Kinemage
A kinemage (short for kinetic image) is an interactive graphic scientific illustration. It often is used to visualize molecules, especially proteins although it can also represent other types of 3-dimensional data (such as geometric figures, social networks,[1] or tetrahedra of RNA base composition). The kinemage system is designed to optimize ease of use, interactive performance, and the perception and communication of detailed 3D information. The kinemage information is stored in a text file, human- and machine-readable, that describes the hierarchy of display objects and their properties, and includes optional explanatory text. The kinemage format is a defined chemical MIME type of 'chemical/x-kinemage' with the file extension '.kin'.
Early history
Kinemages were first developed by David Richardson at Duke University School of Medicine, for the Protein Society's journal Protein Science that premiered in January 1992.[2] For its first 5 years (1992–1996), each issue of Protein Science included a supplement on floppy disk of interactive, kinemage 3D computer graphics to illustrate many of the articles, plus the Mage software (cross-platform, free, open-source) to display them;[3] kinemage supplementary material is still available on the journal web site. Mage and RasMol[4] were the first widely used macromolecular graphics programs to support interactive display on personal computers. Kinemages are used for teaching,[5][6] and for textbook supplements,[7][8] individual exploration, and analysis of macromolecular structures.
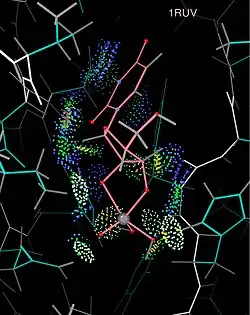
Research uses
More recently, with the availability of a much wider variety of other molecular graphics tools, presentation use of kinemages has been overtaken by a wide variety of research uses, concomitant with new display features and with the development of software that produces kinemage-format output from other types of molecular calculations. All-atom contact analysis[9] adds and optimizes explicit hydrogen atoms,[10] and then uses patches of dot surface to display the hydrogen bond, van der Waals, and steric clash interactions between atoms. The results can be used visually (in kinemages) and quantitatively to analyze the detailed interactions between molecular surfaces,[11][12] most extensively for the purpose of validating and improving the molecular models from experimental x-ray crystallography data.[13][14][15][16] Both Mage and KiNG (see below) have been enhanced for kinemage display of data in higher than 3 dimensions (moving between views in various 3-D projections, coloring and selecting candidate clusters of datapoints, and switching to a parallel coordinates representation), used for instance for defining clusters of favorable RNA backbone conformations in the 7-dimensional space of backbone dihedral angles between one ribose and the next.[17]
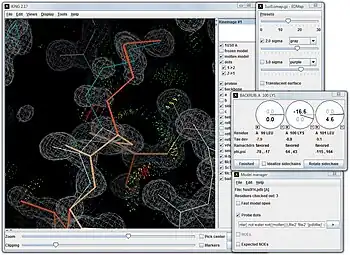
Online web use
KiNG is an open-source kinemage viewer, written in the programming language Java by Ian Davis and Vincent Chen,[18] that can work interactively either standalone on a user machine with no network connection, or as a web service in a web page. The interactive nature of kinemages is their primary purpose and attribute. To appreciate their nature, the demonstration KiNG in browser has two examples that can be moved around in 3D, plus instructions for how to embed a kinemage on a web page.[19] The figure below shows KiNG being used to remodel a lysine sidechain in a high-resolution crystal structure. KiNG is one of the viewers provided on each structure page at the Protein Data Bank site,[20] and displays validation results in 3D on the MolProbity site.[21][22][23] Kinemages can also be shown in immersive virtual reality systems, with the open-source KinImmerse software.[24] All of the kinemage display and all-atom contact software is available free and open-source on the kinemage web site.
References
- Freeman, L. C.; hierarchy; et al. (1998). "Exploring social structure using dynamic three-dimensional color images" (PDF). Social Networks. 20 (2): 109–118. doi:10.1016/S0378-8733(97)00016-6.
- Richardson, D. C.; J.S. Richardson (January 1992). "The kinemage: a tool for scientific communication". Protein Science. 1 (1): 3–9. doi:10.1002/pro.5560010102. PMC 2142077. PMID 1304880.
- Neurath, H. (1992). "Editorial. The Kinemage: A Tool for Scientific Illustration". Protein Science. 5 (11): 2147. doi:10.1002/pro.5560051101. PMC 2143300.
- Sayle, R. (1992). Proceedings of the 10th Eurographics UK 1992 Conference. Abingdon Press, York.
- Richardson, D. C.; J.S. Richardson (1994). "Kinemages - Simple Macromolecular Graphics for Interactive Teaching and Publication". Trends in Biochemical Sciences. 19 (3): 135–138. doi:10.1016/0968-0004(94)90207-0. PMID 8203021.
- Richardson, D. C.; J.S. Richardson (2002). "Teaching Molecular 3-D Literacy". Biochemistry and Molecular Biology Education. 30: 21–26. doi:10.1002/bmb.2002.494030010005.
- Voet, D.; J. G. Voet; C. W. Pratt (1999). Fundamentals of Biochemistry. John Wiley & Sons, New York.
- Branden, C.-I.; J. Tooze (1999). Introduction to Protein Structure (2 ed.). Garland Publishing, Inc., New York.
- Word, J. M.; et al. (1999). "Visualizing and Quantifying Molecular Goodness-of-Fit: Small-probe Contact Dots with Explicit Hydrogen Atoms". Journal of Molecular Biology. 285 (4): 1711–1733. CiteSeerX 10.1.1.119.6173. doi:10.1006/jmbi.1998.2400. PMID 9917407.
- Word, J. M.; et al. (1999). "Asparagine and Glutamine: Using Hydrogen Atom Contacts in the Choice of Side-chain Amide Orientation". Journal of Molecular Biology. 285 (4): 1735–1747. CiteSeerX 10.1.1.323.6971. doi:10.1006/jmbi.1998.2401. PMID 9917408.
- Word, J. M.; et al. (2000). "Exploring steric constraints on protein mutations using MAGE/PROBE". Protein Science. 9 (11): 2251–2259. doi:10.1110/ps.9.11.2251. PMC 2144501. PMID 11152136.
- Richardson, J. S.; Richardson, D.C. (2002). "Natural β-sheet proteins use negative design to avoid edge-to-edge aggregation". Proc. Natl. Acad. Sci. USA. 99 (5): 2754–2759. Bibcode:2002PNAS...99.2754R. doi:10.1073/pnas.052706099. PMC 122420. PMID 11880627.
- Richardson, D. C.; Richardson, J. S. (2001). "MAGE, PROBE, and Kinemages". International Tables for Crystallography. F, chapter 25.2.8: 727–730.
- Richardson, Jane S.; et al. (2003). "New Tools and Data for Improving Structures, Using All-Atom Contacts". Macromolecular Crystallography, Part D. Methods in Enzymology. Vol. 374. pp. 385–412. doi:10.1016/S0076-6879(03)74018-X. ISBN 978-0-12-182777-9. PMID 14696383.
- Higman, V.A..; et al. (2004). "Asparagine and glutamine side-chain conformations in solution and crystal: a comparison for hen egg-white lysozyme using residual dipolar couplings". Journal of Biomolecular NMR. 30 (3): 327–346. doi:10.1007/s10858-004-3218-y. PMID 15754058. S2CID 26047711.
- Arendall III, W. B.; et al. (2005). "A test of enhancing model accuracy in high-throughput crystallography". Journal of Structural and Functional Genomics. 6 (1): 1–11. doi:10.1007/s10969-005-3138-4. PMID 15965733. S2CID 2790812.
- Richardson, J. S.; et al. (2008). "RNA backbone: Consensus all-angle conformers and modular string nomenclature (an RNA Ontology Consortium contribution)". RNA. 14 (3): 465–481. doi:10.1261/rna.657708. PMC 2248255. PMID 18192612.
- Chen, V.B.; et al. (2009). "KING (Kinemage, Next Generation): A versatile interactive molecular and scientific visualization program". Protein Science. 18 (11): 2403–2409. doi:10.1002/pro.250. PMC 2788294. PMID 19768809.
- KiNG in browser
- "Protein Data Bank". Archived from the original on 2008-08-28. Retrieved 2016-12-07.
- MolProbity
- Davis, I. W.; et al. (2007). "MolProbity: all-atom contacts and structure validation for proteins and nucleic acids". Nucleic Acids Research. 35 (Web server issue): W375–W383. doi:10.1093/nar/gkm216. PMC 1933162. PMID 17452350.
- Chen, V. B.; et al. (2010). "MolProbity: all-atom structure validation for macromolecular crystallography". Acta Crystallographica. D 66 (Pt 1): 12–21. doi:10.1107/S0907444909042073. PMC 2803126. PMID 20057044.
- Block, J. N.; et al. (2009). "KinImmerse: Macromolecular VR for NMR ensembles". Source Code for Biology and Medicine. 4: 3. doi:10.1186/1751-0473-4-3. PMC 2650690. PMID 19222844.
External links
- Official website, Duke University original, with examples and software
- kinemage example in a browser
- RCSB Protein Data Bank
- MolProbity: structure validation, with KiNG on-line kinemages