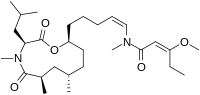
Kanamienamide
Kanamienamides is a complex enol ether containing enamide that is currently undergoing research in regards to its inhibitory activity towards cancer cells. The synthesis of kanamienamide consists of several chemical techniques, including CBS asymmetric reduction, Stork-Zhao-Wittig olefination, Cu-mediated amide coupling with vinyl iodide, Evans asymmetric alkylation, and ring-closing metathesis.[1] Kanamienamide is a natural product found in Moorea bouillonii which is a cyanobacterium.[2]
 | |
| Names | |
|---|---|
| Preferred IUPAC name
(2E)-3-Methoxy-N-{(1Z)-5-[(3S,6R,8S,11S)-4,6,8-trimethyl-3-(2-methylpropyl)-2,5-dioxo-1-oxa-4-azacycloundecan-11-yl]pent-1-en-1-yl}pent-2-enamide | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
| |
| |
| Properties | |
| C28H48N2O5 | |
| Molar mass | 492.701 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
References
- Prabhakar Reddy, D.; Zhang, Ning; Yu, Zhimei; Wang, Zhen; He, Yun (2017-10-02). "Total Synthesis of Kanamienamide". The Journal of Organic Chemistry. 82 (20): 11262–11268. doi:10.1021/acs.joc.7b01984. ISSN 0022-3263. PMID 28944669.
- Sumimoto, Shimpei; Iwasaki, Arihiro; Ohno, Osamu; Sueyoshi, Kosuke; Teruya, Toshiaki; Suenaga, Kiyotake (13 September 2016). "Kanamienamide, an Enamide with an Enol Ether from the Marine Cyanobacterium". Organic Letters. 18 (19): 4884–4887. doi:10.1021/acs.orglett.6b02364. PMID 27623268.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.