Pentafluorophenol
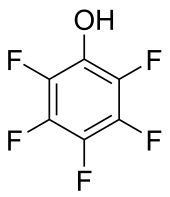
Pentafluorophenol is the organofluorine compound (specifically a fluoroalcohol) with the formula C6F5OH. This is the perfluorinated analogue of phenol. It is a white odorless solid that melts just above room temperature. With a pKa of 5.5, it is one of the most acidic phenols.
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Pentafluorophenol | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.011.123 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C6F5OH | |
| Molar mass | 184.065 g·mol−1 |
| Appearance | white solid or colorless liquid |
| Melting point | 32.8 °C (91.0 °F; 305.9 K) |
| Boiling point | 145.6 °C (294.1 °F; 418.8 K) |
| Hazards | |
| GHS labelling: | |
  | |
| Danger | |
| H302, H312, H314, H315, H319, H335 | |
| P260, P261, P264, P270, P271, P280, P301+P312, P301+P330+P331, P302+P352, P303+P361+P353, P304+P340, P305+P351+P338, P310, P312, P321, P322, P330, P332+P313, P337+P313, P362, P363, P403+P233, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Uses
Pentafluorophenol is used to prepare pentafluorophenyl esters, which are active esters useful in peptide synthesis.[1]
Environmental hazards
Pentafluorophenol is considered hazardous because of oral, dermal and inhalation toxicity and because it causes severe skin burns and eye damage.[2][3]
References
- Jones K, DeAmicis C (2009). "Pentafluorophenol". Encyclopedia of Reagents for Organic Synthesis. pp. 1–9. doi:10.1002/047084289X. hdl:10261/236866.
- "Pentafluorophenol SAFETY DATA SHEET". Thermo Fisher Scientific. January 18, 2018. Retrieved 26 February 2021.
- "Pentafluorophenol". PubChem. National Center for Biotechnology Information. February 20, 2021. Retrieved 26 February 2021.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.