Rock hyrax midden
A rock hyrax midden is a stratified accumulation of fecal pellets and a brown amber-like a urinary product known as hyraceum excreted by the rock hyrax and closely related species.[1]
Hyrax middens form very slowly (ranging from ~5 years to >1000 years for 1 mm of hyraceum accumulation[2][3]), over long periods of time, with many spanning tens of thousands of years[4] and some dating as far back as ~70,000 years.[5] Hyrax middens contain a diverse range of paleoenvironmental proxies, including fossil pollen and stable carbon, nitrogen and hydrogen isotopes.[5][4][6][7][8] Combined with the antiquity of hyrax middens, and the often-continuous nature of their deposition, hyrax middens have become a valuable means of reconstructing past environmental and climate change[1]
Rock hyraxes are known to use communal latrines.[9][10] These sites are often found in sheltered locations, where the threat of predation is limited, and middens form when they are protected from the elements. At well-protected sites, it may accumulate in deposits in excess of a meter thick and several meters across.[2][5] The thickness of hyrax middens depends on the nature of the shelter and the regional climate history and geology. Hyraceum shows hygroscopic properties and periods of increased precipitation or elevated ambient humidity will destroy existing middens, while more arid periods allow their development/preservation.[1] Thicker formations tend to occur in shallow shelters that during more arid periods, presumably provided sufficient shelter from rainfall for substantial midden accumulations, but under wetter conditions no longer provide adequate protection, resulting in the removal of the more soluble components of the midden. At poorly protected sites in arid regions hyrax urine leaves a white, calcium carbonate[11] precipitate on the rocks. Varying degrees of protection result in varying degrees of midden preservation. Small overhangs, vertical fractures in cap rocks, and groundwater flow along weakness in the shelter’s architecture may lead to midden degradation if rainfall exceeds a certain amount and/or intensity. The thickest middens have been found at sites composed of massive, horizontally bedded rock such as granite and quartzites with between ~30 and 480 mm of annual rainfall.[1] In more humid environments (>800 mm mean annual rainfall), there is little to no evidence of hyraceum accumulation, and middens typically resemble piles of compost, as the masticated plant material in the pellets rapidly decomposes. Hyraceum-rich middens do not typically form in coastal situations, despite the presence of hyraxes, and it is considered that the ambient humidity of the air and the occurrence of coastal fogs preclude midden development[1]
Comparisons with fossilised herbivore middens
Studies of other herbivore midden remains have been very effective in palaeoenvironmental studies in dryland regions on several continents. In the southwestern United States pack rat middens have provided an unprecedented record of environmental changes over the last 40,000 years.[12][13][14] As a result of this work, the vegetation dynamics of this area are some of the best understood for any of the world’s drylands at this timescale, and the critical data provided have dramatically helped define the range of regional climate variability. This work has also lead to important perspectives on ecological theory,[15] which have impacted on management strategies[16][17] by allowing a distinction to be made between anthropogenic environmental impacts and natural processes.
Midden studies have also been undertaken in Australia[18][19][20][21] and South America.[22][23][24] This work has highlighted a fundamental difference between middens from these regions and hyrax middens. American and Australian middens are essentially nests composed of sticks and other macrobotanical remains. These middens are generally reported have no clear stratigraphy, and researchers have thus adopted the methodology of processing them as single samples that provide a palaeoenvironmental snapshot.[25] Hyrax middens, on the other hand, are primarily urino-fecal deposits, and are deposited progressively as a series of layers.[1] This diachroneity is one of the fundamental advantages of hyrax middens over nest middens, which are only secondarily preserved as the animals urinate in their shelters.
Examinations of the internal and external structure of hyrax middens suggest flow/deposition dynamics similar to speleothems (cave deposits, e.g. stalactites), with the fresh urine flowing across the surface of the midden, then drying and crystallising, preserving the stratigraphic integrity of the midden. The general morphology of middens is often characterised by (1) lobate forms, (2) undulating weathering features on exposed midden faces, and (3) in some cases the formation of thin (1-3 mm in diameter) stalactites on the underside of some middens. As a result, questions over the potential for post-depositional remobilisation of hyraceum may be raised. The examination of over 150 middens, however, has confirmed the visible stratigraphic integrity of the middens, and while some surficial alteration of exposed surfaces can occur, consistently coherent age-depth models, and the nearly vertical exposed external faces of the middens indicate that, once dry, hyraceum is not prone to significant remobilization.[1]
Hyrax midden structure, accumulation rates and age
Hyrax midden structures and accumulation rates can vary considerably based on the relative proportion of their two primary components, pellets and hyraceum, which is determined by the architecture of the site itself. Depending on the shape and irregularities of the floor of the site in question, pellets are likely to either accumulate (in concave structures) or roll away (in convex or inclined structures). Whereas hyrax urine will deposit only a very thin film of hyraceum after evaporation, pellets are usually 0.5-1 cm in diameter, and thereby accumulate much more quickly.[7], with deep piles accumulating perhaps within just a few years, or even months. Compared to this, we have observed that middens composed primarily of hyraceum accumulate much more slowly; generally between ~5 to >1000 years/mm.[3][2] The rate of hyraceum accumulation depends on the morphology of the midden, the architecture of the site, as well as presumably the size of the hyrax colony, and as such net rates can be highly variable[5][4]
Hyrax midden ages
Radiocarbon ages from hyraceum are not subject to reservoir effects or the inclusion of new carbon.[26][27] This is primarily a function of middens being isolated systems, and that through respiration the hyraceum is brought into equilibrium with atmospheric 14C at the time of deposition. Published data show that hyrax middens can be of considerable antiquity, and middens from the Groenfontein site in the Cederberg Mountains of South Africa are considered to have begun accumulating ~70,000 years ago[5]
It has been commonly observed that many middens are no longer actively accumulating. Often this is controlled by the shelters in which they are found, with accumulation ceasing when the middens grow to such an extent that the hyraxes can no longer physically enter the shelters. Until recently, field sampling was limited to the collection of middens that were most accessible and easiest to sample. In many cases this meant that the individual sampled middens were relatively thin (<5 cm) with aggregate records subsequently constructed from fragments of as many as 25 separate middens.[28][29] (Scott and Woodborne, 2007a, b). With recent developments in sampling tools and techniques, larger, more stratigraphically coherent middens are more regularly sampled, which better represent the full period of accumulation at a given site[7][30][5]
Composition of hyrax middens
The very nature of hyrax middens implies that they comprise a mixture of materials, which include animal metabolic products, undigested food, and any allochtonous material blown into the middens or deposited via feet or fur.[31] In terms of organic matter, the existence of such potentially distinct sources (i.e. extraneous organic matter and animal metabolites) implies that a range of information concerning inter alia: animal diet, animal behaviour, metabolic responses to environmental stress, changing behaviour, as well the wider palaeoecological setting of the site may all be preserved within hyraceum.
Chemical composition
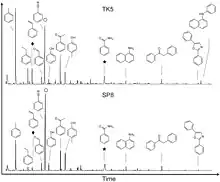
Hyraceum essentially comprises a mix of organic compounds, soluble salts, calcium carbonate and the mineral sylvite.[32][33] More recent data from Raman Spectroscopy and Fourier Transform Infrared (FTIR) Spectroscopy demonstrate the presence of a number of CaCO3 polymorphs, the abundance of sylvite (KCl) and an organic component[34]
The organic components within hyraceum have been investigated using pyrolysis-GC/MS (py-GC/MS) and GC/MS analysis of solvent-extractable lipids.[35] Py-GC/MS is commonly applied to elucidate macromolecular organic matter structure and composition. Py-GC/MS measurements on samples from two distant sites, Spitzkoppe, Namibia and Truitjes Kraal, Western Cape Province, South Africa produced remarkably similar suites of pyrolysis products,[31] despite their contrasting environmental settings. The pyrolysis products were dominated by aromatic compounds; notably the nitrogenous compounds benzonitrile and benzamide. Pyrolysis in the presence of a methylating agent tetramethylammonium hydroxide (TMAH) implied that benzamide is a monomer of a larger polymeric structure, the major organic component of the hyraceum OM.[35] This is further supported by the ubiquity of benzamide within solvent extracts and it is probable that it is derived from hippuric or benzoic acid, which are common metabolites in ruminants.[36] Given its abundance, the metabolite (or metabolite product) benzamide is likely the major source of organic nitrogen and carbon measured in bulk stable isotope analyses,[7][5][30][35] and can therefore provide insights into animal diet and its isotopic signature. Interestingly, common plant-derived pyrolysis products, such as lignin were not detected using py-GC/MS, although low molecular weight polysaccharide pyrolysis products (e.g. acetyl furan, furaldehyde, dimethyl furan) were found in trace amounts[31]
That such plant-derived compounds might be identified with this technique following more detailed analytical pyrolysis protocols is implied by new FTIR analyses of the organic fraction, which support the basic pyrolysis-based interpretation of Spitzkoppe and Truitjes Kraal midden chemical compositions. The Spitzkoppe FTIR spectra following carbonate removal contains a broad absorption band at w3300 cm1 as well as sharper absorptions from w1600 to 1700, 1400, 1130, 770 and 690 cm-1. Multiplets between 1560 and 1640 cm-1 have been reported as being due to NeH bending in primary amines.[37][38], while a signal at w1650 cm1 is representative of C==O stretches of the amide band. The spectra thus bear a strong resemblance to benzamide.[38][39] FTIR spectra from the Truitjes Kraal midden, which is rich in faecal material, shows some resemblance to that of cellulose, with strong broad bands at 3400 cm-1 and 1050 cm-1, and some weaker broad bands at 1730 and1670 cm-1. Overall, the FTIR spectra and previous studies reveal a complex mixture of salts and organic compounds, with the latter incorporating aromatics, polysaccharides, amines, amides and other carbonyl-containing compounds. There are also clear similarities with the spectrum of benzamide, particularly at Spitzkoppe, which is consistent with the pyrolysis data[31]
Palaeoenvironmental proxies
Part of the extraordinary potential of hyrax middens as palaeoenvironmental archives is the large range of proxies that are contained within them. Initially, when their diachronic nature was less evident, they were viewed as the poor relation to the better studied pack rat middens.[40][41] While pack rat middens are rich in identifiable macrofossils, which can be directly dated and provide high taxonomic resolution, hyrax middens are poor in macroremains. Those that are found are almost exclusively masticated material that has been incorporated into the deposits as faecal pellets. While some studies have analysed these midden components,[40][41] more recent work suggests that this approach does not maximise the full potential of hyrax middens as palaeoenvironmental archives[1]
Hyrax middens contain a suite of proxies that have the potential to provide clear insights into past climate and vegetation change. Working within the context of the middens’ stratigraphy, and building on robust chronologies indicating predictable and consistent accumulation rates, sampling methodologies are now more akin to those applied to speleothems rather than to packrat middens. Whereas the early focus was on small (<1 kg), accessible middens and in some cases in-situ sub-sampling, it is now standard practice to collect larger (10-70 kg) segments of the best-developed middens.[5][6][7] The segments are then split and polished in a controlled environment, and subsamples for radiocarbon dating and proxy analysis. That multiple proxies can be analysed from the same subsample allows for direct comparability, and much more reliable insights into the interrelationships between the systems being studied. This is valuable when comparing proxies that reflect vegetation change (e.g. fossil pollen) and those that are primarily influenced by climate (e.g. δ15N), as the relative roles of climatic forcing versus vegetation dynamics related to competitive processes within an ecosystem can be better resolved, resulting in a fuller and more reliable understanding of palaeoenvironmental dynamics[3][7]
Pollen
Hyrax middens contain well-preserved micro plant material including pollen, which is sealed in middens by hyraceum, protecting it from microbial activity and decay. The earliest study of fossil pollen from a hyrax midden was undertaken in the late 1950s by Pons and Quézel.[42] in the Hoggar Massif of Algeria, whereas the first palynological analyses of southern African middens were undertaken during the late 1980s and early 1990s,[43][44][45][46] and demonstrated that hyrax middens are very useful as pollen and microfossil traps.[43][45] Subsequently, hyrax middens have been become an important archive for fossil pollen analysis in South Africa[47][28][48][49][50] and Namibia.[51][52][3] Studies of fossil pollen in hyrax middens have also been undertaken in Jordan[41] Ethiopia,[53] Yemen[54] and Oman[55]
Taphonomy, preservation and concentrations
Middens are excellent traps for pollen derived from the local and regional surroundings either via the alimentary channel of the animals (excreted in pellets) or via deposition on the middens. The airborne pollen rain is incorporated by (1) collecting on the surface of the midden, (2) being brought in on the fur of the hyraxes, or (3) being ingested as dust on dietary items such as plant leaves or drinking water.[43][45][1] The dietary component may also represent the ingestion of flowers, which may result in the occasional over-representation of pollen of certain plant species in the pellet fraction of certain middens. A clear benefit of midden pollen spectra over wetland pollen spectra is that they may more clearly reflect terrestrial vegetation, without the high proportions of hydrophilic elements found in wetland sequences, which is particularly problematic in some dryland pollen records.[56] Furthermore, as the pollen found in hyraceum is not exclusively wind-transported, usually under-represented entomophilous plants are more clearly represented.
Preservation of pollen sealed in hyraceum is usually very good, but the degradation of pollen grains has been occasionally observed in loose pellets or middens semi-exposed to the elements, such as in dolerite shelters in the central grassland region of South Africa, where some Asteraceae pollen have apparently lost their ektexine (L. Scott, unpublished observation). Compared to other available palaeoarchives in the region, such as fluvial sediments or paleosols, and to more widely used pollen records from peat bog and lakes, middens contain high fossil pollen concentrations; usually between 1 and 2 x 105 pollen grains per gram of sample. Pollen concentrations are high even in poorly productive ecosystems such as the Namib Desert margins.[57] Concentrations increase markedly when analysing pollen contents from pellets, reaching 5-30 x 105 pollen grains/gram of sample.
Interpretation of pollen data
There are some potential drawbacks for the palynological analysis of middens, however, as the diverse taphonomic vectors can complicate interpretations if they are not adequately considered and controlled for. Pollen spectra from pellets - reflecting the animal’s needs or preferences on a particular day - may contrast strongly with pollen spectra preserved in hyraceum, and which thought to be primarily brought to the midden via the fur of the hyraxes (which is collected as it moves through the vegetation around its shelter) and the wind. The degree to which dietary biases affect pollen spectra in pellets is a subject that is not fully understood. While Scott and Cooremans[58] have shown that at the biome scale, fresh pellets reflect vegetation of the region from which they were collected, including the seasonal variations within vegetation types, most published studies also indicate significant differences both between modern pellets, and between modern pellets and surface sediment samples from the same site.[59][57] A number of options might explain this, but it is assumed that as any given pellet represents what was eaten in the last day(s), there will be substantial inter-seasonal and inter-annual variation in the pollen preserved.
In most fossil pollen archives, wind-pollinated plants may dominate the natural pollen rain. Pollen production, however, is likely to have a less significant influence on the pollen that hyraxes ingest, and considering the wide variety of plants that they may eat, it may be possible to control for the taxon over-representation resulting from a production bias while still attaining a reasonable representation of the local vegetation.[60][58][33]
A study from the Lower Omo Basin of Ethiopia collected several dozen pellets from different areas around the study site, aggregated them into a single sample, and compared them to the local vegetation.[53]
Structured studies to clarify the relative influence of regional (aeolian) and local (fur) signals in the pollen preserved in hyraceum remain to be completed. At least in some cases, aeolian inputs appear to be negligible as some middens that have accumulated in vertical cracks - precluding the incorporation of pellets and direct contact with the animals - have been found to be devoid of pollen. If aeolian pollen does represent a small percentage of the pollen preserved in hyraceum then it might be inferred that hyraceum pollen assemblages reflect primarily local vegetation cover from within the animals’ primary feeding range.[1]
Stable isotopes
As hyrax middens have been developed as palaeoenvironmental archives, there has been increasing emphasis on the application of stable isotope analyses to midden sequences. Initially this focussed on the use of bulk 13C data, with an emphasis on identifying changes in the relative abundance of C3/C4/CAM vegetation and associated palaeoecological/palaeoenvironmental inferences.[61] This is useful in climatic transition zones, such as the Western Cape Province of South Africa, where modern rainfall seasonality has a strong impact on C3/C4 grass distributions.[62][61] δ13C records can also be used in some ecoregions, such as the dry savannah at Spitzkoppe in Namibia, as an indicator of the reliability of grass cover. As hyraxes will preferentially graze (grasses are C4 in the region), more depleted δ13C values from hyrax middens have been interpreted as evidence that the animals were forced to obtain a greater proportion of their diet from trees and shrubs, which are less susceptible to extended periods of drought.[6] However, these data do not necessarily provide a direct and unambiguous indicator of past arid/humid shifts. As such, other studies have focussed on the use of δ15N data as a potential proxy for water availability in the environment[6][7][4][5]
δ15N of hyrax middens as an indicator of past hydrologic change
In palaeoclimatology, the variables for which reconstructions are most often sought are humidity and temperature. Unfortunately, direct, or even reliable, proxies for these are rarely available, and it is necessary to make several inferential steps in order to interpret their past variability. Recent work on hyrax middens has shown that δ15N records from middens may provide a clearer, more direct estimation of water availability than previously possible in southern Africa, e.g..[5][4][2][6]
As described by Chase et al.[1] it has long been understood that the 15N abundance in animal tissues is influenced by diet, climate and/or physiology.[63][64][65] In terms of diet, a clear distinction exists between δ15N values in carnivores and herbivores, with enrichment in 15N occurring up trophic levels.[66] Among herbivores, a link between increased δ15N values in animal tissues and aridity was identified very early,[63][66] but it was thought to be predominantly a function of the animals’ metabolism. Ambrose and DeNiro,[64][67] based in part on the apparent lack of relationship between 15N/14N ratios in plants and the amount of rainfall,[63] developed a model to account for the enrichment of 15N in animal tissues based on physiological mechanisms of water conservation and nitrogen isotope mass balance. In this model, under arid conditions drought-tolerant herbivores concentrate their urine and excrete more 15N-depleted urea, leaving the body enriched in 15N. Conversely, water-dependent species that do not concentrate their urine were observed to have smaller δ15N ranges and lower mean values in their tissues.[64] This predicted differentiation between drought-tolerant and water-dependent species, however, only found partial support in South Africa.[68] This study suggested that animals in arid regions are likely to eat lower protein diets (%N decreases with increasing aridity[69]), and that the additional protein produced by symbiotic bacteria in the animals’ digestive tracts would essentially result in a shift in trophic level and an enrichment of 15N in the animals’ tissues. Similarly, Codron and Codron[70] found no significant difference in faecal δ15N between drought-tolerant and water-dependent herbivores, but did identify a significant correlation between %N and δ15N.
In contrast to the initial findings of Heaton et al.[63], subsequent studies of soils and plants across aridity gradients, indicate a clear negative correlation between 15N and rainfall.[71][72][69][73] As this was the original impetus for the construction of the mass balance model and its corollaries, these models, and their implications for interpreting δ15N records in plant and animals tissues, should be reconsidered. Although a strong relationship has been established between soil and plant δ15N, the link with rainfall is sometimes considered to be less robust (e.g.[70]). One of the primary difficulties in determining the relationship between precipitation and δ15N values in soils, plants, animal tissues and excrement is the means by which precipitation is determined. It has been noted by Handley et al.[71] that δ15N in soils and plants may change substantially across a landscape as a function of variations in soil moisture. Since soil moisture varies as a result of subtle changes in topography, aspect and soil type, particularly in drylands where sparse vegetation and poorly-formed soils exacerbate the heterogeneity of the biogeochemical landscape,[74] the common practice of using rainfall records from the nearest gauge and/or interpolated from regional stations will inevitably weaken the significance of any correlation. Soil moisture and δ15N also vary significantly over short, sub-seasonal timescales[71] and, combined, these fine-scale spatio-temporal variations need to be adequately controlled for if reliable δ15N-climate correlations are to be identified.
If we accept that plant δ15N is determined by soil δ15N, and the link with climate, while identified, has been imperfectly explored, it remains to determine to what extent variations in plant d15N account for the variations identified in animal tissue and/or excrement. Murphy and Bowman[75][76] investigated variations in grass and kangaroo bone δ15N from across Australia and demonstrated a remarkably consistent relationship between plant and bone δ15N signals. Moisture availability, through its influence on the isotopic signature of plants/diet, was inferred as the primary control on animal δ15N, with metabolism having no clear effect. It is interesting to note that Ambrose and DeNiro’s[64] findings are not inconsistent with these results, as drought-tolerant species can inhabit more arid regions with less regular rainfall (higher, wider δ15N range) while water-dependent animals will be more restricted to well-watered areas (lower, smaller δ15N range). To extend the findings of Murphy and Bowman[75][76] to the study of excrement and hyrax middens, one can consider the studies of (1) Codron and Codron,[70] which concluded that faecal δ15N correspond to changes in plant δ15N, and (2) Sponheimer et al.[77], which found that, while preferential urinary excretion of isotopically light nitrogen may occur under conditions of disequilibrium, an unstressed animal at “steady state” will have equivalent dietary and excreta δ15N. Since faecal and animal δ15N track plant δ15N, and under normal conditions total excreta δ15N is equivalent to dietary (plant) δ15N, it follows that urinary δ15N, while perhaps more negative relative to dietary δ15N,[77] will reflect trends in plant δ15N and water availability.
Hyrax middens thus provide an optimal archive for the study of δ15N as a proxy for long-term environmental change. The effects of contemporary ecosystem variability are mitigated by the spatial and temporal averaging intrinsic in hyraxes’ wide dietary preferences, restricted range, the probable contribution of multiple individuals to a single δ15N sample, and the relatively long periods of time incorporated into each sample. In these archives, microtopographic variations in soil moisture (and thus δ15N) are accounted for by the feeding habits of the hyrax, and it is expected that the spatio-temporal averaging will allow for the reliable identification of long-term changes in water availability as reflected in variations in midden δ15N. Over long timescales (102-103 yr), this expectation is borne out, and the potential of hyrax middens as diachronic palaeoclimatic records has been supported by strong similarities between variations in δ15N records and a range of palaeoenvironmental proxies reflecting changes in precipitation.[5][4][2][6]
References
- Chase, Brian M.; Scott, Louis; Meadows, Michael E.; Gil-Romera, Graciela; Boom, Arnoud; Carr, Andrew S.; Reimer, Paula J.; Truc, Loïc; Valsecchi, Veruschka; Quick, Lynne J. (2012). "Rock hyrax middens: a palaeoenvironmental archive for southern African drylands". Quaternary Science Reviews. 56: 107–125. Bibcode:2012QSRv...56..107C. doi:10.1016/j.quascirev.2012.08.018 – via Elsevier Science Direct.
- Chase, Brian M.; Carr, Andrew S.; Boom, Arnoud; Reimer, Paula J. (2022). "Climate variability along the margin of the southern African monsoon region at the end of the African Humid Period". Quaternary Science Reviews. 291. Bibcode:2022QSRv..29107663C. doi:10.1016/j.quascirev.2022.107663. S2CID 251359250 – via Elsevier Science Direct.
- Chase, Brian M.; Boom, Arnoud; Carr, Andrew S.; Meadows, Michael E.; Lim, Sophak (2023). "A ca. 39,000-year record of vegetation and climate change from the margin of the Namib Sand Sea". Quaternary Research: 1–11. doi:10.1017/qua.2023.29. S2CID 259886588 – via Cambridge Core.
- Chase, Brian M.; Niedermeyer, Eva; Boom, Arnoud; Carr, Andrew S.; Chevalier, Manuel; He, Feng; Meadows, Michael E.; Ogle, Neil; Reimer, Paula J. (2019). "Orbital controls on Namib Desert hydroclimate over the past 50,000 years". Geology. 47 (9): 867–871. Bibcode:2019Geo....47..867C. doi:10.1130/G46334.1. S2CID 201305809 – via Geological Society of America.
- Chase, Brian M.; Carr, Andrew S.; Boom, Arnoud; Tyrrell, Genevieve; Reimer, Paula J. (2023). "Linking upwelling intensity and orbital-scale climate variability in South Africa's winter rainfall zone: insights from a ~70,000-year hyrax midden record". Quaternary Science Advances. 12. Bibcode:2023QSAdv..1200110C. doi:10.1016/j.qsa.2023.100110. S2CID 260694557 – via Elsevier Science Direct.
- Chase, Brian M.; Meadows, Michael E.; Scott, Louis; Thomas, David S.G.; Marais, Eugene; Sealy, Judith; Reimer, Paula J. (2009). "A record of rapid Holocene climate change preserved in hyrax middens from southwestern Africa". Geology. 37 (8): 703–706. Bibcode:2009Geo....37..703C. doi:10.1130/G30053A.1 – via The Geological Society of America.
- Chase, Brian M.; Boom, Arnoud; Carr, Andrew S.; Carre, Matthieu; Chevalier, Manuel; Meadows, Michael E.; Pedro, Joel B.; Stager, J. Curt; Reimer, Paula J. (2015). "Evolving Southern Hemisphere response to abrupt deglacial North Atlantic climate change events". Quaternary Science Reviews. 121: 132–136. doi:10.1016/j.quascirev.2015.05.023. S2CID 126734882 – via Elsevier Science Direct.
- Scott, Louis; Marais, Eugene; Brook, George A. (2004). "Fossil hyrax dung and evidence of Late Pleistocene and Holocene vegetation types in the Namib Desert". Journal of Quaternary Science. 19 (8): 829–832. Bibcode:2004JQS....19..829S. doi:10.1002/jqs.870. S2CID 85560796 – via Wiley Online Library.
- Sale, J.B. (1960). "The Hyracoidea: a review of the systematic position and biology of the hyrax" (PDF). Journal of East African Natural History Society – via Biodiversity Library.
- Louw, Elza; Louw, G.N.; Retief, C.P. (1972). "Thermolability, heat tolerance and renal function in the dassie or hyrax (Procavaia capensis)". Zoologica Africana. 7 (2): 451–469. doi:10.1080/00445096.1972.11447455 – via Taylor & Francis Online.
- Leon, B.; Belonje, P.C. (1979). "Calcium, phosphorus and magnesium excretion in the rock hyrax, Procavia capensis". Comparative Biochemistry and Physiology Part A: Physiology. 64: 67–72. doi:10.1016/0300-9629(79)90431-6 – via Elsevier Science Direct.
- King, James E.; van Devender, Thomas R. (1977). "Pollen analysis of fossil packrat middens from the Sonoran Desert". Quaternary Research. 8 (2): 191–204. Bibcode:1977QuRes...8..191K. doi:10.1016/0033-5894(77)90045-X. S2CID 128594218 – via Elsevier Science Direct.
- Betancourt, Julio L.; Davis, Owen K. (1984). "Packrat middens from Canyon de Chelly, northeastern Arizona: paleoecological and archaeological implications". Quaternary Research. 21 (1): 56–64. Bibcode:1984QuRes..21...56B. doi:10.1016/0033-5894(84)90089-9. S2CID 129171197 – via Elsevier Science Direct.
- Anderson, R. Scott; Betancourt, Julio L.; Mead, Jim I.; Hevly, Richard H.; Adam, David P. (2000). "Middle- and late-Wisconsin paleobotanic and paleoclimatic records from the southern Colorado Plateau, USA". Palaeogeography, Palaeoclimatology, Palaeoecology. 155 (1–2): 31–57. Bibcode:2000PPP...155...31A. doi:10.1016/S0031-0182(99)00093-0 – via Elsevier Science Direct.
- Betancourt, Julio L.; van Devender, Thomas R.; Martin, Paul S. (1990). Packrat Middens: The Last 40,000 Years of Biotic Change (1st ed.). University of Arizona Press.
- Betancourt, Julio L.; Schuster, William S.; Mitton, Jeffry B.; Anderson, R. Scott (1991). "Fossil and Genetic History of a Pinyon Pine (Pinus Edulis) Isolate". Ecology. 72 (5): 1685–1697. doi:10.2307/1940968. JSTOR 1940968 – via JSTOR.
- Betancourt, Julio L.; Pierson, Elizabeth A.; Rylander, Kate A.; Fairchild-Parks, James A.; Dean, Jeffrey S. (1993). "Influence of history and climate on New Mexico pinyon-juniper woodlands., Managing Pinyon-Juniper for Sustainability and Social Needs" (PDF). Department of Agriculture, Forest General Technical Reports – via USDA.
- Pearson, Stuart (1999). "Late Holocene biological records from the middens of stick-nest rats in the central Australian arid zone". Quaternary International. 59 (1): 39–46. Bibcode:1999QuInt..59...39P. doi:10.1016/S1040-6182(98)00070-6 – via Elsevier Science Direct.
- Pearson, Stuart; Dodson, John R. (1993). "Stick-nest rat middens as sources of paleoecological data in Australian deserts". Quaternary Research. 39 (3): 347–354. Bibcode:1993QuRes..39..347P. doi:10.1006/qres.1993.1041. S2CID 129163635 – via Elsevier Science Direct.
- McCarthy, Lynne; Head, Lesley; Quade, Jay (1996). "Holocene palaeoecology of the northern Flinders Ranges, South Australia, based on stick-nest rat (Leporillus spp.) middens: a preliminary overview". Quaternary Research. 123 (1–4): 205–218. Bibcode:1996PPP...123..205M. doi:10.1016/0031-0182(96)01113-3 – via Elsevier Science Direct.
- Pearson, Stuart; Betancourt, Julio L. (2002). "Understanding arid environments using fossil rodent middens". Journal of Arid Environments. 50 (3): 499–511. Bibcode:2002JArEn..50..499P. doi:10.1006/jare.2001.0901 – via Elsevier Science Direct.
- Holmgren, Camille A.; Roselle, E.; Latorre, Claudio; Betancourt, Julio L. (2008). "Late-Holocene fossil rodent middens from the Arica region of northernmost Chile". Journal of Arid Environments. 72 (5): 677–686. Bibcode:2008JArEn..72..677H. doi:10.1016/j.jaridenv.2007.09.003 – via Elsevier Science Direct.
- Diaz, Francisca P.; Latorre, Claudio; Maldonado, Antonio; Quade, Jay; Betancourt, Julio L. (2012). "Rodent middens reveal episodic, long-distance plant colonizations across the hyperarid Atacama Desert over the last 34,000 years". Journal of Biogeography. 39 (3): 510–525. doi:10.1111/j.1365-2699.2011.02617.x. S2CID 83370136 – via Wiley Online Library.
- Gayo, E.M.; Latorre, C.; Santoro, C.M.; Maldonado, A.; De Pol-Holz, R. (2012). "Hydroclimate variability in the low-elevation Atacama Desert over the last 2500 yr". Climate of the Past. 8 (1): 287–306. Bibcode:2012CliPa...8..287G. doi:10.5194/cp-8-287-2012.
- Spaulding, W.G., Betancourt, J.L., Croft, L.K., Cole, K.L., 1990. Packrat middens: their composition and methods of analysis, in: Betancourt, J.L., Devender, T.R.V., Martin, P.S. (Eds.), Packrat Middens: The Last 40,000 Years of Biotic Change. University of Arizona Press, pp. 59-84
- Kaufman, A., Broecker, W., 1965. Comparison of Th230 and C14 Ages for Carbonate Materials from Lakes Lahontan and Bonneville. J. Geophys. Res. 70, 4039-4054.
- Grimm, E.C., Maher Jr, L.J., Nelson, D.M., 2009. The magnitude of error in conventional bulk-sediment radiocarbon dates from central North America. Quaternary Research 72, 301-308.
- Scott, Louis; Woodborne, Stephan (2007). "Pollen analysis and dating of Late Quaternary faecal deposits (hyraceum) in the Cederberg, Western Cape, South Africa". Review of Palaeobotany and Palynology. 144 (3–4): 123–34. Bibcode:2007RPaPa.144..123S. doi:10.1016/j.revpalbo.2006.07.004 – via Elsevier Science Direct.
- Scott, Louis; Woodborne, Stephan (2007). "Vegetation history inferred from pollen in Late Quaternary faecal deposits (hyraceum) in the Cape winter-rain region and its bearing on past climates in South Africa". Quaternary Science Reviews. 26 (7–8): 941–953. Bibcode:2007QSRv...26..941S. doi:10.1016/j.quascirev.2006.12.012 – via Elsevier Science Direct.
- Chase, Brian M.; Meadows, Michael E.; Scott, Louis; Thomas, David S.G.; Marais, Eugene; Sealy, Judith; Reimer, Paula J. (2009). "A record of rapid Holocene climate change preserved in hyrax middens from southwestern Africa". Geology. 37 (8): 703–706. Bibcode:2009Geo....37..703C. doi:10.1130/G30053A.1 – via The Geological Society of America.
- Chase, Brian M.; Scott, Louis; Meadows, Michael E.; Gil-Romera, Graciela; Boom, Arnoud; Carr, Andrew S.; Reimer, Paula J.; Truc, Loïc; Valsecchi, Veruschka; Quick, Lynne J. (2012). "Rock hyrax middens: a palaeoenvironmental archive for southern African drylands". Quaternary Science Reviews. 56: 107–125. Bibcode:2012QSRv...56..107C. doi:10.1016/j.quascirev.2012.08.018 – via Elsevier Science Direct.
- Leon, B.; Belonje, P.C. (1979). "Calcium, phosphorus and magnesium excretion in the rock hyrax, Procavia capensis". Comparative Biochemistry and Physiology Part A: Physiology. 64: 67–72. doi:10.1016/0300-9629(79)90431-6 – via Elsevier Science Direct.
- Scott, Louis (1994). "Palynology of Late Pleistocene hyrax middens, southwestern Cape Province, South Africa: a preliminary report". Historical Biology. 9 (1–2): 71–81. doi:10.1080/10292389409380489 – via Taylor & Francis Online.
- Prinsloo, L.C., 2007. Rock hyraces: a cause of San rock art deterioration? Journal of Raman Spectroscopy 38, 496-503.
- Carr, Andrew S.; Boom, Arnoud; Chase, Brian M. (2010). "The potential of plant biomarker evidence derived from rock hyrax middens as an indicator of palaeoenvironmental change". Palaeogeography, Palaeoclimatology, Palaeoecology. 285 (3–4): 321–330. Bibcode:2010PPP...285..321C. doi:10.1016/j.palaeo.2009.11.029 – via Elsevier Science Direct.
- Bristow, A.W., Whitehead, D.C., Cockburn, J.E., 1992. Nitrogenous constituents in the urine of cattle, sheep and goats. Journal of the Science of Food and Agriculture 59, 387-394.
- Williams, D.H., Fleming, I., 1989. Spectroscopic Methods in Organic Chemistry. McGraw-Hill International.
- Brittain, H.G., 2009. Vibrational Spectroscopic Studies of Cocrystals and Salts. 1. The Benzamide-Benzoic Acid System. Crystal Growth and Design 9, 2492-2499
- Kniseley, R.N., Fassel, V.A., Farquhar, E.L., Gray, L.S., 1962. Infrared spectra of nitrogen containing compounds I – benzamide. Spectrochimica Acta 18, 1217-1229.
- Fall, P.L., 1990. Deforestation in southern Jordan: evidence from fossil hyrax middens, in: Entjes-Nieborg, G., van Zeist, W. (Eds.), Man's Role in Shaping the Eastern Mediterranean Landscape. Balkema, Rotterdam.
- Fall, P.L., Lindquist, C.A., Falconer, S.E., 1990. Fossil hyrax middens from the Middle East: a record of paleovegetation and human disturbance, in: Betancourt, J.L., Devender, T.R.V., Martin, P.S. (Eds.), Packrat Middens: The Last 40,000 Years of Biotic Change. University of Arizona Press, pp. 408-427
- Pons, A., Quézel, P., 1958. Première remarques sur l'étude palynologique d'un guano fossile du Hoggar. Comptes Rendus des Scéances de L'Académie des Sciences 244, 2290-2292.
- Scott, L., 1990a. Hyrax (Procaviidae) and dassie rat (Petromuridae) middens in palaeoenvironmental studies in Africa, in: Betancourt, J.L., van Devender, T.R., Martin, P.S. (Eds.), Packrat Middens: The Last 40,000 Years of Biotic Change. University of Arizona Press, Tucson, pp. 408-427.
- Scott, L., 1990b. Palynological evidence for late Quaternary environmental change in southern Africa. Palaeoecology of Africa 21, 259-268.
- Scott, L., Bousman, C.B., 1990. Palynological analysis of hyrax middens from Southern Africa. Palaeogeography, Palaeoclimatology, Palaeoecology 76, 367-379.
- Bousman, B., Scott, L., 1994. Climate or overgrazing: the palynological evidence for vegetation change in the eastern Karoo. South African Journal of Science 90, 575-578.
- Scott, Louis; Bousman, C.B.; Nyakale, M. (2005). "Holocene pollen from swamp, cave and hyrax dung deposits at Blydefontein (Kikvorsberge), Karoo, South Africa". Quaternary International. 129 (1): 45–59. Bibcode:2005QuInt.129...49S. doi:10.1016/j.quaint.2004.04.006 – via Elsevier Science Direct.
- Quick, Lynne J.; Chase, Brian M.; Meadows, Michael E.; Scott, Louis; Reimer, Paula J. (2011). "A 19.5 kyr vegetation history from the central Cederberg Mountains, South Africa: Palynological evidence from rock hyrax middens". Palaeogeography, Palaeoclimatology, Palaeoecology. 309 (3–4): 253–270. Bibcode:2011PPP...309..253Q. doi:10.1016/j.palaeo.2011.06.008 – via Elsevier Science Direct.
- Lim, Sophak; Chase, Brian M.; Chevalier, Manuel; Reimer, Paula J. (2016). "50,000 years of vegetation and climate change in the southern Namib Desert, Pella, South Africa". Palaeogeography, Palaeoclimatology, Palaeoecology. 451: 197–209. Bibcode:2016PPP...451..197L. doi:10.1016/j.palaeo.2016.03.001. S2CID 49311191 – via Elsevier Science Direct.
- Valsecchi, Verushka; Chase, Brian M.; Slingsby, Jasper A.; Carr, Andrew S.; Quick, Lynne J.; Meadows, Michael E.; Cheddadi, Rachid; Reimer, Paula J. (2016). "A high resolution 15,600-year pollen and microcharcoal record from the Cederberg Mountains, South Africa". Palaeogeography, Palaeoclimatology, Palaeoecology. 387: 6–16. doi:10.1016/j.palaeo.2013.07.009 – via Elsevier Science Direct.
- Scott, Louis; Gil Romera, Graciela; Marais, Eugene; Brook, George A. (2018). "Pollen in fossil hyrax dung from Marine Isotope Stages 2 and 3 reveals past environments in Namibia". Quaternary International. 464 (Part A): 260–272. Bibcode:2018QuInt.464..260S. doi:10.1016/j.quaint.2017.06.054 – via Elsevier Science Direct.
- Scott, Louis; Gil Romera, Graciela; Marais, Eugene; Brook, George A. (2022). "Holocene environmental change along the central Namib Desert escarpment derived from hyrax and owl dung". Review of Palaeobotany and Palynology. 305. Bibcode:2022RPaPa.30504746S. doi:10.1016/j.revpalbo.2022.104746. S2CID 251509326 – via Elsevier Science Direct.
- Gil-Romera, G., Lamb, H.F., Turton, D., Sevilla-Callejo, M., Umer, M., 2010. Long-term resilience, bush encroachment patterns and local knowledge in a Northeast African savanna. Global Environmental Change 20, 612-626.
- Ivory, S.J., Cole, K.L., Anderson, R.S., Anderson, A., McCorriston, J., 2021. Human landscape modification and expansion of tropical woodland in southern Arabia during the mid-Holocene from rock hyrax middens. Journal of Biogeography 48, 2588-2603.
- Horisk, K.E., Ivory, S.J., McCorriston, J., McHale, M., Al Mehri, A., Anderson, A., Anderson, R.S., Al Kathiri, A.A., 2023. Vegetation dynamics in Dhofar, Oman, from the Late Holocene to present inferred from rock hyrax middens. Quaternary Research, 1-18.
- Horowitz, A., 1992. Palynology of Arid Lands. Elsevier, Amsterdam.
- Gil Romera, Graciela; Scott, Louis; Marais, Eugene; Brook, George A. (2007). "Late Holocene environmental change in the northwestern Namib Desert margin: new fossil pollen evidence from hyrax middens". Palaeogeography, Palaeoclimatology, Palaeoecology. 249 (1–2): 1–17. Bibcode:2007PPP...249....1G. doi:10.1016/j.palaeo.2007.01.002.
- Scott, Louis; Cooremans, B. (1992). "Pollen in Recent Procavia (Hyrax), Petromus (Dassie Rat) and Bird Dung in South Africa". Journal of Biogeography. 19 (2): 205–215. doi:10.2307/2845506. JSTOR 2845506 – via JSTOR.
- Hubbard, Richard N.L.B.; Sampson, C. Garth (1993). "Rainfall estimates derived from the pollen content of modern hyrax dung: an evaluation". South African Journal of Science. 89: 199–204.
- Carrion, Jose S.; Scott, Louis; Vogel, John C. (1999). "Twentieth century changes in montane vegetation in the eastern Free State, South Africa, derived from palynology of hyrax dung middens". Journal of Quaternary Science. 14 (1): 1–16. doi:10.1002/(SICI)1099-1417(199902)14:1<1::AID-JQS412>3.0.CO;2-Y – via Wiley Online Library.
- Scott, Louis; Vogel, John C. (2000). "Evidence for environmental conditions during the last 20 000 years in Southern Africa from 13C in fossil hyrax dung". Global and Planetary Change. 26 (1–3): 207–215. Bibcode:2000GPC....26..207S. doi:10.1016/S0921-8181(00)00045-X.
- Meadows, Michael E.; Chase, Brian M.; Seliane, Mamo (2010). "Holocene palaeoenvironments of the Cederberg and Swartruggens mountains, Western Cape, South Africa: Pollen and stable isotope evidence from hyrax dung middens". Journal of Arid Environments. 74 (7): 786–793. Bibcode:2010JArEn..74..786M. doi:10.1016/j.jaridenv.2009.04.020 – via Elsevier Science Direct.
- Heaton, T.H.E., Vogel, J.C., von la Chevallerie, G., Collet, G., 1986. Climate influence on the isotopic composition of bone nitrogen. Nature 322, 822-823.
- Ambrose, S.H., DeNiro, M.J., 1986. The isotopic ecology of East African mammals. Oecologia 69, 395-406.
- Ambrose, S.H., 1991. Effects of diet, climate and physiology on nitrogen isotope abundances in terrestrial foodwebs. Journal of Archaeological Science 18, 293-317.
- Schoeninger, M.J., DeNiro, M.J., 1984. Nitrogen and carbon isotopic composition of bone collagen from marine and terrestrial animals. Geochimica et Cosmochimica Acta 48, 625-639.
- Ambrose, S.H., DeNiro, M.J., 1986. Reconstruction of African human diet using bone collagen carbon and nitrogen isotope ratios. Nature 319, 321-324.
- Sealy, J.C., van der Merwe, N.J., Thorp, J.A.L., Lanham, J.L., 1987. Nitrogen isotopic ecology in southern Africa: implications for environmental and dietary tracing. Geochimica et Cosmochimica Acta 51, 2707-2717.
- Aranibar, J.N., Otter, L., Macko, S.A., Feral, C.J.W., Epstein, H.E., Dowty, P.R., Eckardt, F., Shugart, H.H., Swap, R.J., 2004. Nitrogen cycling in the soil-plant system along a precipitation gradient in the Kalahari sands. Global Change Biology 10, 359-373.
- Codron, D., Codron, J., 2009. Reliability of d13C and d15N in faeces for reconstructing savanna herbivore diet. Mammalian Biology - Zeitschrift fur Saugetierkunde 74, 36-48.
- Handley, L.L., Austin, A.T., Stewart, G.R., Robinson, D., Scrimgeour, C.M., Raven, J.A., Heaton, T.H.E., Schmidt, S., 1999. The 15N natural abundance (d15N) of ecosystem samples reflects measures of water availability. Functional Plant Biology 26, 185-199.
- Schwarcz, H.P., Dupras, T.L., Fairgrieve, S.I., 1999. 15N enrichment in the Sahara: in search of a global relationship. Journal of Archaeological Science 26, 629-636.
- Swap, R.J., Aranibar, J.N., Dowty, P.R., Gilhooly, W.P., Macko, S.A., 2004. Natural abundance of 13C and 15N in C3 and C4 vegetation of southern Africa: patterns and implications. Global Change Biology 10, 350-358.
- Schade, J.D., Hobbie, S.E., 2005. Spatial and temporal variation in islands of fertility in the Sonoran Desert. Biogeochemistry 73, 541-553.
- Murphy, B.P., Bowman, D.M.J.S., 2006. Kangaroo metabolism does not cause the relationship between bone collagen d15N and water availability. Functional Ecology 20, 1062-1069.
- Murphy, B.P., Bowman, D.M.J.S., 2009. The carbon and nitrogen isotope composition of Australian grasses in relation to climate. Functional Ecology 23, 1040-1049.
- Sponheimer, M., Robinson, T.F., Roeder, B.L., Passey, B.H., Ayliffe, L.K., Cerling, T.E., Dearing, M.D., Ehleringer, J.R., 2003b. An experimental study of nitrogen flux in llamas: is 14N preferentially excreted? Journal of Archaeological Science 30, 1649-1655.