Inverse agonist
In pharmacology, an inverse agonist is a drug that binds to the same receptor as an agonist but induces a pharmacological response opposite to that of the agonist.
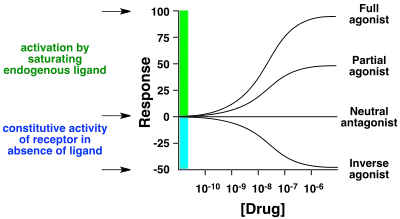
A neutral antagonist has no activity in the absence of an agonist or inverse agonist but can block the activity of either;[1] they are in fact sometimes called blockers (examples include alpha blockers, beta blockers, and calcium channel blockers). Inverse agonists have opposite actions to those of agonists but the effects of both of these can be blocked by antagonists.[2]
A prerequisite for an inverse agonist response is that the receptor must have a constitutive (also known as intrinsic or basal) level of activity in the absence of any ligand.[3] An agonist increases the activity of a receptor above its basal level, whereas an inverse agonist decreases the activity below the basal level.
The efficacy of a full agonist is by definition 100%, a neutral antagonist has 0% efficacy, and an inverse agonist has < 0% (i.e., negative) efficacy.
Examples
Receptors for which inverse agonists have been identified include the GABAA, melanocortin, mu opioid, histamine and beta adrenergic receptors. Both endogenous and exogenous inverse agonists have been identified, as have drugs at ligand gated ion channels and at G protein-coupled receptors.
Ligand gated ion channel inverse agonists
An example of a receptor site that possesses basal activity and for which inverse agonists have been identified is the GABAA receptors. Agonists for GABAA receptors (such as muscimol) create a relaxant effect, whereas inverse agonists have agitation effects (for example, Ro15-4513) or even convulsive and anxiogenic effects (certain beta-carbolines).[4][5]
G protein-coupled receptor inverse agonists
Two known endogenous inverse agonists are the Agouti-related peptide (AgRP) and its associated peptide Agouti signalling peptide (ASIP). AgRP and ASIP appear naturally in humans and bind melanocortin receptors 4 and 1 (Mc4R and Mc1R), respectively, with nanomolar affinities.[6]
The opioid antagonists naloxone and naltrexone act as neutral antagonists of the mu opioid receptors under basal conditions, but as inverse agonists when an opioid such as morphine as bound to the same channel. 6α-naltrexo, 6β-naltrexol, 6β-naloxol, and 6β-naltrexamine acted neutral antagonists regardless of opioid binding and caused significantly reduced withdrawal jumping when compared to naloxone and naltrexone.[7]
Nearly all antihistamines acting at H1 receptors and H2 receptors have been shown to be inverse agonists.[8]
The beta blockers carvedilol and bucindolol have been shown to be low level inverse agonists at beta adrenoceptors.[8]
Mechanisms of action
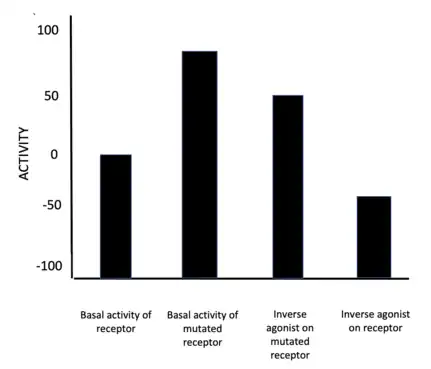
Like agonists, inverse agonists have their own unique ways of inducing pharmacological and physiological responses depending on many factors, such as the type of inverse agonist, the type of receptor, mutants of receptors, binding affinities and whether the effects are exerted acutely or chronically based on receptor population density.[9] Because of this, they exhibit a spectrum of activity below the Intrinsic activity level.[9][10] Changes in constitutive activity of receptors affect response levels from ligands like inverse agonists.[11]
To illustrate, mechanistic models have been made for how inverse agonists induce their responses on G protein-coupled receptors (GPCRs). Many types of Inverse agonists for GPCRs have been shown to exhibit the following conventionally accepted mechanism.
Based on the Extended Ternary complex model, the mechanism contends that inverse agonists switch the receptor from an active state to an inactive state by undergoing conformational changes.[12] Under this model, current thinking is that the GPCRs can exist in a continuum of active and inactive states when no ligand is present.[12] Inverse agonists stabilize the inactive states, thereby suppressing agonist-independent activity.[12] However, the implementation of 'constitutively active mutants'[12] of GPCRs change their intrinsic activity.[9][10] Thus, the effect an inverse agonist has on a receptor depends on the basal activity of the receptor, assuming the inverse agonist has the same binding affinity (as shown in the figure 2).
See also
References
- Kenakin T (April 2004). "Principles: receptor theory in pharmacology". Trends in Pharmacological Sciences. 25 (4): 186–92. doi:10.1016/j.tips.2004.02.012. PMID 15063082.
- Nutt D, Stahl S, Blier P, Drago F, Zohar J, Wilson S (January 2017). "Inverse agonists - What do they mean for psychiatry?". European Neuropsychopharmacology. 27 (1): 87–90. doi:10.1016/j.euroneuro.2016.11.013. hdl:10044/1/43624. PMID 27955830. S2CID 25113284.
- Berg, Kelly A; Clarke, William P (2018-08-06). "Making Sense of Pharmacology: Inverse Agonism and Functional Selectivity". International Journal of Neuropsychopharmacology. 21 (10): 962–977. doi:10.1093/ijnp/pyy071. ISSN 1461-1457. PMC 6165953. PMID 30085126.
- Mehta AK, Ticku MK (August 1988). "Ethanol potentiation of GABAergic transmission in cultured spinal cord neurons involves gamma-aminobutyric acid voltage-gated chloride channels". The Journal of Pharmacology and Experimental Therapeutics. 246 (2): 558–64. PMID 2457076. Archived from the original on 2021-05-31. Retrieved 2008-04-21.
- Sieghart W (January 1994). "Pharmacology of benzodiazepine receptors: an update". Journal of Psychiatry & Neuroscience. 19 (1): 24–9. PMC 1188559. PMID 8148363.
- Ollmann MM, Lamoreux ML, Wilson BD, Barsh GS (February 1998). "Interaction of Agouti protein with the melanocortin 1 receptor in vitro and in vivo". Genes & Development. 12 (3): 316–30. doi:10.1101/gad.12.3.316. PMC 316484. PMID 9450927.
- Wang OD, Raehal KM, Bilsky EJ, Sadée W (June 2001). "Inverse agonists and neutral antagonists at mu opioid receptor (MOR): possible role of basal receptor signaling in narcotic dependence". Journal of Neurochemistry. 77 (3): 1590–600. doi:10.1046/j.1471-4159.2001.00362.x. PMID 11413242. S2CID 10026688.
- Khilnani G, Khilnani AK (2011). "Inverse agonism and its therapeutic significance". Indian J Pharmacol. 43 (5): 492–501. doi:10.4103/0253-7613.84947. PMC 3195115. PMID 22021988.
- Prather, Paul L. (2004-01-05). "Inverse agonists: tools to reveal ligand-specific conformations of G protein-coupled receptors". Science's STKE: Signal Transduction Knowledge Environment. 2004 (215): pe1. doi:10.1126/stke.2152004pe1. ISSN 1525-8882. PMID 14722344. S2CID 22336235.
- Hirayama, Shigeto; Fujii, Hideaki (2020). "δ Opioid Receptor Inverse Agonists and their In Vivo Pharmacological Effects". Current Topics in Medicinal Chemistry. 20 (31): 2889–2902. doi:10.2174/1568026620666200402115654. ISSN 1873-4294. PMID 32238139. S2CID 214767114.
- Berg, Kelly A.; Clarke, William P. (2018-10-01). "Making Sense of Pharmacology: Inverse Agonism and Functional Selectivity". The International Journal of Neuropsychopharmacology. 21 (10): 962–977. doi:10.1093/ijnp/pyy071. ISSN 1469-5111. PMC 6165953. PMID 30085126.
- Strange, Philip G. (February 2002). "Mechanisms of inverse agonism at G-protein-coupled receptors". Trends in Pharmacological Sciences. 23 (2): 89–95. doi:10.1016/s0165-6147(02)01993-4. ISSN 0165-6147. PMID 11830266.
External links
- Jeffries WB (1999-02-17). "Inverse Agonists for Medical Students". Office of Medical Education - Courses - IDC 105 Principles of Pharmacology. Creighton University School of Medicine - Department of Pharmacology. Retrieved 2008-08-12.
- Inverse Agonists: An Illustrated Tutorial Panesar K, Guzman F. Pharmacology Corner. 2012