Protochlorophyllide reductase
In enzymology, protochlorophyllide reductases (POR)[2][3] are enzymes that catalyze the conversion from protochlorophyllide to chlorophyllide a. They are oxidoreductases participating in the biosynthetic pathway to chlorophylls.[4][5]

| light-dependent protochlorophyllide reductase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| EC no. | 1.3.1.33 | ||||||||
| CAS no. | 68518-04-7 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / QuickGO | ||||||||
| |||||||||
| light-independent protochlorophyllide reductase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
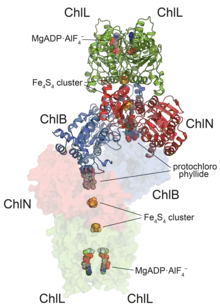 Crystallographic structure of heterooctamer of a dark-operative protochlorophyllide oxidoreductase from Prochlorococcus marinus.[1] | |||||||||
| Identifiers | |||||||||
| EC no. | 1.3.7.7 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| |||||||||
There are two structurally unrelated proteins with this sort of activity, referred to as light-dependent (LPOR) and dark-operative (DPOR). The light- and NADPH-dependent reductase is part of the short-chain dehydrogenase/reductase (SDR) superfamily and is found in plants and oxygenic photosynthetic bacteria,[6][7] while the ATP-dependent dark-operative version is a completely different protein, consisting of three subunits that exhibit significant sequence and quaternary structure similarity to the three subunits of nitrogenase.[8] This enzyme may be evolutionary older; due to its bound iron-sulfur clusters is highly sensitive to free oxygen and does not function if the atmospheric oxygen concentration exceeds about 3%.[9] It is possible that evolutionary pressure associated with the great oxidation event resulted in the development of the light-dependent system.
The light-dependent version (EC 1.3.1.33) uses NADPH:
- protochlorophyllide + NADPH + H+ chlorophyllide a + NADP+
While the light-independent or dark-operative version (EC 1.3.7.7) uses ATP and ferredoxin:[10][11][12]
- protochlorophyllide a + reduced ferredoxin + 2 ATP + 2 H2O = chlorophyllide a + oxidized ferredoxin + 2 ADP + 2 phosphate
Light-dependent
The light-dependent version has the accepted name protochlorophyllide reductase. The systematic name is chlorophyllide-a :NADP+ 7,8-oxidoreductase. Other names in common use include NADPH2-protochlorophyllide oxidoreductase, NADPH-protochlorophyllide oxidoreductase, NADPH-protochlorophyllide reductase, protochlorophyllide oxidoreductase, and protochlorophyllide photooxidoreductase.
LPOR is one of only three known light-dependent enzymes. The enzyme enables light-dependent protochlorophyllide reduction via direct local hydride transfer from NADPH and a longer-range proton transfer along a defined structural pathway.[13] LPOR is a ~40kDa monomeric enzyme, for which the structure has been solved by X-ray crystallography. It is part of the SDR superfamily, which includes alcohol dehydrogenase, and consists of a Rossman-fold NADPH-binding site and a substrate-specific C-terminal segment region. The protochlorophyllide substrate is thought to bind to a cavity near the nicotinamide end of the bound NADPH.[7][13] LPOR is primarily found in plants and oxygenic photosynthetic bacteria, as well as in some algae.
Light-independent
The light-independent version has the accepted name of ferredoxin:protochlorophyllide reductase (ATP-dependent). Systematically it is known as ATP-dependent ferredoxin:protochlorophyllide-a 7,8-oxidoreductase. Other names in common use include light-independent protochlorophyllide reductase and dark-operative protochlorophyllide reductase (DPOR).
DPOR is a nitrogenase homologue[8] and adopts an almost identical overall architecture arrangement to both nitrogenase as well as the downstream chlorophyllide a reductase (COR). The enzyme consists of a catalytic heterotetramer and two transiently-bound ATPase dimers (right).[14] Similar to nitrogenase, the reduction mechanism relies on an electron transfer from the iron-sulfur cluster of the ATPase domain, through a secondary cluster on the catalytic heterotetramer and finally to the protochlorophyllide-bound active site (which, distinct from nitrogenase, does not contain FeMoco). The reduction requires significantly less input than the nitrogenase reaction, requiring only a 2-electron reduction and 4 ATP equivalents, and as such may require an auto-inhibitory mechanism to avoid over-activity.[15]
DPOR can alternatively take as its substrate the compound with a second vinyl group (instead of an ethyl group) in the structure, in which case the reaction is
- 3,8-divinylprotochlorophyllide + reduced ferredoxin + 2 ATP + 2 H2O 3,8-divinylchlorophyllide a + oxidized ferredoxin + 2 ADP + 2 phosphate
This enzyme is present in photosynthetic bacteria, cyanobacteria, green algae and gymnosperms.[4][16]
See also
References
- PDB: 2ynm; Moser J, Lange C, Krausze J, Rebelein J, Schubert WD, Ribbe MW, Heinz DW, Jahn D (2013). "Structure of ADP-aluminium fluoride-stabilized protochlorophyllide oxidoreductase complex". Proc Natl Acad Sci U S A. 110 (6): 2094–2098. Bibcode:2013PNAS..110.2094M. doi:10.1073/pnas.1218303110. PMC 3568340. PMID 23341615.
- Griffiths WT (1978). "Reconstitution of chlorophyllide formation by isolated etioplast membranes". Biochem. J. 174 (3): 681–92. doi:10.1042/bj1740681. PMC 1185970. PMID 31865.
- Apel K, Santel HJ, Redlinger TE, Falk H (1980). "The protochlorophyllide holochrome of barley (Hordeum vulgare L.) Isolation and characterization of the NADPH:protochlorophyllide oxidoreductase". Eur. J. Biochem. 111 (1): 251–8. doi:10.1111/j.1432-1033.1980.tb06100.x. PMID 7439188.
- Willows, Robert D. (2003). "Biosynthesis of chlorophylls from protoporphyrin IX". Natural Product Reports. 20 (6): 327–341. doi:10.1039/B110549N. PMID 12828371.
- Bollivar, David W. (2007). "Recent advances in chlorophyll biosynthesis". Photosynthesis Research. 90 (2): 173–194. doi:10.1007/s11120-006-9076-6. PMID 17370354. S2CID 23808539.
- Nomata, Jiro; Kondo, Toru; Mizoguchi, Tadashi; Tamiaki, Hitoshi; Itoh, Shigeru; Fujita, Yuichi (May 2015). "Dark-operative protochlorophyllide oxidoreductase generates substrate radicals by an iron-sulphur cluster in bacteriochlorophyll biosynthesis". Scientific Reports. 4 (1): 5455. doi:10.1038/srep05455. ISSN 2045-2322. PMC 4071322. PMID 24965831.
- Dong, Chen-Song; Zhang, Wei-Lun; Wang, Qiao; Li, Yu-Shuai; Wang, Xiao; Zhang, Min; Liu, Lin (2020-04-14). "Crystal structures of cyanobacterial light-dependent protochlorophyllide oxidoreductase". Proceedings of the National Academy of Sciences. 117 (15): 8455–8461. doi:10.1073/pnas.1920244117. ISSN 0027-8424. PMC 7165480. PMID 32234783.
- Yuichi Fujita and Carl E. Bauer (2000). Reconstitution of Light-independent Protochlorophyllide Reductase from Purified Bchl and BchN-BchB Subunits. J. Biol. Chem., Vol. 275, Issue 31, 23583-23588.
- S.Yamazaki, J.Nomata, Y.Fujita (2006) Differential operation of dual protochlorophyllide reductases for chlorophyll biosynthesis in response to environmental oxygen levels in the cyanobacterium Leptolyngbya boryana. Plant Physiology, 2006, 142, 911-922
- Fujita Y, Matsumoto H, Takahashi Y, Matsubara H (March 1993). "Identification of a nifDK-like gene (ORF467) involved in the biosynthesis of chlorophyll in the cyanobacterium Plectonema boryanum". Plant & Cell Physiology. 34 (2): 305–14. PMID 8199775.
- Nomata J, Ogawa T, Kitashima M, Inoue K, Fujita Y (April 2008). "NB-protein (BchN-BchB) of dark-operative protochlorophyllide reductase is the catalytic component containing oxygen-tolerant Fe-S clusters". FEBS Letters. 582 (9): 1346–50. doi:10.1016/j.febslet.2008.03.018. PMID 18358835.
- Muraki N, Nomata J, Ebata K, Mizoguchi T, Shiba T, Tamiaki H, et al. (May 2010). "X-ray crystal structure of the light-independent protochlorophyllide reductase". Nature. 465 (7294): 110–4. Bibcode:2010Natur.465..110M. doi:10.1038/nature08950. PMID 20400946. S2CID 4427639.
- Zhang, Shaowei; Heyes, Derren J.; Feng, Lingling; Sun, Wenli; Johannissen, Linus O.; Liu, Huanting; Levy, Colin W.; Li, Xuemei; Yang, Ji; Yu, Xiaolan; Lin, Min (2019-10-31). "Structural basis for enzymatic photocatalysis in chlorophyll biosynthesis". Nature. 574 (7780): 722–725. Bibcode:2019Natur.574..722Z. doi:10.1038/s41586-019-1685-2. ISSN 0028-0836. PMID 31645759. S2CID 204849396.
- Moser, Jürgen; Lange, Christiane; Krausze, Joern; Rebelein, Johannes; Schubert, Wolf-Dieter; Ribbe, Markus W.; Heinz, Dirk W.; Jahn, Dieter (2013-02-05). "Structure of ADP-aluminium fluoride-stabilized protochlorophyllide oxidoreductase complex". Proceedings of the National Academy of Sciences. 110 (6): 2094–2098. Bibcode:2013PNAS..110.2094M. doi:10.1073/pnas.1218303110. ISSN 0027-8424. PMC 3568340. PMID 23341615.
- Corless, Elliot I.; Saad Imran, Syed Muhammad; Watkins, Maxwell B.; Bacik, John-Paul; Mattice, Jenna R.; Patterson, Angela; Danyal, Karamatullah; Soffe, Mark; Kitelinger, Robert; Seefeldt, Lance C.; Origanti, Sofia (January 2021). "The flexible N-terminus of BchL autoinhibits activity through interaction with its [4Fe-4S] cluster and released upon ATP binding". Journal of Biological Chemistry. 296: 100107. doi:10.1074/jbc.RA120.016278. PMC 7948495. PMID 33219127.
- Bollivar DW (November 2006). "Recent advances in chlorophyll biosynthesis". Photosynthesis Research. 90 (2): 173–94. doi:10.1007/s11120-006-9076-6. PMID 17370354. S2CID 23808539.
Ferredoxin:protochlorophyllide+reductase+(ATP-dependent) at the U.S. National Library of Medicine Medical Subject Headings (MeSH)