Leprosy
Leprosy, also known as Hansen's disease (HD), is a long-term infection by the bacteria Mycobacterium leprae or Mycobacterium lepromatosis.[4][7] Infection can lead to damage of the nerves, respiratory tract, skin, and eyes.[4] This nerve damage may result in a lack of ability to feel pain, which can lead to the loss of parts of a person's extremities from repeated injuries or infection through unnoticed wounds.[3] An infected person may also experience muscle weakness and poor eyesight.[3] Leprosy symptoms may begin within one year, but, for some people, symptoms may take 20 years or more to occur.[4]
| Leprosy | |
|---|---|
| Other names | Hansen's disease (HD)[1] |
 | |
| Rash on the chest and abdomen caused by leprosy | |
| Pronunciation |
|
| Specialty | Infectious diseases |
| Symptoms | Decreased ability to feel pain[3] |
| Causes | Mycobacterium leprae or Mycobacterium lepromatosis[4][5] |
| Risk factors | Close contact with a case of leprosy, living in poverty[3][6] |
| Treatment | Multidrug therapy[4] |
| Medication | Rifampicin, dapsone, clofazimine[3] |
| Frequency | 209,000 (2018)[4] |
| Named after | Gerhard Armauer Hansen |
Leprosy is spread between people, although extensive contact is necessary.[3][8] Leprosy has a low pathogenicity, and 95% of people who contract M. leprae do not develop the disease.[9] Spread is thought to occur through a cough or contact with fluid from the nose of a person infected by leprosy.[8][9] Genetic factors and immune function play a role in how easily a person catches the disease.[9][10] Leprosy does not spread during pregnancy to the unborn child or through sexual contact.[8] Leprosy occurs more commonly among people living in poverty.[3] There are two main types of the disease – paucibacillary and multibacillary, which differ in the number of bacteria present.[3] A person with paucibacillary disease has five or fewer poorly pigmented, numb skin patches, while a person with multibacillary disease has more than five skin patches.[3] The diagnosis is confirmed by finding acid-fast bacilli in a biopsy of the skin.[3]
Leprosy is curable with multidrug therapy.[4] Treatment of paucibacillary leprosy is with the medications dapsone, rifampicin, and clofazimine for six months.[9] Treatment for multibacillary leprosy uses the same medications for 12 months.[9] A number of other antibiotics may also be used.[3] These treatments are provided free of charge by the World Health Organization.[4]
Leprosy is not highly contagious.[11] People with leprosy can live with their families and go to school and work.[12] In the 1980s, there were 5.2 million cases globally, but this decreased to fewer than 200 thousand by 2020.[4][13][14] Most new cases occur in 14 countries, with India accounting for more than half.[3][4] In the 20 years from 1994 to 2014, 16 million people worldwide were cured of leprosy.[4] About 200 cases per year are reported in the United States.[15] Central Florida accounted for 81% of cases in Florida and nearly 1 out of 5 leprosy cases nationwide.[16] Separating people affected by leprosy by placing them in leper colonies still occurs in some areas of India,[17] China,[18] the African continent,[11] and Thailand.[19]
Leprosy has affected humanity for thousands of years.[3] The disease takes its name from the Greek word λέπρᾱ (léprā), from λεπῐ́ς (lepís; 'scale'), while the term "Hansen's disease" is named after the Norwegian physician Gerhard Armauer Hansen.[3] Leprosy has historically been associated with social stigma, which continues to be a barrier to self-reporting and early treatment.[4] Some consider the word leper offensive, preferring the phrase "person affected with leprosy".[20] Leprosy is classified as a neglected tropical disease.[21] World Leprosy Day was started in 1954 to draw awareness to those affected by leprosy.[22][4]
Signs and symptoms
Common symptoms present in the different types of leprosy include a runny nose; dry scalp; eye problems; skin lesions; muscle weakness; reddish skin; smooth, shiny, diffuse thickening of facial skin, ear, and hand; loss of sensation in fingers and toes; thickening of peripheral nerves; a flat nose from destruction of nasal cartilages; and changes in phonation and other aspects of speech production.[23] In addition, atrophy of the testes and impotence may occur.[24]
Leprosy can affect people in different ways.[9] The average incubation period is five years.[4] People may begin to notice symptoms within the first year or up to 20 years after infection.[4] The first noticeable sign of leprosy is often the development of pale or pink coloured patches of skin that may be insensitive to temperature or pain.[25] Patches of discolored skin are sometimes accompanied or preceded by nerve problems including numbness or tenderness in the hands or feet.[25][26] Secondary infections (additional bacterial or viral infections) can result in tissue loss, causing fingers and toes to become shortened and deformed, as cartilage is absorbed into the body.[27][28] A person's immune response differs depending on the form of leprosy.[29]
Approximately 30% of people affected with leprosy experience nerve damage.[30] The nerve damage sustained is reversible when treated early, but becomes permanent when appropriate treatment is delayed by several months. Damage to nerves may cause loss of muscle function, leading to paralysis. It may also lead to sensation abnormalities or numbness, which may lead to additional infections, ulcerations, and joint deformities.[30]
.jpg.webp) Paucibacillary leprosy (PB): Pale skin patch with loss of sensation
Paucibacillary leprosy (PB): Pale skin patch with loss of sensation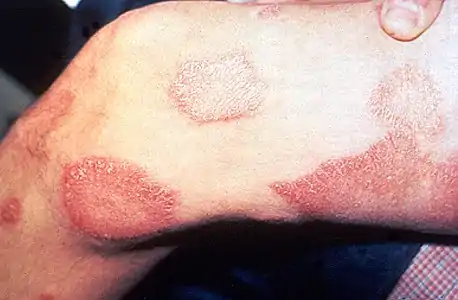 Skin lesions on the thigh of a person with leprosy
Skin lesions on the thigh of a person with leprosy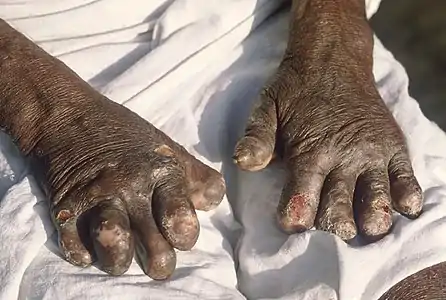 Hands deformed by leprosy
Hands deformed by leprosy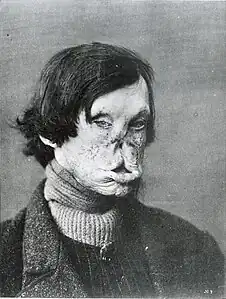 Face deformed by leprosy
Face deformed by leprosy
Cause
M. leprae and M. lepromatosis

M. leprae and M. lepromatosis are the mycobacteria that cause leprosy.[30] M. lepromatosis is a relatively newly identified mycobacterium isolated from a fatal case of diffuse lepromatous leprosy in 2008.[5][31] M. lepromatosis is indistinguishable clinically from M. leprae.[32]
M. leprae is an intracellular, acid-fast bacterium that is aerobic and rod-shaped.[33] M. leprae is surrounded by the waxy cell envelope coating characteristic of the genus Mycobacterium.[33]
Genetically, M. leprae and M. lepromatosis lack the genes that are necessary for independent growth.[34] M. leprae and M. lepromatosis are obligate intracellular pathogens, and cannot be grown (cultured) in the laboratory.[34] The inability to culture M. leprae and M. lepromatosis has resulted in a difficulty definitively identifying the bacterial organism under a strict interpretation of Koch's postulates.[5][34]
While the causative organisms have to date been impossible to culture in vitro, it has been possible to grow them in animals such as mice and armadillos.[35][36]
Naturally occurring infection has been reported in nonhuman primates (including the African chimpanzee, the sooty mangabey, and the cynomolgus macaque), armadillos,[37] and red squirrels.[38] Multilocus sequence typing of the armadillo M. leprae strains suggests that they were of human origin for at most a few hundred years.[39] Thus, it is suspected that armadillos first acquired the organism incidentally from early American explorers.[40] This incidental transmission was sustained in the armadillo population, and it may be transmitted back to humans, making leprosy a zoonotic disease (spread between humans and animals).[40]
Red squirrels (Sciurus vulgaris), a threatened species in Great Britain, were found to carry leprosy in November 2016.[41] It has been suggested that the trade in red squirrel fur, highly prized in the medieval period and intensively traded, may have been responsible for the leprosy epidemic in medieval Europe.[42] A pre-Norman era skull excavated in Hoxne, Suffolk, in 2017 was found to carry DNA from a strain of Mycobacterium leprae, which closely matched the strain carried by modern red squirrels on Brownsea Island, UK.[42][43]
Risk factors
The greatest risk factor for developing leprosy is contact with another person infected by leprosy.[4] People who are exposed to a person who has leprosy are 5–8 times more likely to develop leprosy than members of the general population.[6] Leprosy also occurs more commonly among those living in poverty.[3] Not all people who are infected with M. leprae develop symptoms.[44][45]
Conditions that reduce immune function, such as malnutrition, other illnesses, or genetic mutations, may increase the risk of developing leprosy.[6] Infection with HIV does not appear to increase the risk of developing leprosy.[46] Certain genetic factors in the person exposed have been associated with developing lepromatous or tuberculoid leprosy.[47]
Transmission
Transmission of leprosy occurs during close contact with those who are infected.[4] Transmission of leprosy is through the upper respiratory tract.[9][48] Older research suggested the skin as the main route of transmission, but research has increasingly favored the respiratory route.[49] Transmission occurs through inhalation of bacilli present in upper airway secretion.[50]
Leprosy is not sexually transmitted and is not spread through pregnancy to the unborn child.[4][8] The majority (95%) of people who are exposed to M. leprae do not develop leprosy; casual contact such as shaking hands and sitting next to someone with leprosy does not lead to transmission.[4][51] People are considered non-infectious 72 hours after starting appropriate multi-drug therapy.[52]
Two exit routes of M. leprae from the human body often described are the skin and the nasal mucosa, although their relative importance is not clear. Lepromatous cases show large numbers of organisms deep in the dermis, but whether they reach the skin surface in sufficient numbers is doubtful.[53]
Leprosy may also be transmitted to humans by armadillos, although the mechanism is not fully understood.[8][54][55]
Genetics
| Name | Locus | OMIM | Gene |
|---|---|---|---|
| LPRS1 | 10p13 | 609888 | |
| LPRS2 | 6q25 | 607572 | PARK2, PACRG |
| LPRS3 | 4q32 | 246300 | TLR2 |
| LPRS4 | 6p21.3 | 610988 | LTA |
| LPRS5 | 4p14 | 613223 | TLR1 |
| LPRS6 | 13q14.11 | 613407 |
Not all people who are infected or exposed to M. leprae develop leprosy, and genetic factors are suspected to play a role in susceptibility to an infection.[56] Cases of leprosy often cluster in families and several genetic variants have been identified.[56] In many people who are exposed, the immune system is able to eliminate the leprosy bacteria during the early infection stage before severe symptoms develop.[57] A genetic defect in cell-mediated immunity may cause a person to be susceptible to develop leprosy symptoms after exposure to the bacteria.[58] The region of DNA responsible for this variability is also involved in Parkinson's disease, giving rise to current speculation that the two disorders may be linked at the biochemical level.[58]
Mechanism
Most leprosy complications are the result of nerve damage. The nerve damage occurs from direct invasion by the M. leprae bacteria and a person's immune response resulting in inflammation.[30] The molecular mechanism underlying how M. leprae produces the symptoms of leprosy is not clear,[14] but M. leprae has been shown to bind to Schwann cells, which may lead to nerve injury including demyelination and a loss of nerve function (specifically a loss of axonal conductance).[59] Numerous molecular mechanisms have been associated with this nerve damage including the presence of a laminin-binding protein and the glycoconjugate (PGL-1) on the surface of M. leprae that can bind to laminin on peripheral nerves.[59]
As part of the human immune response, white blood cell-derived macrophages may engulf M. leprae by phagocytosis.[59]
In the initial stages, small sensory and autonomic nerve fibers in the skin of a person with leprosy are damaged.[30] This damage usually results in hair loss to the area, a loss of the ability to sweat, and numbness (decreased ability to detect sensations such as temperature and touch). Further peripheral nerve damage may result in skin dryness, more numbness, and muscle weaknesses or paralysis in the area affected.[30] The skin can crack and if the skin injuries are not carefully cared for, there is a risk for a secondary infection that can lead to more severe damage.[30]
Diagnosis
In countries where people are frequently infected, a person is considered to have leprosy if they have one of the following two signs:
Skin lesions can be single or many, and usually hypopigmented, although occasionally reddish or copper-colored.[4] The lesions may be flat (macules), raised (papules), or solid elevated areas (nodular).[4] Experiencing sensory loss at the skin lesion is a feature that can help determine if the lesion is caused by leprosy or by another disorder such as tinea versicolor.[4][60] Thickened nerves are associated with leprosy and can be accompanied by loss of sensation or muscle weakness, but muscle weakness without the characteristic skin lesion and sensory loss is not considered a reliable sign of leprosy.[4]
In some cases, acid-fast leprosy bacilli in skin smears are considered diagnostic; however, the diagnosis is typically made without laboratory tests, based on symptoms.[4] If a person has a new leprosy diagnosis and already has a visible disability caused by leprosy, the diagnosis is considered late.[30]
In countries or areas where leprosy is uncommon, such as the United States, diagnosis of leprosy is often delayed because healthcare providers are unaware of leprosy and its symptoms.[61] Early diagnosis and treatment prevent nerve involvement, the hallmark of leprosy, and the disability it causes.[4][61]
There is no recommended test to diagnose latent leprosy in people without symptoms.[9] Few people with latent leprosy test positive for anti PGL-1.[44] The presence of M. leprae bacterial DNA can be identified using a polymerase chain reaction (PCR)-based technique.[62] This molecular test alone is not sufficient to diagnose a person, but this approach may be used to identify someone who is at high risk of developing or transmitting leprosy such as those with few lesions or an atypical clinical presentation.[62][63]
Classification
Several different approaches for classifying leprosy exist. There are similarities between the classification approaches.
- The World Health Organization system distinguishes "paucibacillary" and "multibacillary" based upon the proliferation of bacteria.[64] ("pauci-" refers to a small quantity.)
- The Ridley-Jopling scale provides five gradations.[65][66][67]
- The ICD-10, though developed by the WHO, uses Ridley-Jopling and not the WHO system. It also adds an indeterminate ("I") entry.[53]
- In MeSH, three groupings are used.
| WHO | Ridley-Jopling | ICD-10 | MeSH | Description | Lepromin test |
|---|---|---|---|---|---|
| Paucibacillary | tuberculoid ("TT"), borderline tuberculoid ("BT") |
A30.1, A30.2 | Tuberculoid | It is characterized by one or more hypopigmented skin macules and patches where skin sensations are lost because of damaged peripheral nerves that have been attacked by the human host's immune cells. TT is characterized by the formation of epithelioid cell granulomas with a large number of epithelioid cells. In this form of leprosy Mycobacterium leprae are either absent from the lesion or occur in very small numbers. This type of leprosy is most benign.[59][68] | Positive |
| Multibacillary | midborderline or borderline ("BB") |
A30.3 | Borderline | Borderline leprosy is of intermediate severity and is the most common form. Skin lesions resemble tuberculoid leprosy, but are more numerous and irregular; large patches may affect a whole limb, and peripheral nerve involvement with weakness and loss of sensation is common. This type is unstable and may become more like lepromatous leprosy or may undergo a reversal reaction, becoming more like the tuberculoid form. | Negative |
| Multibacillary | borderline lepromatous ("BL"), and lepromatous ("LL") |
A30.4, A30.5 | Lepromatous | It is associated with symmetric skin lesions, nodules, plaques, thickened dermis, and frequent involvement of the nasal mucosa resulting in nasal congestion and nose bleeds, but, typically, detectable nerve damage is late. Loss of eyebrows and lashes can be seen in advanced disease.[69] LL is characterized by the absence of epithelioid cells in the lesions. In this form of leprosy, Mycobacteria leprae are found in lesions in large numbers. This is the most unfavorable clinical variant of leprosy, which occurs with a generalized lesion of the skin, mucous membranes, eyes, peripheral nerves, lymph nodes, and internal organs.[59][68] | Negative |
Leprosy may also occur with only neural involvement, without skin lesions.[4][70][71][72][73][74]
Complications
Leprosy may cause the victim to lose limbs and digits but not directly. M. leprae attacks nerve endings and destroys the body's ability to feel pain and injury. Without feeling pain, people with leprosy have an increased risk of injuring themselves. Injuries become infected and result in tissue loss. Fingers, toes, and limbs become shortened and deformed as the tissue is absorbed into the body.[75]
Prevention
Early detection of the disease is important, since physical and neurological damage may be irreversible even if cured.[4] Medications can decrease the risk of those living with people who have leprosy from acquiring the disease and likely those with whom people with leprosy come into contact outside the home.[14] The WHO recommends that preventive medicine be given to people who are in close contact with someone who has leprosy.[9] The suggested preventive treatment is a single dose of rifampicin (SDR) in adults and children over 2 years old who do not already have leprosy or tuberculosis.[9] Preventive treatment is associated with a 57% reduction in infections within 2 years and a 30% reduction in infections within 6 years.[9]
The Bacillus Calmette–Guérin (BCG) vaccine offers a variable amount of protection against leprosy in addition to its closely related target of tuberculosis.[76] It appears to be 26% to 41% effective (based on controlled trials) and about 60% effective based on observational studies with two doses possibly working better than one.[77][78] The WHO concluded in 2018 that the BCG vaccine at birth reduces leprosy risk and is recommended in countries with high incidence of TB and people who have leprosy.[79] People living in the same home as a person with leprosy are suggested to take a BCG booster which may improve their immunity by 56%.[80][81] Development of a more effective vaccine is ongoing.[14][82][83][84]
A novel vaccine called LepVax entered clinical trials in 2017 with the first encouraging results reported on 24 participants published in 2020.[85][86] If successful, this would be the first leprosy-specific vaccine available.
Treatment
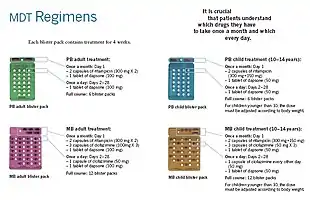
Anti-leprosy medication
A number of leprostatic agents are available for treatment. A three-drug regimen of rifampicin, dapsone and clofazimine is recommended for all people with leprosy, for six months for paucibacillary leprosy and 12 months for multibacillary leprosy.[9]
Multidrug therapy (MDT) remains highly effective, and people are no longer infectious after the first monthly dose.[4] It is safe and easy to use under field conditions because of its presentation in calendar blister packs.[4] Post-treatment relapse rates remain low.[4] Resistance has been reported in several countries, although the number of cases is small.[87] People with rifampicin-resistant leprosy may be treated with second line drugs such as fluoroquinolones, minocycline, or clarithromycin, but the treatment duration is 24 months because of their lower bactericidal activity.[88] Evidence on the potential benefits and harms of alternative regimens for drug-resistant leprosy is not available.[9]
Skin changes
For people with nerve damage, protective footwear may help prevent ulcers and secondary infection.[30] Canvas shoes may be better than PVC boots.[30] There may be no difference between double rocker shoes and below-knee plaster.[30]
Topical ketanserin seems to have a better effect on ulcer healing than clioquinol cream or zinc paste, but the evidence for this is weak.[30] Phenytoin applied to the skin improves skin changes to a greater degree when compared to saline dressings.[30]
Outcomes
Although leprosy has been curable since the mid-20th century, left untreated it can cause permanent physical impairments and damage to a person's nerves, skin, eyes, and limbs.[4] Despite leprosy not being very infectious and having a low pathogenicity, there is still significant stigma and prejudice associated with the disease.[89] Because of this stigma, leprosy can affect a person's participation in social activities and may also affect the lives of their family and friends.[89] People with leprosy are also at a higher risk for problems with their mental well-being.[89] The social stigma may contribute to problems obtaining employment, financial difficulties, and social isolation.[89] Efforts to reduce discrimination and reduce the stigma surrounding leprosy may help improve outcomes for people with leprosy.[90]
Epidemiology

In 2018, there were 208,619 new cases of leprosy recorded, a slight decrease from 2017.[94] In 2015, 94% of the new leprosy cases were confined to 14 countries.[95] India reported the greatest number of new cases (60% of reported cases), followed by Brazil (13%) and Indonesia (8%).[95] Although the number of cases worldwide continues to fall, there are parts of the world where leprosy is more common, including Brazil, South Asia (India, Nepal, Bhutan), some parts of Africa (Tanzania, Madagascar, Mozambique), and the western Pacific.[95] About 150 to 250 cases are diagnosed in the United States each year.[96]
In the 1960s, there were tens of millions of leprosy cases recorded when the bacteria started to develop resistance to dapsone, the most common treatment option at the time.[4][14] International (e.g., the WHO's "Global Strategy for Reducing Disease Burden Due to Leprosy") and national (e.g., the International Federation of Anti-Leprosy Associations) initiatives have reduced the total number and the number of new cases of the disease.[14][97]
Disease burden
The number of new leprosy cases is difficult to measure and monitor because of leprosy's long incubation period, delays in diagnosis after onset of the disease, and lack of medical care in affected areas.[98] The registered prevalence of the disease is used to determine disease burden.[99] Registered prevalence is a useful proxy indicator of the disease burden, as it reflects the number of active leprosy cases diagnosed with the disease and receiving treatment with MDT at a given point in time.[99] The prevalence rate is defined as the number of cases registered for MDT treatment among the population in which the cases have occurred, again at a given point in time.[99]
| Year | 2005 | 2006 | 2007 | 2008 | 2009 | 2010 | 2011 | 2012 | 2013 | 2014 | 2015 | 2016 | 2017 | 2018 | 2019 | 2020 | 2021 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| No. of new cases[100] | 296,479 | 258,980 | 252,541 | 249,018 | 244,797 | 228,488 | 224,344 | 232,847 | 215,636 | 213,861 | 211,945 | 217,927 | 210,973 | 208,613 | 202,166 | 127,506 | 140,546 |
History

Historical distribution
Using comparative genomics, in 2005, geneticists traced the origins and worldwide distribution of leprosy from East Africa or the Near East along human migration routes. They found four strains of M. leprae with specific regional locations:[101] Monot et al. (2005) determined that leprosy originated in East Africa or the Near East and traveled with humans along their migration routes, including those of trade in goods and slaves. The four strains of M. leprae are based in specific geographic regions where each predominantly occurs:[101]
- strain 1 in Asia, the Pacific region, and East Africa;
- strain 2 in Ethiopia, Malawi, Nepal, north India, and New Caledonia;
- strain 3 in Europe, North Africa, and the Americas;
- strain 4 in West Africa and the Caribbean.
This confirms the spread of the disease along the migration, colonisation, and slave trade routes taken from East Africa to India, West Africa to the New World, and from Africa into Europe and vice versa.[102]
Skeletal remains discovered in 2009 represent the oldest documented evidence for leprosy, dating to the 2nd millennium BC.[103][104] Located at Balathal, Rajasthan, in northwest India, the discoverers suggest that, if the disease did migrate from Africa to India during the 3rd millennium BC "at a time when there was substantial interaction among the Indus Civilization, Mesopotamia, and Egypt, there needs to be additional skeletal and molecular evidence of leprosy in India and Africa to confirm the African origin of the disease".[105] A proven human case was verified by DNA taken from the shrouded remains of a man discovered by researchers from the Hebrew University of Jerusalem in a tomb next to the Old City of Jerusalem, Israel, dated by radiocarbon methods to the first half of the 1st century.[106]
The oldest strains of leprosy known from Europe are from Great Chesterford in southeast England and dating back to AD 415–545. These findings suggest a different path for the spread of leprosy, meaning it may have originated in Western Eurasia. This study also indicates that there were more strains in Europe at the time than previously determined.[107]
Discovery and scientific progress
Literary attestation of leprosy is unclear because of the ambiguity of many early sources, including the Indian Atharvaveda and Kausika Sutra, the Egyptian Ebers papyrus, and the Hebrew Bible's various sections regarding signs of impurity (tzaraath).[108] Clearly leprotic symptoms are attested in the Indian doctor Sushruta's Compendium, originally dating to c. 600 BC but only surviving in emended texts no earlier than the 5th century. They were separately described by Hippocrates in 460 BC. However, Hansen's disease probably did not exist in Greece or the Middle East before the Common Era.[109][110][111] In 1846, Francis Adams produced The Seven Books of Paulus Aegineta which included a commentary on all medical and surgical knowledge and descriptions and remedies to do with leprosy from the Romans, Greeks, and Arabs.[112][113]
Leprosy did not exist in the Americas before colonization by modern Europeans[114] nor did it exist in Polynesia until the middle of the 19th century.[115]

The causative agent of leprosy, M. leprae, was discovered by Gerhard Armauer Hansen in Norway in 1873, making it the first bacterium to be identified as causing disease in humans.[116]
Treatment
The first effective treatment (promin) became available in the 1940s.[117] In the 1950s, dapsone was introduced. The search for further effective antileprosy drugs led to the use of clofazimine and rifampicin in the 1960s and 1970s.[118] Later, Indian scientist Shantaram Yawalkar and his colleagues formulated a combined therapy using rifampicin and dapsone, intended to mitigate bacterial resistance.[119] Multi-drug therapy (MDT) combining all three drugs was first recommended by the WHO in 1981. These three antileprosy drugs are still used in the standard MDT regimens.[120]
Leprosy was once believed to be highly contagious and was treated with mercury, as was syphilis, which was first described in 1530. Many early cases thought to be leprosy could actually have been syphilis.[121]
Resistance has developed to initial treatment. Until the introduction of MDT in the early 1980s, leprosy could not be diagnosed and treated successfully within the community.[122]
Japan still has sanatoriums (although Japan's sanatoriums no longer have active leprosy cases, nor are survivors held in them by law).[123]
The importance of the nasal mucosa in the transmission of M. leprae was recognized as early as 1898 by Schäffer, in particular, that of the ulcerated mucosa.[124] The mechanism of plantar ulceration in leprosy and its treatment was first described by Ernest W. Price.[125]
Etymology
The word "leprosy" comes from the Greek word "λέπος (lépos) – skin" and "λεπερός (leperós) – scaly man".
Society and culture

India
In 1898, the colonial government in British India enacted the Leprosy Act of 1898, which mandated the compulsory segregation of lepers by authorities in newly established leper asylums, where they were segregated by sex to prevent sexual activity. The act, which proved difficult to enforce, was repealed in 1983 by the Indian government after multidrug therapy had become widely available in India. In 1983, the National Leprosy Elimination Programme, previously the National Leprosy Control Programme, changed its methods from surveillance to the treatment of people with leprosy. India still accounts for over half of the global disease burden. According to WHO, new cases in India during 2019 diminished to 114,451 patients (57% of the world's total new cases).[126][127] Until 2019, Indians could justify a petition for divorce with their spouse's diagnosis of leprosy.[128]
Treatment cost
Between 1995 and 1999, the WHO, with the aid of the Nippon Foundation, supplied all endemic countries with free multidrug therapy in blister packs, channeled through ministries of health.[4] This free provision was extended in 2000 and again in 2005, 2010 and 2015 with donations by the multidrug therapy manufacturer Novartis through the WHO. In the latest agreement signed between the company and the WHO in October 2015, the provision of free multidrug therapy by the WHO to all endemic countries will run until the end of 2025.[129][4] At the national level, nongovernment organizations affiliated with the national program will continue to be provided with an appropriate free supply of multidrug therapy by the WHO.[130]
Historical texts
Written accounts of leprosy date back thousands of years. Various skin diseases translated as leprosy appear in the ancient Indian text, the Atharava Veda, by 600 BC.[131] Another Indian text, the Manusmriti (200 BC), prohibited contact with those infected with the disease and made marriage to a person infected with leprosy punishable.[132]
The Hebraic root tsara or tsaraath (צָרַע, – tsaw-rah' – to be struck with leprosy, to be leprous) and the Greek (λεπρός–lepros), are of broader classification than the more narrow use of the term related to Hansen's Disease.[133] Any progressive skin disease (a whitening or splotchy bleaching of the skin, raised manifestations of scales, scabs, infections, rashes, etc....), as well as generalized molds and surface discoloration of any clothing, leather, or discoloration on walls or surfaces throughout homes all, came under the "law of leprosy" (Leviticus 14:54–57).[134] Ancient sources such as the Talmud (Sifra 63) make clear that tzaraath refers to various types of lesions or stains associated with ritual impurity and occurring on cloth, leather, or houses, as well as skin. Traditional Judaism and Jewish rabbinical authorities, both historical and modern, emphasize that the tsaraath of Leviticus is a spiritual ailment with no direct relationship to Hansen's disease or physical contagions. The relation of tsaraath to "leprosy" comes from translations of Hebrew Biblical texts into Greek and ensuing misconceptions.[135]
All three Synoptic Gospels of the New Testament describe instances of Jesus healing people with leprosy (Matthew 8:1–4, Mark 1:40–45, and Luke 5:12–16). The Bible's description of leprosy is congruous (if lacking detail) with the symptoms of modern leprosy, but the relationship between this disease, tzaraath, and Hansen's disease has been disputed.[136] The biblical perception that people with leprosy were unclean can be found in a passage from Leviticus 13: 44–46. While this text defines the leper as impure, it did not explicitly make a moral judgement on those with leprosy.[137] Some Early Christians believed that those affected by leprosy were being punished by God for sinful behavior. Moral associations have persisted throughout history. Pope Gregory the Great (540–604) and Isidor of Seville (560–636) considered people with the disease to be heretics.[138]
Middle Ages

The social perception of leprosy in the general population was in general mixed. On one hand, people feared getting infected with the disease and thought of people suspected of leprosy to be unclean, untrustworthy, and occasionally morally corrupt.[138] On the other hand, Jesus' interaction with lepers, the writing of church leaders and the Christian focus on charitable works led to viewing the lepers as "chosen by God"[139] or seeing the disease as a means of obtaining access to heaven.[140]
Early medieval understanding of leprosy was influenced by early Christian writers such as Gregory of Nazianzus and John Chrysostom, whose writings were later embraced by Byzantine and Latin writers.[141] Gregory, for example, did not only compose sermons urging Christians to assist victims of the disease, but also condemned pagans or Christians who justified rejecting lepers on the allegation that God had sent them the disease to punish them. As cases of leprosy increased during these years in the Eastern Roman Empire, becoming a major health issue, the ecclesiastic leaders of the time discussed how to assist those affected as well as change the attitude of society towards them. They also tried this by using the name "Holy disease" instead of the commonly used "Elephant's disease" (elephantiasis), implying that God did not create this disease to punish people but to purify them for heaven.[142] Although not always successful in persuading the public and a cure was never found by Greek medicians, they created an environment where victims could get palliative care and were never expressly banned from society, as sometimes happened in Western Europe. Theodore Balsamon, a 12th-century jurist in Constantinople, noted that lepers were allowed to enter the same churches, cities and assemblies that healthy people attended.[141]
As the disease became more prevalent in Western Europe in the fifth century, first efforts to set up permanent institutions to house and feed lepers. These efforts were, inclusively, the work of bishops in France at the end of the sixth century, such as in Chalon-sur-Saône.[141] The increase in hospitals or leprosaria (sing. leprosarium) that treated people with leprosy in the 12th and 13th century seems to indicate a rise in cases,[143][144][145] possibly in connection with the increase in urbanisation [146] as well as returning crusaders from the Middle East.[141] France alone had nearly 2,000 leprosaria during this period.[147] Additionally to the new leprosia, further steps were taken by secular and religious leaders to prevent further spread of the disease. The third Lateran Council of 1179 required lepers to have their own priests and churches[146] and a 1346 edict by King Edward expelled lepers from city limits. Segregation from mainstream society became common, and people with leprosy were often required to wear clothing that identified them as such or carry a bell announcing their presence.[147] As in the East, it was the Church who took care of the lepers due to the still persisting moral stigma and who ran the leprosaria.[138][148] Although the leprosaria in Western Europe removed the sick from society, they were never a place to quarantine them or from which they could not leave: lepers would go beg for alms for the upkeep of the leprosaria or meet with their families.[146][141]
19th century
Norway
Norway was the location of a progressive stance on leprosy tracking and treatment and played an influential role in European understanding of the disease. In 1832, Dr. JJ Hjort conducted the first leprosy survey, thus establishing a basis for epidemiological surveys. Subsequent surveys resulted in the establishment of a national leprosy registry to study the causes of leprosy and for tracking the rate of infection.
Early leprosy research throughout Europe was conducted by Norwegian scientists Daniel Cornelius Danielssen and Carl Wilhelm Boeck. Their work resulted in the establishment of the National Leprosy Research and Treatment Center. Danielssen and Boeck believed the cause of leprosy transmission was hereditary. This stance was influential in advocating for the isolation of those infected by sex to prevent reproduction.[149][150][151]
Leprosy and imperialism

Though leprosy rates were again on the decline in the Western world by the 1860s, authorities in the West frequently embraced isolation treatment due to a combination of reasons, including fears of the disease spreading from the Global South, efforts by Christian missionaries and a lack of understanding concerning bacteriology, medical diagnosis and how contagious the disease was.[127] The rapid expansion of Western imperialism during the Victorian era resulted in Westerners coming into increasing contact with regions where the disease was endemic, including British India. English surgeon Henry Vandyke Carter observed isolation treatment for leprosy patients first-hand while visiting Norway, applying these methods in British India with the financial and logistical assistance of Protestant missionaries. Colonialist and religious viewpoints of the disease continued to be a major factor in the treatment and public perception of the disease in the Global South until decolonization in the mid-twentieth century.[127]
United States
The National Leprosarium at Carville, Louisiana, known in 1955 as the Louisiana Leper Home, was the only leprosy hospital on the mainland United States. Leprosy patients from all over the United States were sent to Carville in order to be kept in isolation away from the public, as not much about leprosy transmission was known at the time and stigma against those with leprosy was high (see Leprosy stigma). The Carville leprosarium was known for its innovations in reconstructive surgery for those with leprosy. In 1941, 22 patients at Carville underwent trials for a new drug called promin. The results were described as miraculous, and soon after the success of promin came dapsone, a medicine even more effective in the fight against leprosy.[152]
United States
In 2022 cases of leprosy were reported to be increasing in central Florida.[153]
Stigma
Despite now effective treatment and education efforts, leprosy stigma continues to be problematic in developing countries where the disease is common. Leprosy is most common amongst impoverished populations where social stigma is likely to be compounded by poverty. Fears of ostracism, loss of employment, or expulsion from family and society may contribute to a delayed diagnosis and treatment.[154]
Folk beliefs, lack of education, and religious connotations of the disease continue to influence social perceptions of those affected in many parts of the world. In Brazil, for example, folklore holds that leprosy is a disease transmitted by dogs, or that it is associated with sexual promiscuity, or that it is a punishment for sins or moral transgressions (distinct from other diseases and misfortunes, which are in general thought of as being according to the will of God).[155] Socioeconomic factors also have a direct impact. Lower-class domestic workers who are often employed by those in a higher socioeconomic class may find their employment in jeopardy as physical manifestations of the disease become apparent. Skin discoloration and darker pigmentation resulting from the disease also have social repercussions.[156]
In extreme cases in northern India, leprosy is equated with an "untouchable" status that "often persists long after individuals with leprosy have been cured of the disease, creating lifelong prospects of divorce, eviction, loss of employment, and ostracism from family and social networks."[157]
 Leprosy in Tahiti, c. 1895
Leprosy in Tahiti, c. 1895 A 26-year-old woman with leprous lesions
A 26-year-old woman with leprous lesions A 13-year-old boy with severe leprosy
A 13-year-old boy with severe leprosy
Public policy
A goal of the World Health Organization is to "eliminate leprosy" and in 2016 the organization launched "Global Leprosy Strategy 2016–2020: Accelerating towards a leprosy-free world".[158] Elimination of leprosy is defined as "reducing the proportion of leprosy patients in the community to very low levels, specifically to below one case per 10,000 population".[159] Diagnosis and treatment with multidrug therapy are effective, and a 45% decline in disease burden has occurred since multidrug therapy has become more widely available.[160] The organization emphasizes the importance of fully integrating leprosy treatment into public health services, effective diagnosis and treatment, and access to information.[160] The approach includes supporting an increase in health care professionals who understand the disease, and a coordinated and renewed political commitment that includes coordination between countries and improvements in the methodology for collecting and analysing data.[158]
Interventions in the "Global Leprosy Strategy 2016–2020: Accelerating towards a leprosy-free world":[158]
- Early detection of cases focusing on children to reduce transmission and disabilities.
- Enhanced healthcare services and improved access for people who may be marginalized.
- For countries where leprosy is endemic, further interventions include an improved screening of close contacts, improved treatment regimens, and interventions to reduce stigma and discrimination against people who have leprosy.
Community-based interventions
In some instances in India, community-based rehabilitation is embraced by local governments and NGOs alike. Often, the identity cultivated by a community environment is preferable to reintegration, and models of self-management and collective agency independent of NGOs and government support have been desirable and successful.[161]
Notable cases
- Josephine Cafrine of Seychelles had leprosy from the age of 12 and kept a personal journal that documented her struggles and suffering.[162][163][164] It was published as an autobiography in 1923.[162][163][164][165]
- Saint Damien De Veuster, a Roman Catholic priest from Belgium, himself eventually contracting leprosy, ministered to lepers who had been placed under a government-sanctioned medical quarantine on the island of Molokaʻi in the Kingdom of Hawaiʻi.[166]
- Baldwin IV of Jerusalem was a Christian king of Latin Jerusalem who had leprosy.[167]
- Josefina Guerrero was a Filipino spy during World War II, who used the Japanese fear of her leprosy to listen to their battle plans and deliver the information to the American forces under Douglas MacArthur.[168]
- King Henry IV of England (reigned 1399 to 1413) possibly had leprosy.[169]
- Vietnamese poet Hàn Mặc Tử[170]
- Ōtani Yoshitsugu, a Japanese daimyō[171] (feudal lord).
Leprosy in the media
- English author Graham Greene's novel A Burnt-Out Case is set in a leper colony in Belgian Congo. The story is also predominantly about a disillusioned architect working with a doctor on devising new cure and amenities for mutilated victims of lepers; the title, too, refers to the condition of mutilation and disfigurement in the disease.[172]
- James Michener's novel Hawaii dramatizes the island of Molokai's leper settlement, including Father Damien.
- "Weird Al" Yankovic has a song titled "Party at the Leper Colony" from his album "Poodle Hat".
- Death metal band Death (metal band) has an album titled “Leprosy”.
- Forugh Farrokhzad made a 22-minute documentary about a leprosy colony in Iran in 1962 titled The House Is Black. The film humanizes the people affected and opens by saying that "there is no shortage of ugliness in the world, but by closing our eyes on ugliness, we will intensify it."
- Moloka'i is a novel by Alan Brennert about a leper colony in Hawaii. This novel follows the story of a seven-year-old girl taken from her family and put on Molokai's leper settlement.
- Jack London in 1909 published Koolau the Leper in his Tales of Hawai'i about Molokai and people consigned to it circa 1893.
- The lead character in The Chronicles of Thomas Covenant by Stephen R. Donaldson suffers from leprosy. His condition seems to be cured by the magic of the fantasy land he finds himself in, but he resists believing in its reality, for example, by continuing to perform a regular visual surveillance of extremities as a safety check. Donaldson gained experience with the disease as a young man in India, where his father worked in a missionary for people with leprosy.
- In House of the Dragon, the TV adaptation of George R. R. Martin's Fire and Blood, King Viserys I Targaryen suffers from a debilitating disease where parts of his body develop lesions and slowly rot away over time. Paddy Considine, the actor playing the role, explained on a podcast with Entertainment Weekly that Viserys suffers from "a form of leprosy".[173] Leprosy is not mentioned in the novel, where Viserys instead suffers from various health issues relating to his obesity, including infections and gout.[174]
- The Covenant of Water by 2023 family saga novel by Abraham Verghese
A hero character in the video game The Darkest Dungeon by Red Hook Studios known as the Leper suffers from this disease. He was once a benevolent king who embraced the sick and downtrodden, but abdicated his throne following an assassination attempt after he was infected to wander the world and marvel at its beauty.
Infection of animals
Wild nine-banded armadillos (Dasypus novemcinctus) in south central United States often carry Mycobacterium leprae.[175] This is believed to be because armadillos have a low body temperature. Leprosy lesions appear mainly in cooler body regions such as the skin and mucous membranes of the upper respiratory tract. Because of armadillos' armor, skin lesions are hard to see.[176] Abrasions around the eyes, nose and feet are the most common signs. Infected armadillos make up a large reservoir of M. leprae and may be a source of infection for some humans in the United States or other locations in the armadillos' home range. In armadillo leprosy, lesions do not persist at the site of entry in animals, M. leprae multiply in macrophages at the site of inoculation and lymph nodes.[177]
An outbreak in chimpanzees in West Africa is showing that the bacteria can infect another species and also possibly have additional rodent hosts.[178]
Studies have demonstrated that the disease is endemic in the UK red Eurasian squirrel population, with Mycobacterium leprae and Mycobacterium lepromatosis appearing in different populations. The Mycobacteria leprae strain discovered on Brownsea Island is equated to one thought to have died out in the human population in mediaeval times.[179] Despite this, and speculation regarding past transmission through trade in squirrel furs, there does not seem to be a high risk of squirrel to human transmission from the wild population. Although Leprosy continues to be diagnosed in immigrants to the UK, the last known human case of leprosy arising in the UK was recorded over 200 years ago.[180]
It has been shown that leprosy can reprogram cells in mouse[181][182] and armadillos models[183][184] similarly as how Induced pluripotent stem cells are generated by the transcription factors Myc, Oct3/4, Sox2 and Klf4.
References
- Worobec SM (2008). "Treatment of leprosy/Hansen's disease in the early 21st century". Dermatologic Therapy. 22 (6): 518–537. doi:10.1111/j.1529-8019.2009.01274.x. PMID 19889136. S2CID 42203681.
- "Definition of leprosy". The Free Dictionary. Archived from the original on 22 February 2015. Retrieved 25 January 2015.
- Suzuki K, Akama T, Kawashima A, Yoshihara A, Yotsu RR, Ishii N (February 2012). "Current status of leprosy: epidemiology, basic science and clinical perspectives". The Journal of Dermatology. 39 (2): 121–129. doi:10.1111/j.1346-8138.2011.01370.x. PMID 21973237. S2CID 40027505.
- "Leprosy". World Health Organization (WHO). Archived from the original on 31 January 2021. Retrieved 10 February 2020.
- "New Leprosy Bacterium: Scientists Use Genetic Fingerprint To Nail 'Killing Organism'". ScienceDaily. 28 November 2008. Archived from the original on 13 March 2010. Retrieved 31 January 2010.
- Schreuder PA, Noto S, Richardus JH (January 2016). "Epidemiologic trends of leprosy for the 21st century". Clinics in Dermatology. 34 (1): 24–31. doi:10.1016/j.clindermatol.2015.11.001. PMID 26773620.
- Sotiriou MC, Stryjewska BM, Hill C (September 2016). "Two Cases of Leprosy in Siblings Caused by Mycobacterium lepromatosis and Review of the Literature". The American Journal of Tropical Medicine and Hygiene. 95 (3): 522–527. doi:10.4269/ajtmh.16-0076. PMC 5014252. PMID 27402522.
- "Hansen's Disease (Leprosy) Transmission". cdc.gov. 29 April 2013. Archived from the original on 13 March 2015. Retrieved 28 February 2015.
- Guidelines for the diagnosis, treatment and prevention of leprosy. World Health Organization. Regional Office for South-East Asia. 2018. p. xiii. hdl:10665/274127. ISBN 978-92-9022-638-3.
- Montoya D, Modlin RL (2010). Learning from leprosy: insight into the human innate immune response. Advances in Immunology. Vol. 105. pp. 1–24. doi:10.1016/S0065-2776(10)05001-7. ISBN 978-0-12-381302-2. PMID 20510728.
- Byrne JP (2008). Encyclopedia of pestilence, pandemics, and plagues. Westport, Conn.[u.a.]: Greenwood Press. p. 351. ISBN 978-0-313-34102-1.
- CDC (26 January 2018). "World Leprosy Day". Centers for Disease Control and Prevention. Archived from the original on 15 June 2019. Retrieved 4 July 2019.
- "Global leprosy situation, 2012". Weekly Epidemiological Record. 87 (34): 317–328. August 2012. PMID 22919737.
- Rodrigues LC, Lockwood DN (June 2011). "Leprosy now: epidemiology, progress, challenges, and research gaps". The Lancet. Infectious Diseases. 11 (6): 464–470. doi:10.1016/S1473-3099(11)70006-8. PMID 21616456.
- "Hansen's Disease Data & Statistics". Health Resources and Services Administration. Archived from the original on 4 January 2015. Retrieved 12 January 2015.
- "Central Florida is a hot spot for leprosy, report says". CNN. August 2023.
- Walsh F (31 March 2007). "The hidden suffering of India's lepers". BBC News. Archived from the original on 29 May 2007.
- Lyn TE (13 September 2006). "Ignorance breeds leper colonies in China". Independat News & Media. Archived from the original on 8 April 2010. Retrieved 31 January 2010.
- Pisuthipan A (6 July 2020). "Forgotten victims of the virus". Bangkok Post. Archived from the original on 28 August 2021. Retrieved 6 July 2020.
- Nunzi E, Massone C, eds. (2012). Leprosy a practical guide. Milan: Springer. p. 326. ISBN 978-88-470-2376-5. Archived from the original on 8 September 2017.
- "Neglected Tropical Diseases". cdc.gov. 6 June 2011. Archived from the original on 4 December 2014. Retrieved 28 November 2014.
- McMenamin D (2011). Leprosy and stigma in the South Pacific: a region-by-region history with first person accounts. Jefferson, N.C.: McFarland. p. 17. ISBN 978-0-7864-6323-7. Archived from the original on 19 May 2016.
- "Signs and Symptoms | Hansen's Disease (Leprosy) | CDC". www.cdc.gov. 22 October 2018. Archived from the original on 22 July 2019. Retrieved 22 July 2019.
- "Pathogenesis and Pathology of Leprosy". International Textbook of Leprosy. 11 February 2016. Archived from the original on 22 July 2019. Retrieved 22 July 2019.
- WHO Expert Committee on Leprosy – Eight report (PDF). World Health Organization (WHO). 2012. pp. 11–12. ISBN 978-9241209687. Archived from the original (PDF) on 5 August 2013. Retrieved 9 May 2018.
- Talhari C, Talhari S, Penna GO (2015). "Clinical aspects of leprosy". Clinics in Dermatology. 33 (1): 26–37. doi:10.1016/j.clindermatol.2014.07.002. PMID 25432808.
- Kulkarni GS (2008). Textbook of Orthopedics and Trauma (2nd ed.). Jaypee Brothers Publishers. p. 779. ISBN 978-81-8448-242-3.
- "Q and A about leprosy". American Leprosy Missions. Archived from the original on 4 October 2012. Retrieved 22 January 2011.
Do fingers and toes fall off when someone gets leprosy? No. The bacillus attacks nerve endings and destroys the body's ability to feel pain and injury. Without feeling pain, people injure themselves on fire, thorns, rocks, even hot coffee cups. Injuries become infected and result in tissue loss. Fingers and toes become shortened and deformed as the cartilage is absorbed into the body.
- de Sousa JR, Sotto MN, Simões Quaresma JA (28 November 2017). "Leprosy As a Complex Infection: Breakdown of the Th1 and Th2 Immune Paradigm in the Immunopathogenesis of the Disease". Frontiers in Immunology. 8: 1635. doi:10.3389/fimmu.2017.01635. PMC 5712391. PMID 29234318.
- Reinar LM, Forsetlund L, Lehman LF, Brurberg KG (July 2019). "Interventions for ulceration and other skin changes caused by nerve damage in leprosy". The Cochrane Database of Systematic Reviews. 2019 (7): CD012235. doi:10.1002/14651858.CD012235.pub2. PMC 6699662. PMID 31425632.
- Ryan KU, Ray CJ, eds. (2004). Sherris Medical Microbiology (4th ed.). McGraw Hill. pp. 451–53. ISBN 978-0-8385-8529-0. OCLC 61405904.
- "Genomics Insights into the Biology and Evolution of Leprosy Bacilli". International Textbook of Leprosy. 11 February 2016. Archived from the original on 12 February 2019. Retrieved 11 February 2019.
- McMurray DN (1996). "Mycobacteria and Nocardia". In Baron S; et al. (eds.). Baron's Medical Microbiology (4th ed.). Univ of Texas Medical Branch. ISBN 978-0-9631172-1-2. OCLC 33838234. Archived from the original on 12 February 2009.
- Bhattacharya S, Vijayalakshmi N, Parija SC (October 2002). "Uncultivable bacteria: implications and recent trends towards identification". Indian Journal of Medical Microbiology. 20 (4): 174–177. doi:10.1016/S0255-0857(21)03184-4. PMID 17657065.
- "WHO | Microbiology: culture in vitro". World Health Organization (WHO). Archived from the original on 9 August 2020. Retrieved 22 July 2019.
- "The Armadillo Model for Leprosy". International Textbook of Leprosy. 11 February 2016. Archived from the original on 22 July 2019. Retrieved 22 July 2019.
- Loughry WJ, Truman RW, McDonough CM, Tilak MK, Garnier S, et al. (2009) "Is leprosy spreading among nine-banded armadillos in the southeastern United States?" J Wildl Dis 45: 144–52.
- Meredith A, Del Pozo J, Smith S, Milne E, Stevenson K, McLuckie J (September 2014). "Leprosy in red squirrels in Scotland". The Veterinary Record. 175 (11): 285–286. doi:10.1136/vr.g5680. PMID 25234460. S2CID 207046489.
- Monot M, Honoré N, Garnier T, Araoz R, Coppee JY, et al. (2005). "On the origin of leprosy". Science 308: 1040–42.
- Han XY, Silva FJ (February 2014). "On the age of leprosy". PLOS Neglected Tropical Diseases. 8 (2): e2544. doi:10.1371/journal.pntd.0002544. PMC 3923669. PMID 24551248.
- "Red squirrels in the British Isles are infected with leprosy bacilli" Archived 12 June 2022 at the Wayback Machine, Dr. Andrej Benjak, Prof Anna Meredith and others. Science, 11 November 2016. Archived 12 June 2022 at the Wayback Machine. Retrieved 11 November 2016.
- "Could squirrel fur trade have contributed to England's medieval leprosy outbreak?". ScienceDaily. Archived from the original on 22 November 2018. Retrieved 21 November 2018.
- Inskip S, Taylor GM, Anderson S, Stewart G (November 2017). "Leprosy in pre-Norman Suffolk, UK: biomolecular and geochemical analysis of the woman from Hoxne". Journal of Medical Microbiology. 66 (11): 1640–1649. doi:10.1099/jmm.0.000606. PMID 28984227. S2CID 33997231.
- Penna ML, Penna GO, Iglesias PC, Natal S, Rodrigues LC (May 2016). "Anti-PGL-1 Positivity as a Risk Marker for the Development of Leprosy among Contacts of Leprosy Cases: Systematic Review and Meta-analysis". PLOS Neglected Tropical Diseases. 10 (5): e0004703. doi:10.1371/journal.pntd.0004703. PMC 4871561. PMID 27192199.
- Alcaïs A, Mira M, Casanova JL, Schurr E, Abel L (February 2005). "Genetic dissection of immunity in leprosy". Current Opinion in Immunology. 17 (1): 44–48. doi:10.1016/j.coi.2004.11.006. PMID 15653309.
- Lockwood DN, Lambert SM (January 2011). "Human immunodeficiency virus and leprosy: an update". Dermatologic Clinics. 29 (1): 125–128. doi:10.1016/j.det.2010.08.016. PMID 21095536.
- "Epidemiology of Leprosy". International Textbook of Leprosy. 11 February 2016. Archived from the original on 23 July 2019. Retrieved 30 July 2019.
- Chavarro-Portillo B, Soto CY, Guerrero MI (September 2019). "Mycobacterium leprae's evolution and environmental adaptation". Acta Tropica. 197: 105041. doi:10.1016/j.actatropica.2019.105041. PMID 31152726. S2CID 173188912.
- Eichelmann K, González González SE, Salas-Alanis JC, Ocampo-Candiani J (September 2013). "Leprosy. An update: definition, pathogenesis, classification, diagnosis, and treatment". Actas Dermo-Sifiliograficas. 104 (7): 554–563. doi:10.1016/j.adengl.2012.03.028. PMID 23870850. S2CID 3442319.
- Joel Carlos Lastória JC, Milanez Morgado de Abreu MA (March–April 2014). "Leprosy: review of the epidemiological, clinical, and etiopathogenic aspects - Part 1". An Bras Dermatol. 89 (2): 205–218. doi:10.1590/abd1806-4841.20142450. PMC 4008049. PMID 24770495.
- "Hansen's Disease (Leprosy) Transmission". cdc.gov. 29 April 2013. Archived from the original on 13 March 2015. Retrieved 28 February 2015.
- Lockwood DN, Kumar B (June 2004). "Treatment of leprosy". BMJ. 328 (7454): 1447–1448. doi:10.1136/bmj.328.7454.1447. PMC 428501. PMID 15205269.
- "What Is Leprosy?"| from News-Medical.Net – Latest Medical News and Research from Around the World. Web. 20 Nov. 2010. "What is Leprosy?". News-Medical.net. 18 November 2009. Archived from the original on 6 June 2013. Retrieved 14 May 2013..
- Truman RW, Singh P, Sharma R, Busso P, Rougemont J, Paniz-Mondolfi A, et al. (April 2011). "Probable zoonotic leprosy in the southern United States". The New England Journal of Medicine. 364 (17): 1626–1633. doi:10.1056/NEJMoa1010536. PMC 3138484. PMID 21524213.
- "Hansen's Disease (Leprosy) Transmission". cdc.gov. 29 April 2013. Archived from the original on 13 March 2015. Retrieved 28 February 2015.
- Cambri G, Mira MT (20 July 2018). "Genetic Susceptibility to Leprosy-From Classic Immune-Related Candidate Genes to Hypothesis-Free, Whole Genome Approaches". Frontiers in Immunology. 9: 1674. doi:10.3389/fimmu.2018.01674. PMC 6062607. PMID 30079069.
- Cook GC (2009). Manson's tropical diseases (22nd ed.). [Edinburgh]: Saunders. p. 1056. ISBN 978-1-4160-4470-3. Archived from the original on 4 September 2017.
- Buschman E, Skamene E (June 2004). "Linkage of leprosy susceptibility to Parkinson's disease genes". International Journal of Leprosy and Other Mycobacterial Diseases. 72 (2): 169–170. doi:10.1489/1544-581X(2004)072<0169:LOLSTP>2.0.CO;2. PMID 15301585. S2CID 43103579.
- Bhat RM, Prakash C (2012). "Leprosy: an overview of pathophysiology". Interdisciplinary Perspectives on Infectious Diseases. 2012: 181089. doi:10.1155/2012/181089. PMC 3440852. PMID 22988457.
- Moschella SL, Garcia-Albea V (September 2016). "International Textbook of Leprosy" (PDF). Differential Diagnosis of Leprosy. p. 3, Section 2.3. Archived (PDF) from the original on 16 July 2020. Retrieved 4 July 2019.
- U.S. Department of Health and Human Services, Health Resources and Services Administration. (n.d.). National Hansen's disease (leprosy) program Retrieved from "National Hansen's Disease (Leprosy) Program". Archived from the original on 10 February 2011. Retrieved 12 May 2013.
- Martinez AN, Talhari C, Moraes MO, Talhari S (April 2014). "PCR-based techniques for leprosy diagnosis: from the laboratory to the clinic". PLOS Neglected Tropical Diseases. 8 (4): e2655. doi:10.1371/journal.pntd.0002655. PMC 3983108. PMID 24722358.
- Tatipally S, Srikantam A, Kasetty S (October 2018). "Polymerase Chain Reaction (PCR) as a Potential Point of Care Laboratory Test for Leprosy Diagnosis-A Systematic Review". Tropical Medicine and Infectious Disease. 3 (4): 107. doi:10.3390/tropicalmed3040107. PMC 6306935. PMID 30275432.
- Smith DS (19 August 2008). "Leprosy: Overview". eMedicine Infectious Diseases. Archived from the original on 18 February 2010. Retrieved 1 February 2010.
- Singh N, Manucha V, Bhattacharya SN, Arora VK, Bhatia A (June 2004). "Pitfalls in the cytological classification of borderline leprosy in the Ridley-Jopling scale". Diagnostic Cytopathology. 30 (6): 386–388. doi:10.1002/dc.20012. PMID 15176024. S2CID 29757876.
- Ridley DS, Jopling WH (1966). "Classification of leprosy according to immunity. A five-group system". International Journal of Leprosy and Other Mycobacterial Diseases. 34 (3): 255–273. PMID 5950347.
- James WD, Berger TG, Elston DM, Odom RB (2006). Andrews' Diseases of the Skin: clinical Dermatology. Saunders Elsevier. pp. 344–46. ISBN 978-0-7216-2921-6.
- Lastória JC, Abreu MA (2014). "Leprosy: a review of laboratory and therapeutic aspects--part 2". Anais Brasileiros de Dermatologia. 89 (3): 389–401. doi:10.1590/abd1806-4841.20142460. PMC 4056695. PMID 24937811.
- Kumar, Bhushan; Uprety, Shraddha; Dogra, Sunil (11 February 2016). "Clinical Diagnosis of Leprosy". International Textbook of Leprosy. Archived from the original on 13 February 2019. Retrieved 12 February 2019.
- Jardim MR, Antunes SL, Santos AR, Nascimento OJ, Nery JA, Sales AM, et al. (July 2003). "Criteria for diagnosis of pure neural leprosy". Journal of Neurology. 250 (7): 806–809. doi:10.1007/s00415-003-1081-5. PMID 12883921. S2CID 20070335.
- Mendiratta V, Khan A, Jain A (2006). "Primary neuritic leprosy: a reappraisal at a tertiary care hospital". Indian Journal of Leprosy. 78 (3): 261–267. PMID 17120509.
- Ishida Y, Pecorini L, Guglielmelli E (July 2000). "Three cases of pure neuritic (PN) leprosy at detection in which skin lesions became visible during their course". Nihon Hansenbyo Gakkai Zasshi = Japanese Journal of Leprosy. 69 (2): 101–106. doi:10.5025/hansen.69.101. PMID 10979277.
- Mishra B, Mukherjee A, Girdhar A, Husain S, Malaviya GN, Girdhar BK (1995). "Neuritic leprosy: further progression and significance". Acta Leprologica. 9 (4): 187–194. PMID 8711979.
- Talwar S, Jha PK, Tiwari VD (September 1992). "Neuritic leprosy: epidemiology and therapeutic responsiveness". Leprosy Review. 63 (3): 263–268. doi:10.5935/0305-7518.19920031. PMID 1406021.
- "Embrace a village - FAQ". Archived from the original on 2 February 2023. Retrieved 2 February 2023.
- Duthie MS, Gillis TP, Reed SG (November 2011). "Advances and hurdles on the way toward a leprosy vaccine". Human Vaccines. 7 (11): 1172–1183. doi:10.4161/hv.7.11.16848. PMC 3323495. PMID 22048122.
- Setia MS, Steinmaus C, Ho CS, Rutherford GW (March 2006). "The role of BCG in prevention of leprosy: a meta-analysis". The Lancet. Infectious Diseases. 6 (3): 162–170. doi:10.1016/S1473-3099(06)70412-1. PMID 16500597.
- Merle CS, Cunha SS, Rodrigues LC (February 2010). "BCG vaccination and leprosy protection: review of current evidence and status of BCG in leprosy control". Expert Review of Vaccines. 9 (2): 209–222. doi:10.1586/ERV.09.161. PMID 20109030. S2CID 34309843.
- World Health Organization (June 2018). "BCG vaccine: WHO position paper, February 2018 - Recommendations". Vaccine. 36 (24): 3408–3410. doi:10.1016/j.vaccine.2018.03.009. PMID 29609965. S2CID 4570754.
- Moraes MO, Düppre NC (January 2021). "Leprosy post-exposure prophylaxis: innovation and precision public health". The Lancet. Global Health. 9 (1): e8–e9. doi:10.1016/S2214-109X(20)30512-X. PMID 33338461.
- Yamazaki-Nakashimada MA, Unzueta A, Berenise Gámez-González L, González-Saldaña N, Sorensen RU (August 2020). "BCG: a vaccine with multiple faces". Human Vaccines & Immunotherapeutics. 16 (8): 1841–1850. doi:10.1080/21645515.2019.1706930. PMC 7482865. PMID 31995448.
- "Leprosy Vaccine". American Leprosy Missions. Archived from the original on 15 November 2015. Retrieved 20 October 2015.
- "Trial set for world's first leprosy vaccine". The Guardian. 6 June 2014. Archived from the original on 11 October 2015. Retrieved 20 October 2015.
- "China's Mars plans, leprosy vaccine and self-driving taxis". Nature. 537 (7618): 12–13. September 2016. Bibcode:2016Natur.537...12.. doi:10.1038/537012a. PMID 27582199.
- Clinical trial number NCT03302897 for "Phase 1 LEP-F1 + GLA-SE Vaccine Trial in Healthy Adult Volunteers" at ClinicalTrials.gov
- Duthie MS, Frevol A, Day T, Coler RN, Vergara J, Rolf T, et al. (February 2020). "A phase 1 antigen dose escalation trial to evaluate safety, tolerability and immunogenicity of the leprosy vaccine candidate LepVax (LEP-F1 + GLA-SE) in healthy adults". Vaccine. 38 (7): 1700–1707. doi:10.1016/j.vaccine.2019.12.050. PMID 31899025. S2CID 209677501.
- "WHO | MDT and drug resistance". World Health Organization (WHO). Archived from the original on 4 October 2014. Retrieved 22 July 2019.
- Reibel F, Cambau E, Aubry A (September 2015). "Update on the epidemiology, diagnosis, and treatment of leprosy". Médecine et Maladies Infectieuses. 45 (9): 383–393. doi:10.1016/j.medmal.2015.09.002. PMID 26428602.
- Somar P, Waltz MM, van Brakel WH (2020). "The impact of leprosy on the mental wellbeing of leprosy-affected persons and their family members - a systematic review". Global Mental Health. 7: e15. doi:10.1017/gmh.2020.3. PMC 7379324. PMID 32742673.
- Rao D, Elshafei A, Nguyen M, Hatzenbuehler ML, Frey S, Go VF (February 2019). "A systematic review of multi-level stigma interventions: state of the science and future directions". BMC Medicine. 17 (1): 41. doi:10.1186/s12916-018-1244-y. PMC 6377735. PMID 30770756.
- "Global leprosy update, 2016: accelerating reduction of disease burden". Weekly Epidemiological Record. 92 (35): 501–519. September 2017. hdl:10665/258841. PMID 28861986.
- "Leprosy new case detection rates, 2016". World Health Organization (WHO). Archived from the original on 19 December 2019. Retrieved 19 December 2019.
- "Mortality and Burden of Disease Estimates for WHO Member States in 2002" (xls). World Health Organization (WHO). 2002. Archived from the original on 16 January 2013.
- "WHO | Leprosy: new data show steady decline in new cases". WHO. Archived from the original on 22 October 2019. Retrieved 26 February 2020.
- "WHO | Global leprosy update, 2015: time for action, accountability and inclusion". World Health Organization (WHO). Archived from the original on 18 October 2016. Retrieved 14 January 2019.
- Maggie Veatch (21 February 2019). "Leprosy still lurks in United States, study says". CNN. Archived from the original on 20 August 2020. Retrieved 24 February 2019.
- "About ILEP". ILEP. Archived from the original on 12 August 2014. Retrieved 25 August 2014.
- "Epidemiology of Leprosy". International Textbook of Leprosy. 11 February 2016. Archived from the original on 23 July 2019. Retrieved 23 July 2019.
- Organization, World Health (1985). Epidemiology of leprosy in relation to control. Report of a WHO Study Group. World Health Organization technical report series. Vol. 716. World Health Organization. pp. 1–60. hdl:10665/40171. ISBN 978-92-4-120716-4. OCLC 12095109. PMID 3925646.
- "Number of new leprosy cases". World Health Organization (WHO). 13 September 2021. Archived from the original on 26 September 2021. Retrieved 3 June 2022.
- Monot, Marc; Honoré, Nadine; Garnier, Thierry; Araoz, Romul; Coppée, Jean-Yves; Lacroix, Céline; Sow, Samba; Spencer, John S.; Truman, Richard W.; Williams, Diana L.; Gelber, Robert; Virmond, Marcos; Flageul, Béatrice; Cho, Sang-Nae; Ji, Baohong; Paniz-Mondolfi, Alberto; Convit, Jacinto; Young, Saroj; Fine, Paul E.; Rasolofo, Voahangy; Brennan, Patrick J.; Cole, Stewart T. (2005). "On the origin of leprosy" (PDF). Science. 308 (5724): 1040–1042. doi:10.1126/science/1109759. PMID 15894530. S2CID 86109194. Archived (PDF) from the original on 25 January 2023. Retrieved 22 November 2022.
- Monot M, Honoré N, Garnier T, Araoz R, Coppée JY, Lacroix C, et al. (May 2005). "On the origin of leprosy" (PDF). Science. 308 (5724): 1040–1042. doi:10.1126/science/1109759. PMID 15894530. S2CID 86109194. Archived (PDF) from the original on 25 January 2023. Retrieved 22 November 2022.
- Robbins G, Tripathy VM, Misra VN, Mohanty RK, Shinde VS, Gray KM, Schug MD (May 2009). "Ancient skeletal evidence for leprosy in India (2000 B.C.)". PLOS ONE. 4 (5): e5669. Bibcode:2009PLoSO...4.5669R. doi:10.1371/journal.pone.0005669. PMC 2682583. PMID 19479078.
- Robbins Schug G, Blevins KE, Cox B, Gray K, Mushrif-Tripathy V (December 2013). "Infection, disease, and biosocial processes at the end of the Indus Civilization". PLOS ONE. 8 (12): e84814. Bibcode:2013PLoSO...884814R. doi:10.1371/journal.pone.0084814. PMC 3866234. PMID 24358372.
- Robbins G, Tripathy VM, Misra VN, Mohanty RK, Shinde VS, Gray KM, Schug MD (May 2009). "Ancient skeletal evidence for leprosy in India (2000 B.C.)". PLOS ONE. 4 (5): e5669. Bibcode:2009PLoSO...4.5669R. doi:10.1371/journal.pone.0005669. PMC 2682583. PMID 19479078.
- "DNA of Jesus-Era Shrouded Man in Jerusalem Reveals Earliest Case of Leprosy". ScienceDaily. 16 December 2009. Archived from the original on 20 December 2009. Retrieved 31 January 2010.
- Schuenemann VJ, Avanzi C, Krause-Kyora B, Seitz A, Herbig A, Inskip S, et al. (May 2018). "Ancient genomes reveal a high diversity of Mycobacterium leprae in medieval Europe". PLOS Pathogens. 14 (5): e1006997. doi:10.1371/journal.ppat.1006997. PMC 5944922. PMID 29746563.
- Lendrum FC (1954). "The Name 'Leprosy'". Etc: A Review of General Semantics. Vol. 12. Institute of General Semantics. pp. 37–47. JSTOR 24234298. Archived from the original on 13 April 2022. Retrieved 13 April 2022.
- Haubrich, William S. (2003). Medical Meanings: A Glossary of Word Origins. ACP Press. p. 133. ISBN 978-1-930513-49-5.
- Wilkins, Michael; Evans, Craig A.; Bock, Darrell; Köstenberger, Andreas J. (1 October 2013). The Gospels and Acts. B&H. p. 194. ISBN 978-1-4336-8101-1. Archived from the original on 12 January 2023. Retrieved 15 July 2018.
- Encyclopedia of Jewish Medical Ethics. Feldheim Publishers. 2003. p. 951. ISBN 978-1-58330-592-8.
- Adams, Francis (1678). The Seven Books of Paulus Aegineta: Translated from the Greek with Commentary Embracing a Complete View of the Knowledge Possessed by the Greeks, Romans and Arabians on all Subjects Connected with Medicine and Surgery. London: Sydenham Society.
- Roman: Celsus, Pliny, Serenus Samonicus, Scribonius Largus, Caelius Aurelianus, Themison, Octavius Horatianus, Marcellus the Emperic; Greek: Aretaeus, Plutarch, Galen, Oribasius, Aetius (Aëtius of Amida or Sicamus Aëtius), Actuarius, Nonnus, Psellus, Leo, Myrepsus; Arabic: Scrapion, Avenzoar, Albucasis, Haly Abbas translated by Stephanus Antiochensis, Alsharavius, Rhases (Abū Bakr al-Rāzī), and Guido de Cauliaco.
- Rotberg, Robert I. (2001). Population History and the Family: A Journal of Interdisciplinary History Reader. MIT Press. p. 132. ISBN 978-0-262-68130-8.
- Montgomerie JZ (1988). "Leprosy in New Zealand". The Journal of the Polynesian Society. 97 (2): 115–152. PMID 11617451. Archived from the original on 11 February 2018. Retrieved 3 September 2019.
- Irgens LM (March 2002). "Oppdagelsen av leprabasillen" [The discovery of the leprosy bacillus]. Tidsskrift for den Norske Laegeforening (in Norwegian). 122 (7): 708–709. PMID 11998735.
- Andrew Baum; et al. (1997). Cambridge handbook of psychology, health and medicine. Cambridge, UK: Cambridge University Press. p. 521. ISBN 978-0-521-43686-1. Archived from the original on 11 June 2016.
- Rees RJ, Pearson JM, Waters MF (January 1970). "Experimental and clinical studies on rifampicin in treatment of leprosy". British Medical Journal. 1 (5688): 89–92. doi:10.1136/bmj.1.5688.89. PMC 1699176. PMID 4903972.
- Yawalkar SJ, McDougall AC, Languillon J, Ghosh S, Hajra SK, Opromolla DV, Tonello CJ (May 1982). "Once-monthly rifampicin plus daily dapsone in initial treatment of lepromatous leprosy". Lancet. 1 (8283): 1199–1202. doi:10.1016/S0140-6736(82)92334-0. PMID 6122970. S2CID 38629414.
- "Treatment | Hansen's Disease (Leprosy)". Centers for Disease Control and Prevention. 22 October 2018. Archived from the original on 7 October 2022. Retrieved 7 October 2022.
- "Syphilis through history". Archived 2013-05-13 at the Wayback Machine Encyclopædia Britannica.
- "Communicable Diseases Department, Leprosy FAQ". World Health Organization (WHO). 25 May 2006. Archived from the original on 1 February 2010. Retrieved 31 January 2010.
- Japan repealed its "Leprosy Prevention Laws" in 1996, but former patients still reside in sanatoriums.
- "Koizumi apologises for leper colonies". BBC News. 25 May 2001. Archived from the original on 17 April 2009.
- "Former Hansen's disease patients still struggling with prejudice". Japan Times. 7 June 2007. Archived from the original on 26 August 2009.
- Arch Dermato Syphilis 1898; 44:159–174
- Vernon G (February 2022). "Dr E W Price, the discoverer of podoconiosis". Journal of Medical Biography. 30 (1): 2–5. doi:10.1177/0967772019888406. PMID 31735101. S2CID 208142196.
- "Global leprosy (Hansen disease) update, 2019: time to step-up prevention initiatives". Weekly Epidemiological Record. 95 (36): 417–40. 4 September 2020. hdl:10665/334140.
- Gussow, Zachary (2021) [1989]. Leprosy, Racism, And Public Health: Social Policy In Chronic Disease Control. Routledge. ISBN 978-0-3670-0292-3.
- "Hindu Marriage Act: Parliament passes law removing leprosy as ground for divorce". The Economic Times. 13 February 2019. Archived from the original on 15 July 2020. Retrieved 14 July 2020.
- leprosy Archived 14 April 2021 at the Wayback Machine Novartis (accessed on 2021-04-14)
- "WHO donated MDT". World Health Organization (WHO). Archived from the original on 11 October 2014. Retrieved 23 July 2019.
- Singh KS, Pandey BD (March 2012). "Leprosy – Hidden Disease?". Science Reporter. 49 (3). Archived from the original on 3 August 2020. Retrieved 4 August 2019.
- Jacob JT, Franco-Paredes C (January 2008). "The stigmatization of leprosy in India and its impact on future approaches to elimination and control". PLOS Neglected Tropical Diseases. 2 (1): e113. doi:10.1371/journal.pntd.0000113. PMC 2217676. PMID 18235845.
- Grzybowski A, Nita M (January 2016). "Leprosy in the Bible". Clinics in Dermatology. 34 (1): 3–7. doi:10.1016/j.clindermatol.2015.10.003. PMID 26773616.
- [See: Orr, James, M.A., D.D. General Editor. "Entry for 'Leper; Leprosy'". "International Standard Bible Encyclopedia". 1915. Access-date=January 6, 2017
- Shurpin Y (6 April 2022). "Is Tzaraat Leprosy?". Chabad.org. Archived from the original on 6 April 2022. Retrieved 6 April 2022.
- van der Loos H (1968). The Miracles of Jesus. Brill Archive. p. 464. Archived from the original on 1 May 2023. Retrieved 21 May 2022.
- Lewis G (December 1987). "A Lesson from Leviticus: Leprosy". Man. 22 (4): 598. doi:10.2307/2803354. JSTOR 2803354.
- Covey HC (2001). "People with leprosy (Hansen's disease) during the Middle Ages" (PDF). Social Science Journal. 38 (2): 315–21. doi:10.1016/S0362-3319(01)00116-1. S2CID 145166840. Archived from the original (PDF) on 15 August 2016. Retrieved 25 June 2016.
- Brenner E (2010). "Recent Perspectives on Leprosy in Medieval Western Europe". History Compass. 8 (5): 388–406. doi:10.1111/j.1478-0542.2009.00674.x. Archived from the original on 1 May 2023. Retrieved 10 November 2022.
- Barber M (July 1994). "The Order of Saint Lazarus and the Crusades". The Catholic Historical Review. 80 (3): 439–456.
- Miller TS, Smith-Savage R (2006). "Medieval Leprosy Reconsidered". International Social Science Review. 81 (1/2): 16–28. JSTOR 41887256. Archived from the original on 10 November 2022. Retrieved 10 November 2022.
- Miller TS, Nesbitt JW (19 April 2014). Walking Corpses: Leprosy in Byzantium and the Medieval West. Cornell University Press. ISBN 978-0801470769.
- Le Goff J (1990). The Medieval world. London: Collins & Brown. ISBN 978-1-85585-081-1.
- Clay R (1909). The Medieval Hospitals of EnglishLand. Cornell University Library. ISBN 978-1-112-20443-2.
- Rubin S (1974). Medieval English medicine. New York: Barnes & Noble Books: Newton Abbot: David & Charles. ISBN 978-0-06-496016-8.
- "Medieval Leprosy". Intriguing history. 24 April 2017. Archived from the original on 6 December 2022. Retrieved 10 November 2022.
- Covey HC (1 June 2001). "People with leprosy (Hansen's disease) during the Middle Ages". The Social Science Journal. 38 (2): 315–21. doi:10.1016/S0362-3319(01)00116-1. S2CID 145166840.
- Moore RI (2007). The Formation of a Persecuting Society. Oxford: Blackwell. ISBN 978-1-4051-2964-0.
- Alter A (2010). Genetic susceptibility to leprosy. McGill University (Canada). ISBN 978-0-494-72613-6.
{{cite book}}: CS1 maint: location missing publisher (link) - Svein Atle Skålevåg. "Daniel Cornelius Danielssen". Store norske leksikon. Archived from the original on 13 January 2017. Retrieved 1 January 2017.
- Svein Atle Skålevåg (28 September 2014). "Carl Wilhelm Boeck". Store norske leksikon. Archived from the original on 13 January 2017. Retrieved 1 January 2017.
- Dobson M (2013) [2007]. Disease: The extraordinary stories behind history's deadliest killers. UK: Quercus Editions Ltd. pp. 26–27. ISBN 978-1-4351-5166-6.
- Bhukhan, Aashni; Dunn, Charles; Nathoo, Rajiv (2023). "Case Report of Leprosy in Central Florida, USA, 2022 - Volume 29, Number 8—August 2023 - Emerging Infectious Diseases journal - CDC". Emerging Infectious Diseases. 29 (8): 1698–1700. doi:10.3201/eid2908.220367. PMC 10370849. PMID 37486691.
- admin (11 February 2016). "Stigma Related to Leprosy – A Scientific View". International Textbook of Leprosy. Archived from the original on 7 October 2022. Retrieved 7 October 2022.
- White C (September 2005). "Explaining a complex disease process: talking to patients about Hansen's disease (leprosy) in Brazil". Medical Anthropology Quarterly. 19 (3): 310–330. doi:10.1525/maq.2005.19.3.310. PMID 16222964.
- "Leprosy and Its Stigma Are Both Curable". Office for Science and Society. Archived from the original on 7 October 2022. Retrieved 7 October 2022.
- Barrett R (June 2005). "Self-mortification and the stigma of leprosy in northern India". Medical Anthropology Quarterly. 19 (2): 216–230. doi:10.1525/maq.2005.19.2.216. JSTOR 3655487. PMID 15974328.
- "WHO | The Global Leprosy Strategy". WHO. Archived from the original on 17 September 2006. Retrieved 9 February 2021.
- "WHO | Elimination of leprosy FAQ". WHO. Archived from the original on 18 October 2014. Retrieved 9 February 2021.
- "Leprosy elimination". World Health Organization (WHO). Archived from the original on 14 March 2014. Retrieved 3 July 2019.
- Staples J (2014). "Communities of the afflicted: constituting leprosy through place in South India". Medical Anthropology. 33 (1): 6–20. doi:10.1080/01459740.2012.714021. PMID 24383749. S2CID 24595253. Archived from the original on 13 February 2023. Retrieved 22 November 2022.
- "A look at 6 contested historical events of Seychelles". Seychelles News Agency. Archived from the original on 17 November 2017. Retrieved 15 November 2017.
- "Seychelles: 14 Inspiring Women of Seychelles". Seychelles News Agency (Victoria). 9 March 2017. Archived from the original on 17 November 2017. Retrieved 15 November 2017.
- "Nation Home". Seychelles Nation. 14 January 2016. Archived from the original on 17 November 2017. Retrieved 16 November 2017.
- "Diocesan committee prepares to commemorate Josephine Cafrine's 109th death anniversary". Seychelles Weekly. 8 January 2016. Archived from the original on 4 April 2016. Retrieved 15 November 2017.
- Tayman J (2007). The Colony: The Harrowing True Story of the Exiles of Molokai. New York: Simon and Schuster. ISBN 978-0-7432-3301-9. Archived from the original on 2 January 2016.
- Hamilton, Bernard (2000). The leper king and his Heirs: Baldwin IV and the Crusader Kingdom of Jerusalem. Cambridge: Cambridge University Press. ISBN 978-0-521-64187-6.
- Lazatin H (8 January 2018). "5 Famous Spies Who Made Philippine History". Esquiremag.ph. Archived from the original on 22 September 2021. Retrieved 23 March 2022.
- Webber R (2015). Disease Selection: The Way Disease Changed the World. CABI. p. 8. ISBN 978-1-78064-682-4. Archived from the original on 8 September 2017.
- Cung giu Nguyên (1955). "Contemporary Vietnamese Writing". Books Abroad. University of Oklahoma. 29 (1): 19–25. doi:10.2307/40093803. JSTOR 40093803.
- Bryant A (1995). Sekigahara 1600: The Final Struggle for Power (Campaign Series, 40). Osprey Publishing (UK). ISBN 978-1-85532-395-7. Retrieved 28 February 2010.
- "A Burnt-Out Case | novel by Greene". Encyclopedia Britannica. Archived from the original on 16 August 2021. Retrieved 16 August 2021.
- "EW's West of Westeros: EW goes to the set of House of the Dragon episode 8 on Apple Podcasts". Apple Podcasts. Archived from the original on 20 October 2022. Retrieved 20 October 2022.
- "House of the Dragon: All the Fire and Blood Easter Eggs From Episode 8". Vanity Fair. 10 October 2022. Archived from the original on 26 November 2022. Retrieved 28 December 2022.
- Truman R (September 2005). "Leprosy in wild armadillos". Leprosy Review. 76 (3): 198–208. doi:10.47276/lr.76.3.198. PMID 16248207.
- Sharma R, Lahiri R, Scollard DM, Pena M, Williams DL, Adams LB, et al. (January 2013). "The armadillo: a model for the neuropathy of leprosy and potentially other neurodegenerative diseases". Disease Models & Mechanisms. 6 (1): 19–24. doi:10.1242/dmm.010215. PMC 3529335. PMID 23223615.
- Job CK, Drain V, Truman R, Deming AT, Sanchez RM, Hastings RC (April 1992). "The pathogenesis of leprosy in the nine-banded armadillo and the significance of IgM antibodies to PGL-1". Indian Journal of Leprosy. 64 (2): 137–151. PMID 1607712. INIST:4390813.
- Kupferschmidt K (11 November 2020). "Leprosy, ancient scourge of humans, found to assail wild chimpanzees". Science. Archived from the original on 13 October 2021. Retrieved 1 July 2021.
- "Leprosy in Red Squirrels in the UK". Vet Record. 30 March 2019. Retrieved 25 May 2022.
- "Medieval love of squirrel fur may have helped spread leprosy, study reveals". Guardian. 25 October 2017. Archived from the original on 25 May 2022. Retrieved 25 May 2022.
- Masaki T, Qu J, Cholewa-Waclaw J, Burr K, Raaum R, Rambukkana A (January 2013). "Reprogramming adult Schwann cells to stem cell-like cells by leprosy bacilli promotes dissemination of infection". Cell. 152 (1–2): 51–67. doi:10.1016/j.cell.2012.12.014. PMC 4314110. PMID 23332746.
- "Leprosy bacteria use 'biological alchemy'". BBC News. 17 January 2013. Archived from the original on 18 November 2022. Retrieved 18 November 2022.
- Hess S, Kendall TJ, Pena M, Yamane K, Soong D, Adams L, et al. (November 2022). "In vivo partial reprogramming by bacteria promotes adult liver organ growth without fibrosis and tumorigenesis". Cell Reports. Medicine. 3 (11): 100820. doi:10.1016/j.xcrm.2022.100820. PMC 9729881. PMID 36384103. S2CID 253577148.
- "Leprosy: Ancient disease able to regenerate organs". BBC News. 16 November 2022. Archived from the original on 18 November 2022. Retrieved 18 November 2022.
Further reading
- Pam Fessler (2020). Carville's Cure: Leprosy, Stigma, and the Fight for Justice. Liveright. ISBN 978-1631495038.
.png.webp)