Lithium metal battery
Lithium metal batteries are primary batteries that have metallic lithium as an anode. These types of batteries are also referred to as lithium-metal batteries after lithium-ion batteries had been invented. Most lithium metal batteries are non-rechargeable. However, rechargeable lithium metal batteries are also under development. Since 2007, Dangerous Goods Regulations differentiate between lithium metal batteries (UN 3090) and lithium-ion batteries (UN 3480).[1]

They stand apart from other batteries in their high charge density and high cost per unit. Depending on the design and chemical compounds used, lithium cells can produce voltages from 1.5 V (comparable to a zinc–carbon or alkaline battery) to about 3.7 V.
Disposable primary lithium batteries must be distinguished from secondary lithium-ion or a lithium-polymer,[2] which are rechargeable batteries and contain no metallic lithium. Lithium is especially useful, because its ions can be arranged to move between the anode and the cathode, using an intercalated lithium compound as the cathode material but without using lithium metal as the anode material. Pure lithium will instantly react with water, or even moisture in the air; the lithium in lithium-ion batteries is a less reactive compound.
Lithium batteries are widely used in portable consumer electronic devices. The term "lithium battery" refers to a family of different lithium-metal chemistries, comprising many types of cathodes and electrolytes but all with metallic lithium as the anode. The battery requires from 0.15 to 0.3 kg of lithium per kWh. As designed these primary systems use a charged cathode, that being an electro-active material with crystallographic vacancies that are filled gradually during discharge.
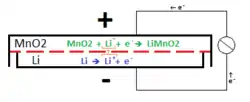
The most common type of lithium cell used in consumer applications uses metallic lithium as the anode and manganese dioxide as the cathode, with a salt of lithium dissolved in an organic solvent as the electrolyte.[3]
History


Lithium is the metal with lowest density and with the greatest electrochemical potential and energy-to-weight ratio. The low atomic weight and small size of its ions also speeds its diffusion, suggesting that it would make an ideal material for batteries.[4] Experimentation with lithium batteries began in 1912 under American physical chemist Gilbert N. Lewis, but commercial lithium batteries did not come to market until the 1970s in the form of the lithium-ion battery.[5][6] Three volt lithium primary cells such as the CR123A type and three volt button cells are still widely used, especially in cameras and very small devices.
Three important developments regarding lithium batteries occurred in the 1980s. In 1980, an American chemist, John B. Goodenough, discovered the LiCoO2 (Lithium cobalt oxide) cathode (positive lead) and a Moroccan research scientist, Rachid Yazami, discovered the graphite anode (negative lead) with the solid electrolyte. In 1981, Japanese chemists Tokio Yamabe and Shizukuni Yata discovered a novel nano-carbonacious-PAS (polyacene)[7] and found that it was very effective for the anode in the conventional liquid electrolyte.[8][9] This led a research team managed by Akira Yoshino of Asahi Chemical, Japan, to build the first lithium-ion battery prototype in 1985, a rechargeable and more stable version of the lithium battery; Sony commercialized the lithium-ion battery in 1991.[10]
In 1997, the lithium polymer battery was released by Sony and Asahi Kasei. These batteries hold their electrolyte in a solid polymer composite instead of in a liquid solvent, and the electrodes and separators are laminated to each other. The latter difference allows the battery to be encased in a flexible wrapping instead of in a rigid metal casing, which means such batteries can be specifically shaped to fit a particular device. This advantage has favored lithium polymer batteries in the design of portable electronic devices such as mobile phones and personal digital assistants, and of radio-controlled aircraft, as such batteries allow for a more flexible and compact design. They generally have a lower energy density than normal lithium-ion batteries.
In 2019, John B. Goodenough, M. Stanley Whittingham, and Akira Yoshino, were awarded the Nobel Prize in Chemistry, for their development of lithium-ion batteries.[11]Chemistries
| Chemistry | Cathode | Electrolyte | Nominal voltage |
Open-circuit voltage |
Wh/kg | Wh/L |
|---|---|---|---|---|---|---|
| Li–MnO2 (IEC code: C), "CR" |
Heat-treated manganese dioxide | Lithium perchlorate in an organic solvent (propylene carbonate and dimethoxyethane in many common cells[12][13][14]) | 3 V | 3.3 V | 280 | 580 |
| "Li–Mn". The most common consumer-grade lithium battery, about 80% of the lithium battery market. Uses inexpensive materials. Suitable for low-drain, long-life, low-cost applications. High energy density per both mass and volume. Operational temperature ranges from -30 °C to 60 °C. Can deliver high pulse currents.[15] With discharge, the internal impedance rises and the terminal voltage decreases. High self-discharge at high temperatures. 1,2-Dimethoxyethane is a REACH candidate substance of very high concern. | ||||||
| Li–(CF)x (IEC code: B), "BR" |
Carbon monofluoride | Lithium tetrafluoroborate in propylene carbonate, dimethoxyethane, or gamma-butyrolactone | 3 V | 3.1 V | 360–500 | 1,000 |
| Cathode material formed by high-temperature intercalation of fluorine gas into graphite powder. Compared to manganese dioxide (CR), which has the same nominal voltage, it provides more reliability.[15] Used for low to moderate current applications in memory and clock backup batteries. Used in aerospace applications, qualified for space since 1976, military applications both terrestrial and marine, in missiles, and in artificial cardiac pacemakers.[16] Operates up to around 80 °C. Very low self-discharge (<0.5%/year at 60 °C, <1%/yr at 85 °C). Developed in the 1970s by Matsushita.[17] | ||||||
| Li–FeS2 (IEC code: F), "FR" |
Iron disulfide | Propylene carbonate, dioxolane, dimethoxyethane | 1.4–1.6 V | 1.8 V | 297 |
|
| "Lithium-iron", "Li/Fe". Called "voltage-compatible" lithium, it can work as a replacement for alkaline batteries with its 1.5 V nominal voltage. As such, Energizer lithium cells of AA[19] and AAA size employ this chemistry. 2.5 times higher lifetime for high current discharge regime than alkaline batteries, better storage life due to lower self-discharge(10–20 years). FeS2 is cheap. Cathode is often designed as a paste of iron sulfide powder mixed with powdered graphite. Variant is Li–CuFeS2. | ||||||
| Li–SOCl2 (IEC code: E) |
Thionyl chloride | Lithium tetrachloroaluminate in thionyl chloride | 3.5 V | 3.65 V | 500–700 | 1,200 |
| Liquid cathode. For low-temperature applications. Can operate down to −55 °C, where it retains over 50% of its rated capacity. A negligible amount of gas generated in nominal use, a limited amount under abuse. Has relatively high internal impedance and limited short-circuit current. High energy density, about 500 Wh/kg. Toxic. Electrolyte reacts with water. Low-current cells are used for portable electronics and memory backup. High-current cells are used in military applications. In long storage, forms passivation layer on the anode, which may lead to temporary voltage delay when put into service. High cost and safety concerns limit use in civilian applications. Can explode when shorted. Underwriters Laboratories require a trained technician for the replacement of these batteries. Hazardous waste, Class 9 Hazmat shipment.[20] Not used for consumer or general-purpose batteries. | ||||||
| Li–SOCl2,BrCl, Li–BCX (IEC code: E) |
Thionyl chloride with bromine chloride | Lithium tetrachloroaluminate in thionyl chloride | 3.7–3.8 V | 3.9 V | 350 | 770 |
| Liquid cathode. A variant of the thionyl chloride battery, with 300 mV higher voltage. The higher voltage drops back to 3.5 V soon as the bromine chloride gets consumed during the first 10–20% of the discharge. The cells with added bromine chloride are thought to be safer when abused. | ||||||
| Li–SO2Cl2 (IEC code: Y) |
Sulfuryl chloride | Lithium tetrachloroaluminate in sulfuryl chloride | 3.7 V | 3.95 V | 330 | 720 |
| Liquid cathode. Similar to thionyl chloride. Discharge does not result in a build-up of elemental sulfur, which is thought to be involved in some hazardous reactions, therefore sulfuryl chloride batteries may be safer. Commercial deployment is hindered by the tendency of the electrolyte to corrode the lithium anodes, reducing the shelf life. Chlorine is added to some cells to make them more resistant to abuse. Sulfuryl chloride cells give less maximum current than thionyl chloride ones, due to polarization of the carbon cathode. Sulfuryl chloride reacts violently with water, releasing hydrogen chloride and sulfuric acid.[21] | ||||||
| Li–SO2 (IEC code: W) |
Sulfur dioxide on teflon-bonded carbon | Lithium bromide in sulfur dioxide with small amount of acetonitrile | 2.85 V | 3.0 V | 250 | 400 |
| Liquid cathode. Can operate down to −55 °C and up to +70 °C. Contains liquid SO2 at high pressure. Requires safety vent, can explode in some conditions. High energy density. High cost. At low temperatures and high currents, performs better than Li–MnO2. Toxic. Acetonitrile forms lithium cyanide, and can form hydrogen cyanide in high temperatures.[22] Used in military applications. Addition of bromine monochloride can boost the voltage to 3.9 V and increase energy density.[23] | ||||||
| Li–I2 | Iodine that has been mixed and heated with poly-2-vinylpyridine (P2VP) to form a solid organic charge transfer complex. | A solid monomolecular layer of crystalline Lithium iodide that conducts lithium ions from the anode to the cathode but does not conduct Iodine.[24] | 2.8 V | 3.1 V | ||
| Solid electrolyte. Very high reliability and low self-discharge rate. Used in medical applications that need a long life, e.g. pacemakers. Does not generate gas even under a short circuit. Solid-state chemistry, limited short-circuit current, suitable only for low-current applications. Terminal voltage decreases with the degree of discharge due to precipitation of lithium iodide. | ||||||
| Li–Ag2CrO4 | Silver chromate | Lithium perchlorate solution | 3.1/2.6 V | 3.45 V | ||
| Very high reliability. Has a 2.6 V plateau after reaching a certain percentage of discharge provides early warning of impending discharge. Developed specifically for medical applications, for example, implanted pacemakers. | ||||||
| Li–Ag2V4O11, Li–SVO, Li–CSVO |
Silver oxide+vanadium pentoxide (SVO) | lithium hexafluorophosphate or lithium hexafluoroarsenate in propylene carbonate with dimethoxyethane | ||||
| Used in medical applications, like implantable defibrillators, neurostimulators, and drug infusion systems. Also projected for use in other electronics, such as emergency locator transmitters. High energy density. Long shelf life. Capable of continuous operation at a nominal temperature of 37 °C.[25] Two-stage discharge with a plateau. Output voltage decreases proportionally to the degree of discharge. Resistant to abuse. | ||||||
| Li–CuO (IEC code: G), "GR" |
Copper(II) oxide | Lithium perchlorate dissolved in dioxolane | 1.5 V | 2.4 V | ||
| Can operate up to 150 °C. Developed as a replacement of zinc–carbon and alkaline batteries. "Voltage up" problem, the high difference between open-circuit and nominal voltage. Produced until the mid-1990s, replaced by lithium–iron sulfide. Current use is limited. | ||||||
| Li–Cu4O(PO4)2 | Copper oxyphosphate | |||||
| See Li–CuO | ||||||
| Li–CuS | Copper sulfide | Lithium salt or a salt such as a tetraalkylammonium chloride dissolved in LiClO4 in an organic solvent that is a mixture of 1,2-dimethoxy ethane, 1,3-dioxolane and 2,5-dimethyloxazole as a stabilizer[26] | 1.5 V | |||
| Li–PbCuS | Lead sulfide and copper sulfide | 1.5 V | 2.2 V | |||
| Li–FeS | Iron sulfide | Propylene carbonate, dioxolane, dimethoxyethane | 1.5–1.2 V | |||
| "Lithium-iron", "Li/Fe". used as a replacement for alkaline batteries. See lithium-iron disulfide. | ||||||
| Li–Bi2Pb2O5 | Lead bismuthate | 1.5 V | 1.8 V | |||
| Replacement of silver-oxide batteries, with higher energy density, lower tendency to leak, and better performance at higher temperatures. | ||||||
| Li–Bi2O3 | Bismuth trioxide | 1.5 V | 2.04 V | |||
| Li–V2O5 | Vanadium pentoxide | 3.3/2.4 V | 3.4 V | 120/260 | 300/660 | |
| Two discharge plateaus. Low-pressure. Rechargeable. Used in reserve batteries. | ||||||
| Li–CuCl2 | Copper chloride | LiAlCl4 or LiGaCl4 in SO2, a liquid, inorganic, non-aqueous electrolyte. | ||||
| Rechargeable. This cell has three voltage plateaus as it discharges (3.3 V, 2.9 V, and 2.5 V).[27] Discharging below the first plateau reduces the life of the cell.[27] The complex salt dissolved in SO2 has a lower vapor pressure at room temperature than pure sulfur dioxide,[28] making the construction simpler and safer than Li–SO2 batteries. | ||||||
| Li/Al–MnO2, "ML" | Manganese dioxide | 3 V |
||||
| Rechargeable. Anode is a Lithium-Aluminum alloy.[29][30] Mainly marketed by Maxell. | ||||||
| Li/Al–V2O5, "VL" | Vanadium pentoxide | 3 V |
||||
| Rechargeable. Anode is a Li–Al alloy.[32] | ||||||
| Li–Se | Selenium | non-aqueous carbonate electrolytes | 1.9 V |
|||
| Li–air | Porous carbon | Organic, aqueous, glass–ceramic (polymer–ceramic composites) | 1,800–660 |
1,600–600 | ||
| Rechargeable. As of 2012, no commercial implementation is available due to difficulties in achieving multiple discharge cycles without losing capacity.[34] There are multiple possible implementations, each having different energy capacities, advantages and disadvantages. In November 2015, a team of University of Cambridge researchers furthered work on lithium-air batteries by developing a charging process capable of prolonging the battery life and battery efficiency. Their work resulted in a battery that delivered high energy densities, more than 90% efficiency, and could be recharged for up to 2,000 times. The lithium-air batteries are described as the "ultimate" batteries because they propose a high theoretical energy density of up to ten times the energy offered by regular lithium-ion batteries. They were first developed in a research environment by Abraham & Jiang in 1996.[35] Toyota promoted a vehicle with a working solid-state battery in September 2021; due to cost, the company plans to use it in a hybrid vehicle in 2025, before scaling up to fully electric vehicles.[36] Other companies working on commercialization include QuantumScape and Solid Power (funded by Ford Motor Company and BMW).[36] | ||||||
| Li–FePO4 | Lithium iron phosphate | ethylene carbonate–dimethyl carbonate (EC–DMC) 1–1 lithium perchlorate (LiClO 4) 1M |
3.0 ~ 3.2 V | 3.2 V | 90–160 |
325 Wh/L (1,200 kJ/L) |
| The specific capacity of LiFePO 4 is higher than that of the related lithium cobalt oxide (LiCoO 2) chemistry, but its energy density is less due to its lower operating voltage. The main drawback of LiFePO 4 is its low electrical conductivity. Because of low cost, low toxicity, well-defined performance, long-term stability, etc. LiFePO 4 is finding a number of roles in vehicle use, utility scale stationary applications, and backup power. | ||||||
University of California San Diego have developed an electrolyte chemistry that allows lithium batteries to run at temperatures as low as -60 °C. The electrolytes also enable electrochemical capacitors to run as low as -80 °C. Previous low-temperature limit is -40 °C. High performance at room temperature is still maintained. This may improve energy density and safety of lithium batteries and electrochemical capacitors.[39]
Applications
Lithium batteries find application in many long-life, critical devices, such as pacemakers and other implantable electronic medical devices. These devices use specialized lithium-iodide batteries designed to last 15 or more years. But for other, less critical applications such as in toys, the lithium battery may actually outlast the device. In such cases, an expensive lithium battery may not be cost-effective.
Lithium batteries can be used in place of ordinary alkaline cells in many devices, such as clocks and cameras. Although they are more costly, lithium cells will provide much longer life, thereby minimizing battery replacement. However, attention must be given to the higher voltage developed by the lithium cells before using them as a drop-in replacement in devices that normally use ordinary zinc cells.

Lithium batteries also prove valuable in oceanographic applications. While lithium battery packs are considerably more expensive than standard oceanographic packs, they hold up to three times the capacity of alkaline packs. The high cost of servicing remote oceanographic instrumentation (usually by ships) often justifies this higher cost.
Sizes and formats
Small lithium batteries are very commonly used in small, portable electronic devices, such as PDAs, watches, camcorders, digital cameras, thermometers, calculators, personal computer BIOS (firmware),[40] communication equipment and remote car locks. They are available in many shapes and sizes, with a common variety being the 3-volt "coin" type manganese variety. The common CR2032 battery is 20 mm diameter and 3.2 mm thick, where the first two digits is the diameter and the last two digits are thickness. A CR2025 is the same 20 mm diameter but 2.5 mm thick.
The heavy electrical demands of many of these devices make lithium batteries a particularly attractive option. In particular, lithium batteries can easily support the brief, heavy current demands of devices such as digital cameras, and they maintain a higher voltage for a longer period than alkaline cells.
Safety issues and regulation
The computer industry's drive to increase battery capacity can test the limits of sensitive components such as the membrane separator, a polyethylene or polypropylene film that is only 20–25 μm thick. The energy density of lithium batteries has more than doubled since they were introduced in 1991. When the battery is made to contain more material, the separator can undergo stress.
Rapid-discharge problems
Lithium batteries can provide extremely high currents and can discharge very rapidly when short-circuited. Although this is useful in applications where high currents are required, a too-rapid discharge of a lithium battery – especially if cobalt is present in the cells' design – can result in overheating of the battery (that lowers the electrical resistance of any cobalt content within the cell), rupture, and even an explosion. Lithium-thionyl chloride batteries are particularly susceptible to this type of discharge. Consumer batteries usually incorporate overcurrent or thermal protection or vents to prevent an explosion.
Air travel
From January 1 2013, much stricter regulations were introduced by IATA regarding the carriage of lithium batteries by air. They were adopted by the International Postal Union; however, some countries, e.g. the UK, have decided that they will not accept lithium batteries unless they are included with the equipment they power.
Because of the above risks, shipping and carriage of lithium batteries are restricted in some situations, particularly the transport of lithium batteries by air.
The United States Transportation Security Administration announced restrictions effective January 1, 2008, on lithium batteries in checked and carry-on luggage. The rules forbid lithium batteries not installed in a device from checked luggage and restrict them in carry-on luggage by total lithium content.[45]
Australia Post prohibited transport of lithium batteries in air mail during 2010.[46]
UK regulations for the transport of lithium batteries were amended by the National Chemical Emergency Centre in 2009.[47]
In late 2009, at least some postal administrations restricted airmail shipping (including Express Mail Service) of lithium batteries, lithium-ion batteries and products containing these (such as laptops and cell phones). Among these countries are Hong Kong, United States, and Japan.[48][49][50]
Methamphetamine labs
Unused lithium batteries provide a convenient source of lithium metal for use as a reducing agent in methamphetamine labs. Specifically, lithium metal reduces pseudoephedrine and ephedrine to methamphetamine in the Birch reduction method, which employs solutions of alkali metals dissolved in anhydrous ammonia.[51][52]
Some jurisdictions have passed laws to restrict lithium battery sales or asked businesses to make voluntary restrictions in an attempt to help curb the creation of illegal meth labs. In 2004 Wal-Mart stores were reported to limit the sale of disposable lithium batteries to three packages in Missouri and four packages in other states.[53]
Health issues on ingestion
Button cell batteries are attractive to small children and are often ingested. In the past 20 years, although there has not been an increase in the total number of button cell batteries ingested in a year, researchers have noted a 6.7-fold increase in the risk that an ingestion would result in a moderate or major complication and 12.5-fold increase in fatalities comparing the last decade to the previous one.[54][55]

The primary mechanism of injury with button battery ingestions is the generation of hydroxide ions, which cause severe chemical burns, at the anode.[57] This is an electrochemical effect of the intact battery, and does not require the casing to be breached or the contents released.[57] Complications include oesophageal strictures, tracheo-oesophageal fistulas, vocal cord paralysis, aorto-oesophageal fistulas, and death.[58] The majority of ingestions are not witnessed; presentations are non-specific; battery voltage has increased; the 20 to 25 mm button battery size are more likely to become lodged at the cricopharyngeal junction; and severe tissue damage can occur within 2 hours. The 3 V, 20 mm CR2032 lithium battery has been implicated in many of the complications from button battery ingestions by children of less than 4 years of age.[59]
While the only cure for an esophageal impaction is endoscopic removal, a 2018 study out of Children's Hospital of Philadelphia by Rachel R. Anfang and colleagues found that early and frequent ingestion of honey or sucralfate suspension prior to the battery's removal can reduce the injury severity to a significant degree.[55] As a result, US-based National Capital Poison Center (Poison Control) recommends the use of honey or sucralfate after known or suspected ingestions to reduce the risk and severity of injury to esophagus, and consequently its nearby structures.[60]
Button batteries can also cause significant necrotic injury when stuck in the nose or ears.[61] Prevention efforts in the US by the National Button Battery Task force in cooperation with industry leaders have led to changes in packaging and battery compartment design in electronic devices to reduce a child's access to these batteries.[62] However, there still is a lack of awareness across the general population and medical community to its dangers. Central Manchester University Hospital Trust warns that "a lot of doctors are unaware that this can cause harm".[63]
Disposal
Regulations for disposal and recycling of batteries vary widely; local governments may have additional requirements over those of national regulations. In the United States, one manufacturer of lithium iron disulfide primary batteries advises that consumer quantities of used cells may be discarded in municipal waste, as the battery does not contain any substances controlled by US Federal regulations.[64] Most lithium batteries, are, however, classified as hazardous waste due to the possibility of fire. Another manufacturer states that while "button" size lithium batteries contain perchlorate, which is regulated as a hazardous waste in California; regulated quantities would not be found in typical consumer use of these cells.[65]
The EPA, however, states that due to limited supply and increasingly high importance, lithium batteries should always be recycled if possible.[66] As well, a rupture in a battery poses a potential fire hazard, thus the EPA states that the average consumer should take lithium batteries to specialized lithium or hazardous materials facilities.
As lithium in used but non-working (i.e. extended storage) button cells is still likely to be in the cathode cup, it is possible to extract commercially useful quantities of the metal from such cells as well as the manganese dioxide and specialist plastics. Some also alloy the lithium with magnesium (Mg) to cut costs.
Since there has been an exponential increase in demand for lithium batteries over time,[67] there have been pushes to find better ways to recycle lithium batteries.[68]
Rechargeable batteries
See also
- List of battery types
- List of battery sizes
- Comparison of battery types
- Battery holder
- Battery recycling
- High capacity oceanographic lithium battery pack
- Lithium–air battery
- Lithium ion manganese oxide battery
- Lithium ion polymer battery
- Lithium iron phosphate battery
- Lithium–sulfur battery
- Lithium-titanate battery
- Nanoarchitectures for lithium-ion batteries
- Polyoxyethylene
- Thin film rechargeable lithium battery
References
- Committee of Experts on the Transport of Dangerous Goods and on the Globally Harmonized System of Classification and Labelling of Chemicals, ed. (2006-12-14). "ST/SG/AC.10/34/Add.1 - Report of the Committee of Experts on its Third Session, Addendum, Annex 1, Amendments to the fourteenth revised edition of the Recommendations on the Transport of Dangerous Goods, Model Regulations" (PDF). Geneva: United Nations. Retrieved 2021-05-13.
- Batscap - La batterie lithium métal polymère Archived 2012-08-08 at the Wayback Machine in batscap.com
- "One Man and His Whippet". Eco Tree Lithium. Retrieved 2022-02-19.
- Winter, Martin; Barnett, Brian; Xu, Kang (30 November 2018). "Before Li Ion Batteries". Chemical Reviews. 118 (23): 11433–11456. doi:10.1021/acs.chemrev.8b00422. PMID 30500179. S2CID 54615265.
- Scrosati, Bruno (4 May 2011). "History of lithium batteries". Journal of Solid State Electrochemistry. 15 (7–8): 1623–1630. doi:10.1007/s10008-011-1386-8. S2CID 98385210.
- Vincent, C (1 October 2000). "Lithium batteries: a 50-year perspective, 1959–2009". Solid State Ionics. 134 (1–2): 159–167. doi:10.1016/S0167-2738(00)00723-2.
- Yamabe, T.; Tanaka, K.; Ohzeki, K.; Yata, S. (1982). "Electronic structure of polyacenacene. A one-dimensional graphite". Solid State Communications. Elsevier BV. 44 (6): 823–825. Bibcode:1982SSCom..44..823Y. doi:10.1016/0038-1098(82)90282-4. ISSN 0038-1098.
- S. Yata, U.S. Patent #4,601,849
- Yata, Shjzukuni; Tanaka, Kazuyoshi; Yamabe, Tokio (1997). "Polyacene (PAS) Batteries". MRS Proceedings. Cambridge University Press (CUP). 496. doi:10.1557/proc-496-15. ISSN 1946-4274.
- Novák, Petr; Müller, Klaus; Santhanam, K. S. V.; Haas, Otto (1997). "Electrochemically Active Polymers for Rechargeable Batteries". Chemical Reviews. American Chemical Society (ACS). 97 (1): 272. doi:10.1021/cr941181o. ISSN 0009-2665. PMID 11848869.
- "The Nobel Prize in Chemistry 2019". NobelPrize.org. Retrieved 2019-10-28.
- "Duracell Primary Lithium Coin Cell Article Information Sheet" (PDF). 2015-07-01. Archived from the original (PDF) on 2018-01-03. Retrieved 2018-01-02.
- "Energizer Product Safety Data Sheet, Coin/Button Lithium Manganese Dioxide Batteries" (PDF). 2017-01-01. Archived from the original (PDF) on 2017-09-08. Retrieved 2018-01-02.
- "Material Safety Data Sheet, Li-Mn Button Cell CR2025" (PDF). 2016-01-01. Archived from the original (PDF) on 2018-01-03. Retrieved 2018-01-02.
- "Electronic Components - Panasonic Industrial Devices". www.panasonic.com. Archived from the original on 2013-07-02.
- Greatbatch W, Holmes CF, Takeuchi ES, Ebel SJ (November 1996). "Lithium/carbon monofluoride (Li/CFx): a new pacemaker battery". Pacing Clin Electrophysiol. 19 (11 Pt 2): 1836–40. doi:10.1111/j.1540-8159.1996.tb03236.x. PMID 8945052. S2CID 11180448.
- "Lithium Poly Carbon Monoflouride". House Of Batteries. Archived from the original on 2007-09-29. Retrieved 2008-02-19.
- "Cylindrical Primary Lithium – Handbook and Application Manual" (PDF). data.energizer.com.
- "Product Datasheet – Energizer L91" (PDF). data.energizer.com. Archived from the original (PDF) on 2015-12-04. Retrieved 2015-10-21.
- Pilarzyk, Jim. "White Paper - Lithium Carbon Monofluoride Coin Cells in Real-Time Clock and Memory Backup Applications". rayovac.com. Rayovac Corporation. Archived from the original on 2007-12-12.
- "Lithium sulfuryl chloride battery". Corrosion-doctors.org. Archived from the original on 2010-11-21. Retrieved 2011-01-19.
- McGraw, Jack (March 7, 1984). "Letter to Dick Bruner, U.S. Defense Logistics Agency". U.S. Environmental Protection Agency. Archived from the original on March 4, 2012.
- "Lithium Batteries Specifications". Lithium-batteries.globalspec.com. Archived from the original on 2007-01-28. Retrieved 2011-01-19.
- Mallela, V. S.; Ilankumaran, V.; Rao, N. S. (2004). "Trends in cardiac pacemaker batteries". Indian Pacing and Electrophysiology Journal. 4 (4): 201–212. PMC 1502062. PMID 16943934.
- Gonzalez, Lina (Summer 2005). "Solid State NMR Investigation of Silver Vanadium Oxide (SVO)". CUNY, Hunter College. Archived from the original on 2006-09-10.
- Engineering Chemistry by RV Gadag and Narayan Shetty ISBN 8188237833
- McDonald, R. C.; Harris, P.; Hossain, S.; Goebel, F. (1992). "Analysis of secondary lithium cells with sulfur dioxide based electrolytes". IEEE 35th International Power Sources Symposium. p. 246. doi:10.1109/IPSS.1992.282033. ISBN 978-0-7803-0552-6. S2CID 98323962.
- US patent 4891281, Kuo, Han C. & Foster, Donald L., "Electrochemical cells having low vapor pressure complexed SO2 electrolytes", issued 01-02-1990, assigned to Duracell Inc.
- "Electronic Components - Panasonic Industrial Devices". www.panasonic.com. Archived from the original on 2013-11-13.
- "Data Sheet: ML2032" (PDF). Maxell. Archived from the original (PDF) on 2018-09-10. Retrieved 10 September 2018.
- "Electronic Components - Panasonic Industrial Devices". www.panasonic.com. Archived from the original on 2013-11-25.
- "Product Safety Data Sheet (VL Series)" (PDF). Panasonic. Retrieved 10 September 2018.
- Eftekhari, Ali (2017). "The rise of lithium–selenium batteries". Sustainable Energy & Fuels. 1: 14–29. doi:10.1039/C6SE00094K.
- Christensen, J.; Albertus, P.; Sanchez-Carrera, R. S.; Lohmann, T.; Kozinsky, B.; Liedtke, R.; Ahmed, J.; Kojic, A. (2012). "A Critical Review of Li/Air Batteries". Journal of the Electrochemical Society. 159 (2): R1. doi:10.1149/2.086202jes.
- Abraham, K. M. (1996). "A Polymer Electrolyte-Based Rechargeable Lithium/Oxygen Battery". Journal of the Electrochemical Society. 143 (1): 1–5. Bibcode:1996JElS..143....1A. doi:10.1149/1.1836378. ISSN 0013-4651. S2CID 96810495.
- Verma, Pranshu (18 May 2022). "Inside the race for a car battery that charges fast — and won't catch fire". The Washington Post.
- "Large-Format, Lithium Iron Phosphate". JCWinnie.biz. 2008-02-23. Archived from the original on 2008-11-18. Retrieved 2012-04-24.
- "Great Power Group, Square Lithium-Ion Battery". Archived from the original on 2020-08-03. Retrieved 2019-12-31.
- "Lithium batteries to run at ultra-low temperatures". WorldOfChemicals. October 9, 2017. Archived from the original on October 10, 2017. Retrieved October 10, 2017.
- Torres, Gabriel (24 November 2004). "Introduction and Lithium Battery". Replacing the Motherboard Battery. hardwaresecrets.com. Archived from the original on 24 December 2013. Retrieved June 20, 2013.
- "BAJ Website | Monthly battery sales statistics". Baj.or.jp. Archived from the original on 2010-12-06. Retrieved 2013-06-12.
- "INOBAT 2008 statistics" (PDF). Archived from the original (PDF) on March 25, 2012.
- "Battery Waste Management - 2006 DEFRA" (PDF). Archived from the original (PDF) on 2013-10-08.
- "Battery Statistics". EPBAEurope.net. European Portable Battery Association. 2000. Archived from the original on 2012-03-21. Retrieved 2015-07-28.
- "Traveling Safe with Batteries". U.S. Department of Transportation. Archived from the original on 2007-12-30. Retrieved 2007-12-29.
- "Customer guidelines for posting lithium batteries" (PDF). AusPost.com.au. Archived from the original (PDF) on 2012-07-06. Retrieved 2012-08-15.
- "Lithium Battery Transport Regulation". The-NCEC.com. Archived from the original on 2013-01-29. Retrieved 2013-04-03.
- "Postage Guide - section 6.3" (PDF). Hong Kong Post. Archived from the original (PDF) on 2014-05-01.
- "349 Miscellaneous Hazardous Materials (Hazard Class 9)". Publication 52 - Hazardous, Restricted, and Perishable Mail. United States Postal Service. February 2015. Archived from the original on 2015-07-29. Retrieved 2015-07-25.
- "I want to send a laptop to overseas. How can I do that ?". Post.JapanPost.jp. Archived from the original on 2011-04-26. Retrieved 2011-01-19.
- "Illinois Attorney General – Basic Understanding of Meth". Illinoisattorneygeneral.gov. Archived from the original on 10 September 2010. Retrieved 6 October 2010.
- Harmon, Aaron R. (2006). "Methamphetamine remediation research act of 2005: Just what the doctor ordered for cleaning up methfields—or sugar pill placebo?" (PDF). North Carolina Journal of Law & Technology. 7. Archived from the original (PDF) on 2008-12-01. Retrieved 5 October 2010.
- Parker, Molly (January 26, 2004). "Meth fear cuts cold-pill access ; Pseudoephedrine used in illegal drug". Chicago Tribune. p. 1. Archived from the original on November 5, 2012.(registration required)
- Litovitz, Toby; Whitaker, N; Clark, L; White, NC; Marsolek, M (June 2010). "Emerging battery-ingestion hazard: clinical implications". Pediatrics. 125 (6): 1168–77. doi:10.1542/peds.2009-3037. PMID 20498173. Archived from the original on 6 October 2017. Retrieved 11 June 2011.
- Anfang, Rachel R.; Jatana, Kris R.; Linn, Rebecca L.; Rhoades, Keith; Fry, Jared; Jacobs, Ian N. (2018-06-11). "pH-neutralizing esophageal irrigations as a novel mitigation strategy for button battery injury". The Laryngoscope. 129 (1): 49–57. doi:10.1002/lary.27312. ISSN 0023-852X. PMID 29889306. S2CID 47004940.
- IEC (ed.). "Clause 9: Marking and packaging". IEC 60086-4:2019 Primary batteries - Part 4: Safety of lithium batteries (PDF). Geneva: IEC. ISBN 978-2-8322-6808-7.
- Jatana, Kris R.; Rhoades, Keith; Milkovich, Scott; Jacobs, Ian N. (2016-11-09). "Basic mechanism of button battery ingestion injuries and novel mitigation strategies after diagnosis and removal". The Laryngoscope. 127 (6): 1276–1282. doi:10.1002/lary.26362. ISSN 0023-852X. PMID 27859311. S2CID 1335692.
- "Parents warned after girl's battery death". Brisbane Times. AAP. July 2, 2013. Archived from the original on July 4, 2013. Retrieved July 2, 2013.
- Litovitz, Toby; Whitaker N; Clark L. (June 2010). "Preventing battery ingestions: an analysis of 8648 cases". Pediatrics. 125 (6): 1178–83. doi:10.1542/peds.2009-3038. PMID 20498172. Archived from the original on 27 May 2014. Retrieved 11 June 2011.
- "Guideline". www.poison.org. Retrieved 2018-07-06.
- Mack, Sharon Kiley, "Tiny lithium battery nearly kills Deer Isle toddler" Archived 2011-08-03 at Wikiwix, Bangor Daily News, July 24, 2011 3:41 pm. Retrieved 2 August 2011
- Jatana, Kris R.; Litovitz, Toby; Reilly, James S.; Koltai, Peter J.; Rider, Gene; Jacobs, Ian N. (2013-09-01). "Pediatric button battery injuries: 2013 task force update". International Journal of Pediatric Otorhinolaryngology. 77 (9): 1392–1399. doi:10.1016/j.ijporl.2013.06.006. ISSN 0165-5876. PMID 23896385.
- "Battery warning after child deaths". BBC News. 2014-10-14. Retrieved 2018-07-06.
- Disposal of Energizer AA and AAA Lithium L92 and L92 Battery Lithium/Iron Disulfide Archived 2013-11-09 at the Wayback Machine, retrieved 2012 Aug 20
- "Electronic Components - Panasonic Industrial Devices". www.panasonic.com. Archived from the original on 2012-08-20. Retrieved 2012-08-20.
- US EPA, OLEM (2019-05-16). "Used Lithium-Ion Batteries". www.epa.gov. Retrieved 2023-09-01.
- "Trends in batteries – Global EV Outlook 2023 – Analysis". IEA. Retrieved 2023-09-01.
- Research, Allied Market. "Lithium-ion Battery Recycling Market to Reach $38.21 Bn, Globally, by 2030 at 36.0% CAGR: Allied Market Research". www.prnewswire.com. Retrieved 2023-09-01.
- Xu, Wu; Wang, Jiulin; Ding, Fei; Chen, Xilin; Nasybulin, Eduard; Zhang, Yaohui; Zhang, Ji-Guang (2014). "Lithium metal anodes for rechargeable batteries". Energy Environ. Sci. 7 (2): 513–537. doi:10.1039/C3EE40795K. ISSN 1754-5692.
- Albertus, Paul; Babinec, Susan; Litzelman, Scott; Newman, Aron (2018). "Status and challenges in enabling the lithium metal electrode for high-energy and low-cost rechargeable batteries". Nature Energy. 3: 16–21. Bibcode:2018NatEn...3...16A. doi:10.1038/s41560-017-0047-2. S2CID 139241677. Retrieved 2021-02-13.
External links
- The 2009 amendments to the regulations regarding transport of Lithium Batteries
- Properties of non-rechargeable lithium batteries
- Brand Neutral Drawings of Lithium Batteries based on ANSI Specifications
- Lithium Thionyl Chloride Battery MSDS and supporting safety information
- Investigation of the fire performance of lithium-ion- and lithium-metal-batteries in various applications and derivative of tactical recommendations (Research Report in German, Forschungsstelle für Brandschutztechnik, Karlsruhe Institute of Technology - KIT) (PDF)
- Intercalation Station (February 9, 2023). "Revival of the lithium metal battery". intercalationstation.substack.com. Retrieved 2023-02-09.

