficient, tin-catalyzed cyclization. The L,L-lactide enantiomer is isolated by distillation and polymerized in the melt to make a crystallizable polymer, which has some applications including textiles and apparel, cutlery, and food packaging. Wal-Mart has announced that it is using/will use PLA for its produce packaging. The NatureWorks PLA process substitutes renewable materials for petroleum feedstocks, doesn't require the use of hazardous organic solvents typical in other PLA processes, and results in a high-quality polymer that is recyclable and compostable.
Carpet tile backings
In 2003 Shaw Industries selected a combination of polyolefin resins as the base polymer of choice for EcoWorx due to the low toxicity of its feedstocks, superior adhesion properties, dimensional stability, and its ability to be recycled. The EcoWorx compound also had to be designed to be compatible with nylon carpet fiber. Although EcoWorx may be recovered from any fiber type, nylon-6 provides a significant advantage. Polyolefins are compatible with known nylon-6 depolymerization methods. PVC interferes with those processes. Nylon-6 chemistry is well-known and not addressed in first-generation production. From its inception, EcoWorx met all of the design criteria necessary to satisfy the needs of the marketplace from a performance, health, and environmental standpoint. Research indicated that separation of the fiber and backing through elutriation, grinding, and air separation proved to be the best way to recover the face and backing components, but an infrastructure for returning postconsumer EcoWorx to the elutriation process was necessary. Research also indicated that the postconsumer carpet tile had a positive economic value at the end of its useful life. EcoWorx is recognized by MBDC as a certified cradle-to-cradle design.

Transesterification of fats
In 2005, Archer Daniels Midland (ADM) and Novozymes won the Greener Synthetic Pathways Award for their enzyme interesterification process. In response to the U.S. Food and Drug Administration (FDA) mandated labeling of trans-fats on nutritional information by January 1, 2006, Novozymes and ADM worked together to develop a clean, enzymatic process for the interesterification of oils and fats by interchanging saturated and unsaturated fatty acids. The result is commercially viable products without trans-fats. In addition to the human health benefits of eliminating trans-fats, the process has reduced the use of toxic chemicals and water, prevents vast amounts of byproducts, and reduces the amount of fats and oils wasted.
Bio-succinic acid
In 2011, the Outstanding Green Chemistry Accomplishments by a Small Business Award went to BioAmber Inc. for integrated production and downstream applications of bio-based succinic acid. Succinic acid is a platform chemical that is an important starting material in the formulations of everyday products. Traditionally, succinic acid is produced from petroleum-based feedstocks. BioAmber has developed process and technology that produces succinic acid from the fermentation of renewable feedstocks at a lower cost and lower energy expenditure than the petroleum equivalent while sequestering CO2 rather than emitting it.[29] However, lower prices of oil precipitated the company into bankruptcy [30] and bio-sourced succinic acid is now barely made.[31]
Laboratory chemicals
Several laboratory chemicals are controversial from the perspective of Green chemistry. The Massachusetts Institute of Technology created a "Green" Alternatives Wizard to help identify alternatives. Ethidium bromide, xylene, mercury, and formaldehyde have been identified as "worst offenders" which have alternatives.[32] Solvents in particular make a large contribution to the environmental impact of chemical manufacturing and there is a growing focus on introducing Greener solvents into the earliest stage of development of these processes: laboratory-scale reaction and purification methods.[33] In the Pharmaceutical Industry, both GSK[34] and Pfizer[35] have published Solvent Selection Guides for their Drug Discovery chemists.
Legislation
The EU
In 2007, The EU put into place the Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH) program, which requires companies to provide data showing that their products are safe. This regulation (1907/2006) ensures not only the assessment of the chemicals' hazards as well as risks during their uses but also includes measures for banning or restricting/authorising uses of specific substances. ECHA, the EU Chemicals Agency in Helsinki, is implementing the regulation whereas the enforcement lies with the EU member states.
United States
The United States formed the Environmental Protection Agency (EPA) in 1970 to protect human and environmental health by creating and enforcing environmental regulation. Green chemistry builds on the EPA’s goals by encouraging chemists and engineers to design chemicals, processes, and products that avoid the creation of toxins and waste.[36]
The U.S. law that governs the majority of industrial chemicals (excluding pesticides, foods, and pharmaceuticals) is the Toxic Substances Control Act (TSCA) of 1976. Examining the role of regulatory programs in shaping the development of green chemistry in the United States, analysts have revealed structural flaws and long-standing weaknesses in TSCA; for example, a 2006 report to the California Legislature concludes that TSCA has produced a domestic chemicals market that discounts the hazardous properties of chemicals relative to their function, price, and performance.[37] Scholars have argued that such market conditions represent a key barrier to the scientific, technical, and commercial success of green chemistry in the U.S., and fundamental policy changes are needed to correct these weaknesses.[38]
Passed in 1990, the Pollution Prevention Act helped foster new approaches for dealing with pollution by preventing environmental problems before they happen.
Green chemistry grew in popularity in the United States after the Pollution Prevention Act of 1990 was passed. This Act declared that pollution should be lowered by improving designs and products rather than treatment and disposal. These regulations encouraged chemists to reimagine pollution and research ways to limit the toxins in the atmosphere. In 1991, the EPA Office of Pollution Prevention and Toxics created a research grant program encouraging the research and recreation of chemical products and processes to limit the impact on the environment and human health.[39] The EPA hosts The Green Chemistry Challenge each year to incentivize the economic and environmental benefits of developing and utilizing green chemistry.[40]
In 2008, the State of California approved two laws aiming to encourage green chemistry, launching the California Green Chemistry Initiative. One of these statutes required California's Department of Toxic Substances Control (DTSC) to develop new regulations to prioritize "chemicals of concern" and promote the substitution of hazardous chemicals with safer alternatives. The resulting regulations took effect in 2013, initiating DTSC's Safer Consumer Products Program.[41]
Scientific journals specialized in green chemistry
Contested definition
There are ambiguities in the definition of green chemistry, and in how it is understood among broader science, policy, and business communities. Even within chemistry, researchers have used the term "green chemistry" to describe a range of work independently of the framework put forward by Anastas and Warner (i.e., the 12 principles).[12] While not all uses of the term are legitimate (see greenwashing), many are, and the authoritative status of any single definition is uncertain. More broadly, the idea of green chemistry can easily be linked (or confused) with related concepts like green engineering, environmental design, or sustainability in general. The complexity and multifaceted nature of green chemistry makes it difficult to devise clear and simple metrics. As a result, "what is green" is often open to debate.[42]
Awards
Several scientific societies have created awards to encourage research in green chemistry.
- Australia's Green Chemistry Challenge Awards overseen by The Royal Australian Chemical Institute (RACI).
- The Canadian Green Chemistry Medal.[43]
- In Italy, Green Chemistry activities center around an inter-university consortium known as INCA.[44]
- In Japan, The Green & Sustainable Chemistry Network oversees the GSC awards program.[45]
- In the United Kingdom, the Green Chemical Technology Awards are given by Crystal Faraday.[46]
- In the US, the Presidential Green Chemistry Challenge Awards recognize individuals and businesses.[47][48]
See also
- Bioremediation – a technique that generally falls outside the scope of green chemistry
- Environmental engineering science
- Green Chemistry (journal) – published by the Royal Society of Chemistry
- Green chemistry metrics
- Green computing – a similar initiative in the area of computing
- Green engineering
- Substitution of dangerous chemicals
- Sustainable engineering
References
- Mutlu, Hatice; Barner, Leonie (2022-06-03). "Getting the Terms Right: Green, Sustainable, or Circular Chemistry?". Macromolecular Chemistry and Physics. 223 (13): 2200111. doi:10.1002/macp.202200111. ISSN 1022-1352. S2CID 249357642.
- "Green Chemistry". United States Environmental Protection Agency. 2006-06-28. Retrieved 2011-03-23.
- Sheldon, R. A.; Arends, I. W. C. E.; Hanefeld, U. (2007). Green Chemistry and Catalysis (PDF). doi:10.1002/9783527611003. ISBN 9783527611003. S2CID 92947071.
- Clark, J. H.; Luque, R.; Matharu, A. S. (2012). "Green Chemistry, Biofuels, and Biorefinery". Annual Review of Chemical and Biomolecular Engineering. 3: 183–207. doi:10.1146/annurev-chembioeng-062011-081014. PMID 22468603.
- Cernansky, R. (2015). "Chemistry: Green refill". Nature. 519 (7543): 379–380. doi:10.1038/nj7543-379a. PMID 25793239.
- Sanderson, K. (2011). "Chemistry: It's not easy being green". Nature. 469 (7328): 18–20. Bibcode:2011Natur.469...18S. doi:10.1038/469018a. PMID 21209638.
- Poliakoff, M.; Licence, P. (2007). "Sustainable technology: Green chemistry". Nature. 450 (7171): 810–812. Bibcode:2007Natur.450..810P. doi:10.1038/450810a. PMID 18064000. S2CID 12340643.
- Clark, J. H. (1999). "Green chemistry: Challenges and opportunities". Green Chemistry. 1: 1–8. doi:10.1039/A807961G.
- Marteel, Anne E.; Davies, Julian A.; Olson, Walter W.; Abraham, Martin A. (2003). "GREEN CHEMISTRY AND ENGINEERING: Drivers, Metrics, and Reduction to Practice". Annual Review of Environment and Resources. 28: 401–428. doi:10.1146/annurev.energy.28.011503.163459.
- Vert, Michel; Doi, Yoshiharu; Hellwich, Karl-Heinz; Hess, Michael; Hodge, Philip; Kubisa, Przemyslaw; Rinaudo, Marguerite; Schué, François (2012). "Terminology for biorelated polymers and applications (IUPAC Recommendations 2012)" (PDF). Pure and Applied Chemistry. 84 (2): 377–410. doi:10.1351/PAC-REC-10-12-04. S2CID 98107080.
- Woodhouse, E. J.; Breyman, S. (2005). "Green chemistry as social movement?". Science, Technology, & Human Values. 30 (2): 199–222. doi:10.1177/0162243904271726. S2CID 146774456.
- Linthorst, J. A. (2009). "An overview: Origins and development of green chemistry". Foundations of Chemistry. 12: 55–68. doi:10.1007/s10698-009-9079-4.
- Anastas, Paul T.; Warner, John C. (1998). Green chemistry: theory and practice. Oxford [England]; New York: Oxford University Press. ISBN 9780198502340.
- "12 Principles of Green Chemistry - American Chemical Society". American Chemical Society. Retrieved 2018-02-16.
- Van Aken, K.; Strekowski, L.; Patiny, L. (2006). "EcoScale, a semi-quantitative tool to select an organic preparation based on economical and ecological parameters". Beilstein Journal of Organic Chemistry. 2 (1): 3. doi:10.1186/1860-5397-2-3. PMC 1409775. PMID 16542013.
- "Green nanotechnology" (PDF). Archived from the original (PDF) on 2016-04-06. Retrieved 2008-03-01.
- Hemant Kumar Daima; Shanker Lal Kothari; Bhargava Suresh Kumar, eds. (2021). Nanotoxicology toxicity evaluation of nanomedicine applications. Boca Raton. ISBN 978-1-000-39991-2. OCLC 1256699945.
{{cite book}}: CS1 maint: location missing publisher (link) - Torok, Bela (2017). Green Chemistry: An Inclusive Approach. Amsterdam: Elsevier. p. Ch 3.15.
- Prat, D.; Pardigon, O.; Flemming, H.-W.; Letestu, S.; Ducandas, V.; Isnard, P.; Guntrum, E.; Senac, T.; Ruisseau, S.; Cruciani, P.; Hosek, P. (2013). "Sanofi's Solvent Selection Guide: A Step Toward More Sustainable Processes". Org. Process Res. Dev. 17 (12): 1517–1525. doi:10.1021/op4002565.
- Sherman, J.; Chin, B.; Huibers, P. D. T.; Garcia-Valls, R.; Hatton, T. A. (1998). "Solvent Replacement for Green Processing". Environ. Health Perspect. 106 (Suppl 1): 253–271. doi:10.2307/3433925. JSTOR 3433925. PMC 1533296. PMID 9539018.
- Isoni, V. (2016). "Q-SAOESS: A methodology to help solvent selection for pharmaceutical manufacture at the early process development stage". Green Chem. 18: 6564. doi:10.1039/C6GC02440H.
- Clarke, Coby J.; Tu, Wei-Chien; Levers, Oliver; Brohl, Andreas; Hallett, Jason P. (2018). "Green and Sustainable Solvents in Chemical Processes". Chemical Reviews. 118 (2): 747–800. doi:10.1021/acs.chemrev.7b00571. hdl:10044/1/59694. PMID 29300087.
- Jessop, Philip (2017). "Green/Alternative Solvents". In Abraham, M. A. (ed.). Encyclopedia of Sustainable Technologies. Elsevier. pp. 611–619. ISBN 9780128046777.
- "The Nobel Prize in Chemistry 2005". The Nobel Foundation. Retrieved 2006-08-04.
- Noyori, R. (2005). "Pursuing practical elegance in chemical synthesis". Chemical Communications (14): 1807–11. doi:10.1039/B502713F. PMID 15795753.
- Baron, M. (2012). "Towards a Greener Pharmacy by More Eco Design" (PDF). Waste and Biomass Valorization. 3 (4): 395–407. doi:10.1007/s12649-012-9146-2. S2CID 109584470.
- Jean-Pierre Schirmann, Paul Bourdauducq "Hydrazine" in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2002. doi:10.1002/14356007.a13_177.
- Kurian, Joseph V (2005). "A New Polymer Platform for the Future – Sorona from Corn Derived 1,3-Propanediol". Journal of Polymers and the Environment. 13 (2): 159–167. doi:10.1007/s10924-005-2947-7. S2CID 137246045.
- "2011 Small Business Award". United States Environmental Protection Agency. 2013-03-12.
- "Succinic acid maker BioAmber is bankrupt". Chemical & Engineering News. 2018-05-13.
- "Succinic acid, once a biobased chemical star, is barely being made". Chemical & Engineering News. 2019-03-20.
- Coombs A. (2009). Green at the Bench Archived 2009-07-10 at the Wayback Machine. The Scientist.
- Bradley, Jean-Claude; Abraham, Michael H.; Acree, William E.; Lang, Andrew (2015). "Predicting Abraham model solvent coefficients". Chemistry Central Journal. 9: 12. doi:10.1186/s13065-015-0085-4. PMC 4369285. PMID 25798192.
- Henderson, R. K.; Jiménez-González, C. N.; Constable, D. J. C.; Alston, S. R.; Inglis, G. G. A.; Fisher, G.; Sherwood, J.; Binks, S. P.; Curzons, A. D. (2011). "Expanding GSK's solvent selection guide – embedding sustainability into solvent selection starting at medicinal chemistry". Green Chemistry. 13 (4): 854. doi:10.1039/c0gc00918k. S2CID 56376990.
- Alfonsi, K.; Colberg, J.; Dunn, P. J.; Fevig, T.; Jennings, S.; Johnson, T. A.; Kleine, H. P.; Knight, C.; Nagy, M. A.; Perry, D. A.; Stefaniak, M. (2008). "Green chemistry tools to influence a medicinal chemistry and research chemistry based organisation". Green Chem. 10: 31–36. doi:10.1039/B711717E. S2CID 97175218.
- "What Is Green Chemistry?". American Chemical Society. Retrieved 2021-01-29.
- Wilson, M. P.; Chia, D. A.; Ehlers, B. C. (2006). "Green chemistry in California: a framework for leadership in chemicals policy and innovation" (PDF). New Solutions. 16 (4): 365–372. doi:10.2190/9584-1330-1647-136p. PMID 17317635. S2CID 43455643.
- Wilson, M. P.; Schwarzman, M. R. (2009). "Toward a new U.S. Chemicals policy: Rebuilding the foundation to advance new science, green chemistry, and environmental health". Environmental Health Perspectives. 117 (8): 1202–9. doi:10.1289/ehp.0800404. PMC 2721862. PMID 19672398.
- "History of Green Chemistry | Center for Green Chemistry & Green Engineering at Yale". greenchemistry.yale.edu. Retrieved 2021-01-29.
- US EPA, OCSPP (2013-02-13). "Information About the Green Chemistry Challenge". US EPA. Retrieved 2021-01-29.
- California Department of Toxic Substances Control. "What is the Safer Consumer Products (SCP) Program?". Retrieved 5 September 2015.
- Matus, K. J. M.; Clark, W. C.; Anastas, P. T.; Zimmerman, J. B. (2012). "Barriers to the Implementation of Green Chemistry in the United States" (PDF). Environmental Science & Technology. 46 (20): 10892–10899. Bibcode:2012EnST...4610892M. doi:10.1021/es3021777. PMID 22963612.
- "Announcing the 2005 Canadian Green Chemistry Medal". RSC Publishing. Retrieved 2006-08-04.
- "Chemistry for the Environment". Interuniversity Consortium. Retrieved 2007-02-15.
- "Green & Sustainable Chemistry Network, Japan". Green & Sustainable Chemistry Network. Archived from the original on 2001-05-13. Retrieved 2006-08-04.
- "2005 Crystal Faraday Green Chemical Technology Awards". Green Chemistry Network. Archived from the original on 2002-12-17. Retrieved 2006-08-04.
- "The Presidential Green Chemistry Awards". United States Environmental Protection Agency. Retrieved 2006-07-31.
- "Information about the Presidential Green Chemistry Challenge". 2013-02-13. Retrieved 2014-08-10.