N-Phenethyl-4-piperidinone
N-Phenethyl-4-piperidinone (NPP) is a derivative of 4-piperidinone with the molecular formula C13H17NO. It is used as an intermediate in the manufacture of chemicals and pharmaceutical drugs such as fentanyl.
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
1-(2-Phenylethyl)piperidin-4-one | |
| Other names
1-Phenethylpiperidin-4-one (no longer recommended) N-Phenylethyl-4-piperidinone N-Phenethyl-4-piperidone | |
| Identifiers | |
3D model (JSmol) |
|
| Abbreviations | NPP |
| ChemSpider | |
| ECHA InfoCard | 100.049.630 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C13H17NO | |
| Molar mass | 203.28 g/mol |
| Density | 1.057 g/cm3 |
| Melting point | 56 to 60 °C (133 to 140 °F; 329 to 333 K) |
| Hazards | |
| GHS labelling:[1] | |
 | |
| Warning | |
| H302 | |
| P264, P270 | |
| NFPA 704 (fire diamond) | |
| Legal status | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Because of its possible use in the illicit manufacture of fentanyl, the United States Drug Enforcement Administration (DEA) placed NPP under control as a List 1 Chemical in 2007. Both domestic sales and domestic importations are thus subject to DEA reporting requirements.[3]
Preparation
N-Phenethyl-4-piperidinone can be prepared from 4-piperidinone and phenethyl bromide in biphasic conditions with a variety of phase transfer catalysts.
Uses
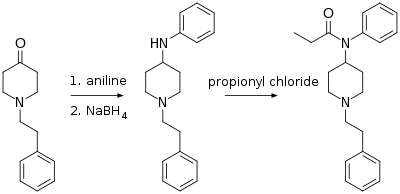
N-Phenethyl-4-piperidinone is useful in the synthesis of pharmaceuticals, primarily fentanyl and its analogs. Paul Janssen (founder of Janssen Pharmaceutica) first synthesized fentanyl in 1960 from Benzylfentanyl.[4] The Seigfried method (shown below and published on The Hive) involves reacting N-phenethyl-4-piperidinone with aniline, and then reducing the imine product with sodium borohydride to 4-anilino-N-phenethylpiperidine (ANPP). This product is reacted with propionyl chloride to form fentanyl.
References
- GHS: PubChem
- Anvisa (2023-03-31). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-04-04). Archived from the original on 2023-08-03. Retrieved 2023-08-15.
- DEA Chemical Handler's Manual: A Guide to Chemical Control Regulations (2022 ed.). United States Department of Justice Drug Enforcement Administration Office of Diversion Control. May 9, 2014.
- Schulz W. "Fentanyl". List of Top Pharmaceuticals. Chemical & Engineering News.