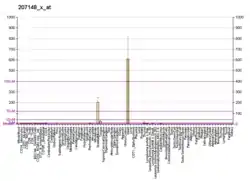
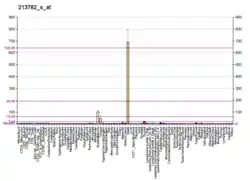
MYOZ2
Myozenin-2, also referred to as Calsarcin-1, is a protein that in humans is encoded by the MYOZ2 gene.[5][6][7] The Calsarcin-1 isoform is a muscle protein expressed in cardiac muscle and slow-twitch skeletal muscle, which functions to tether calcineurin to alpha-actinin at Z-discs, and inhibit the pathological cardiac hypertrophic response. This differs from the fast-skeletal muscle isoform, calsarcin-2.
| MYOZ2 | |||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Aliases | MYOZ2, C4orf5, CMH16, CS-1, myozenin 2, FATZ-2 | ||||||||||||||||||||||||||||||||||||||||||||||||||
| External IDs | OMIM: 605602 MGI: 1913063 HomoloGene: 9583 GeneCards: MYOZ2 | ||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikidata | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
Structure
Calsarcin-1 is a 29.9 kDa protein composed of 264 amino acids.[8][9] Calsarcin-1 and calsarcin-2 are only 31% homologous (94 identical amino acids), exhibiting the highest homology at N- and C-termini. Calsarcin-1 binds to alpha-actinin,[10] gamma-filamin,[11] telethonin,[11] ZASP/Cypher[11] and calcineurin.[10] The binding region of calsarcin-1 to alpha-actinin is localized to amino acids 153-200, and that of calsarcin-1 to calcineurin is amino acids 217-240.[10]
Function
The function of calsarcin-1 in cardiac and slow-skeletal muscle has been illuminated through studies in transgenic animals. Mice lacking the MYOZ2 gene (MYOZ2-/-) are generally sensitized to calcineurin signaling in both muscle types.[11] In slow-skeletal muscle, MYOZ2-/- show increased slow-twitch muscle fibers. In cardiac, MYOZ2-/- show induction of the fetal gene program typical of pathologic hypertrophy, however there was no evidence of hypertrophied morphometry at baseline. However, upon calcineurin activation or pressure overload-induced pathologic hypertrophy, MYOZ2-/- exhibited exaggerated cardiac hypertrophy, demonstrating that calsarcin-1 negatively modulates the function of calcineurin during pathologic hypertrophic remodeling.[11] Additional studies supported these findings in demonstrating that adenoviral overexpression of calsarcin-1 attenuated Gq alpha subunit-stimuated hypertrophy and ANP induction, by Angiotensin II, phenylephrine and endothelin-1 agonists in neonatal cardiomyocytes.[12] Overexpression of calsarcin-1 in mice (CS1Tg) was protective against Angiotensin II-induced pathologic cardiac hypertrophy, evidenced by preserved fractional shortening and contractility, as well as a blunted induction of the fetal hypertrophic gene program and significantly reduced expression of calcineurin-stimulated MCIP1.4 gene expression.[12] Taken together, these studies strongly support a role for calsarcin-1 in suppressing pathologic cardiac hypertrophy.
Clinical Significance
Two missense mutations in MYOZ2, Ser48Pro and Ile246Met, have been shown to be causal for rare forms of familial hypertrophic cardiomyopathy.[13]
References
- GRCh38: Ensembl release 89: ENSG00000172399 - Ensembl, May 2017
- GRCm38: Ensembl release 89: ENSMUSG00000028116 - Ensembl, May 2017
- "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- Andersson B, Wentland MA, Ricafrente JY, Liu W, Gibbs RA (April 1996). "A "double adaptor" method for improved shotgun library construction". Analytical Biochemistry. 236 (1): 107–13. doi:10.1006/abio.1996.0138. PMID 8619474.
- Yu W, Andersson B, Worley KC, Muzny DM, Ding Y, Liu W, Ricafrente JY, Wentland MA, Lennon G, Gibbs RA (April 1997). "Large-scale concatenation cDNA sequencing". Genome Research. 7 (4): 353–8. doi:10.1101/gr.7.4.353. PMC 139146. PMID 9110174.
- "Entrez Gene: MYOZ2 myozenin 2".
- http://www.heartproteome.org/copa/ProteinInfo.aspx?QType=Protein%20ID&QValue=Q9NPC6
- Zong NC, Li H, Li H, Lam MP, Jimenez RC, Kim CS, Deng N, Kim AK, Choi JH, Zelaya I, Liem D, Meyer D, Odeberg J, Fang C, Lu HJ, Xu T, Weiss J, Duan H, Uhlen M, Yates JR, Apweiler R, Ge J, Hermjakob H, Ping P (October 2013). "Integration of cardiac proteome biology and medicine by a specialized knowledgebase". Circulation Research. 113 (9): 1043–53. doi:10.1161/CIRCRESAHA.113.301151. PMC 4076475. PMID 23965338.
- Frey N, Richardson JA, Olson EN (December 2000). "Calsarcins, a novel family of sarcomeric calcineurin-binding proteins". Proceedings of the National Academy of Sciences of the United States of America. 97 (26): 14632–7. Bibcode:2000PNAS...9714632F. doi:10.1073/pnas.260501097. PMC 18970. PMID 11114196.
- Frey N, Barrientos T, Shelton JM, Frank D, Rütten H, Gehring D, Kuhn C, Lutz M, Rothermel B, Bassel-Duby R, Richardson JA, Katus HA, Hill JA, Olson EN (December 2004). "Mice lacking calsarcin-1 are sensitized to calcineurin signaling and show accelerated cardiomyopathy in response to pathological biomechanical stress". Nature Medicine. 10 (12): 1336–43. doi:10.1038/nm1132. PMID 15543153. S2CID 33712044.
- Frank D, Kuhn C, van Eickels M, Gehring D, Hanselmann C, Lippl S, Will R, Katus HA, Frey N (November 2007). "Calsarcin-1 protects against angiotensin-II induced cardiac hypertrophy". Circulation. 116 (22): 2587–96. doi:10.1161/CIRCULATIONAHA.107.711317. PMID 18025526.
- Osio A, Tan L, Chen SN, Lombardi R, Nagueh SF, Shete S, Roberts R, Willerson JT, Marian AJ (March 2007). "Myozenin 2 is a novel gene for human hypertrophic cardiomyopathy". Circulation Research. 100 (6): 766–8. doi:10.1161/01.RES.0000263008.66799.aa. PMC 2775141. PMID 17347475.
Further reading
- Faulkner G, Pallavicini A, Formentin E, Comelli A, Ievolella C, Trevisan S, Bortoletto G, Scannapieco P, Salamon M, Mouly V, Valle G, Lanfranchi G (July 1999). "ZASP: a new Z-band alternatively spliced PDZ-motif protein". The Journal of Cell Biology. 146 (2): 465–75. doi:10.1083/jcb.146.2.465. PMC 3206570. PMID 10427098.
- Frey N, Richardson JA, Olson EN (December 2000). "Calsarcins, a novel family of sarcomeric calcineurin-binding proteins". Proceedings of the National Academy of Sciences of the United States of America. 97 (26): 14632–7. Bibcode:2000PNAS...9714632F. doi:10.1073/pnas.260501097. PMC 18970. PMID 11114196.
- Ahmad F, Gonzalez O, Ramagli L, Xu J, Siciliano MJ, Bachinski LL, Roberts R (December 2000). "Identification and characterization of a novel gene (C4orf5) located on human chromosome 4q with specific expression in cardiac and skeletal muscle". Genomics. 70 (3): 347–53. doi:10.1006/geno.2000.6399. PMID 11161785.
- Takada F, Vander Woude DL, Tong HQ, Thompson TG, Watkins SC, Kunkel LM, Beggs AH (February 2001). "Myozenin: an alpha-actinin- and gamma-filamin-binding protein of skeletal muscle Z lines". Proceedings of the National Academy of Sciences of the United States of America. 98 (4): 1595–600. doi:10.1073/pnas.041609698. PMC 29302. PMID 11171996.
- Frey N, Olson EN (April 2002). "Calsarcin-3, a novel skeletal muscle-specific member of the calsarcin family, interacts with multiple Z-disc proteins". The Journal of Biological Chemistry. 277 (16): 13998–4004. doi:10.1074/jbc.M200712200. PMID 11842093.
- Gontier Y, Taivainen A, Fontao L, Sonnenberg A, van der Flier A, Carpen O, Faulkner G, Borradori L (August 2005). "The Z-disc proteins myotilin and FATZ-1 interact with each other and are connected to the sarcolemma via muscle-specific filamins". Journal of Cell Science. 118 (Pt 16): 3739–49. doi:10.1242/jcs.02484. PMID 16076904.
- Rual JF, Venkatesan K, Hao T, Hirozane-Kishikawa T, Dricot A, Li N, Berriz GF, Gibbons FD, Dreze M, Ayivi-Guedehoussou N, Klitgord N, Simon C, Boxem M, Milstein S, Rosenberg J, Goldberg DS, Zhang LV, Wong SL, Franklin G, Li S, Albala JS, Lim J, Fraughton C, Llamosas E, Cevik S, Bex C, Lamesch P, Sikorski RS, Vandenhaute J, Zoghbi HY, Smolyar A, Bosak S, Sequerra R, Doucette-Stamm L, Cusick ME, Hill DE, Roth FP, Vidal M (October 2005). "Towards a proteome-scale map of the human protein-protein interaction network". Nature. 437 (7062): 1173–8. Bibcode:2005Natur.437.1173R. doi:10.1038/nature04209. PMID 16189514. S2CID 4427026.
- Posch MG, Perrot A, Dietz R, Ozcelik C, Pankuweit S, Ruppert V, Richter A, Maisch B (June 2007). "Mutations in MYOZ1 as well as MYOZ2 encoding the calsarcins are not associated with idiopathic and familial dilated cardiomyopathy" (PDF). Molecular Genetics and Metabolism. 91 (2): 207–8. doi:10.1016/j.ymgme.2007.02.014. PMID 17434779.