Meyers synthesis
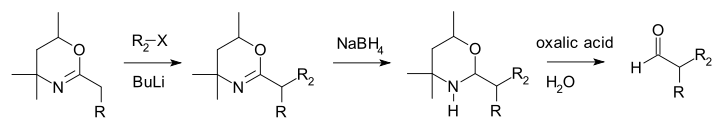
The Meyers synthesis is an organic synthesis for the preparation of unsymmetrical aldehydes via hydrolysis of an oxazine.[1][2][3][4] The reaction is named after the American chemist Albert Meyers.
The starting compound is a dihydro-1,3-oxazine with an alkyl group in the 2 position. The alpha proton is acidic and can be abstracted by a strong base such as butyl lithium and subsequently alkylated by an alkyl halide (haloalkane). In the next step the nitrogen to carbon double bond (imine) is reduced with sodium borohydride and the resulting oxazine (a hemiaminal) hydrolyzed with water and oxalic acid to the aldehyde.
References
- Aldehydes from dihydro-1,3-oxazines. I. Synthesis of aliphatic aldehydes and their C-1 deuterated derivatives Albert I. Meyers, Aiko Nabeya, H. Wayne Adickes, Ieva R. Politzer J. Am. Chem. Soc. 1969; 91(3); 763-764. doi:10.1021/ja01031a053
- Aldehydes from dihydro-1,3-oxazines. II. Synthesis of .alpha.,.beta.-unsaturated aldehydes and their C-1 deuterated derivatives Albert I. Meyers, Aiko Nabeya, H. Wayne Adickes, J. Michael Fitzpatrick, G. Ray Malone, and Ieva R. Politzer pp 764 - 765; J. Am. Chem. Soc. 1969 doi:10.1021/ja01031a054
- Aldehydes from dihydro-1,3-oxazines. III. Synthesis of cycloalkanecarboxaldehydes Albert I. Meyers, H. Wayne Adickes, Ieva R. Politzer, and Warren N. Beverung pp 765 - 767; J. Am. Chem. Soc. 1969 doi:10.1021/ja01031a055
- Organic Syntheses, Coll. Vol. 6, p.905 (1988); Vol. 51, p.24 (1971). http://www.orgsynth.org/orgsyn/pdfs/CV6P0905.pdf
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.