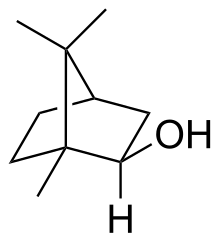
Isoborneol
Isoborneol is a bicyclic organic compound and a terpene derivative. The hydroxyl group in this compound is placed in an exo position. The endo diastereomer is called borneol. Being chiral, isoborneol exists as enantiomers.
 | |
| Names | |
|---|---|
| IUPAC name
(1S,2S,4S)-1,7,7-trimethylbicyclo[2.2.1]heptane-2-ol, (1R,2R,4R)-1,7,7-trimethylbicyclo[2.2.1]heptane-2-ol | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChEBI |
|
| ChEMBL | |
| ChemSpider | |
PubChem CID |
|
| UNII |
|
| UN number | 1312 |
| |
| |
| Properties | |
| C10H18O | |
| Molar mass | 154.253 g·mol−1 |
| Appearance | white or colorless solid |
| Melting point | 212–214 °C (414–417 °F; 485–487 K) + or -; 210–215 °C for rac |
| Hazards | |
| GHS labelling:[1] | |
 | |
| Warning | |
| H228 | |
| P210, P240, P241, P280, P370+P378 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Preparation
Isoborneol is synthesized commercially by hydrolysis of isobornyl acetate. The latter is obtained from treatment of camphene with acetic acid in the presence of a strong acid catalyst.[2]
It can also be produced by reduction of camphor:
Isoborneol derivatives as chiral ligands
Derivatives of isoborneol are used as ligands in asymmetric synthesis.[3]
- (2S)-(−)-3-exo-(morpholino)isoborneol or MIB[4] with a morpholine substituent in the α-hydroxyl position.
- (2S)-(−)-3-exo-(dimethylamino)isoborneol or DAIB[5] with a dimethylamino substituent in the α-hydroxyl position
References
- "(+)-Isoborneol". pubchem.ncbi.nlm.nih.gov. Retrieved 1 December 2022.
- Sell, Charles S. (2006). "Terpenoids". Kirk-Othmer Encyclopedia of Chemical Technology. doi:10.1002/0471238961.2005181602120504.a01.pub2. ISBN 0471238961.
- Yus, Miguel; Ramón, Diego (2007). "Chiral Ligands with an Isoborneol-10-sulfonamide Structure: A Ten-Year Odyssey". Synlett. 2007 (15): 2309–2320. doi:10.1055/s-2007-985602.
- Chen, Y. K.; Jeon, S.-J.; Walsh, P. J.; Nugent, W. A. (2005). "(2S)-(−)-3-exo-(Morpholino)isoborneol". Organic Syntheses. 82: 87. doi:10.15227/orgsyn.082.0087.
- White, J. D.; Wardrop, D. J.; Sundermann, K. F. (2002). "(2S)-(−)-3-exo-(Dimethylamino)isoborneol". Organic Syntheses. 79: 130. doi:10.15227/orgsyn.079.0130.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.
-Camphor_Reduction_V.1.svg.png.webp)