Ruth Clayton
Ruth Clayton (née Freedman;10 June 1925 – 2003) was a lecturer and researcher with an international reputation in eye research at the University of Edinburgh in the Institute of Animal Genetics.[1] She was one of a group of scientists who joined the Institute at a time when it was led by C.H. Waddington and when many of the fundamental aspects of modern biology were being elucidated by forward thinking scientists at the university's King's Buildings campus.[2][3] Leading a large and diverse research group, she applied the newly developing techniques of modern biology to fundamental questions in the fields of developmental biology and pathology of the eye and the brain. Her work was characterised by a rigorously conceptual approach, methodological innovation and a keen interest in the social and ethical implications of scientific and medical research.[1]
Ruth Clayton | |
|---|---|
 | |
| Born | Ruth Moira Freedman 10 June 1925 London, England |
| Died | 11 January 2003 (aged 77) Edinburgh, Scotland |
| Nationality | British |
| Occupation(s) | University lecturer and researcher (reader) |
| Academic background | |
| Alma mater | University of Oxford (MA, Zoology) |
| Academic work | |
| Discipline | Genetics |
| Sub-discipline | Eye Research, embryology, epigenetics, molecular and cellular biology, pathology |
| Main interests | Differentiation and transdifferentiation of ocular tissues, extralenticular crystallins, risk factors for cataract, behavioural teratogenesis |
Education and early life
Clayton was born in London on 10 June 1925. She studied zoology at the University of Oxford, gaining an MA.[1]
Research and career
Institute of Animal Genetics
Clayton joined the Institute of Animal Genetics (University of Edinburgh) in 1947 at a time of rapid expansion under the leadership of the biologist and polymath C.H. Waddington.[1] She was one of the original group of scientists there during the 1950s that included notable figures such as Charlotte Auerbach, Geoffrey Beale, Douglas Scott Falconer, Henrik Kacser, Eric Lucey, Eric Reeve, and Alan Robertson. Waddington's initial solution to staff accommodation problems in Edinburgh was the acquisition of the nearby Mortonhall and an experiment in collegiate living, from which Eric Reeve's wife, the artist Edith Simon drew inspiration for her novel The Past Masters (titled House of Strangers in the USA).[4] In 1965, the Medical Research Council and Wellcome Foundation funded the building of a modern tower block, adjacent to the main Institute of Animal Genetics Crew Building on the university's King's Buildings campus to accommodate the research being undertaken by Waddington and his colleagues.[5] By the 1970s, Clayton, DES Truman, John Campbell, Geoffrey Selman, Joe Jacob, John Bishop, Ken Jones and their research groups occupied the tower, all pursuing different aspects of an explicitly epigenetic research programme.[6] When Ruth Clayton's group outgrew the accommodation available in the tower, she refurbished and occupied the first floor of the Crew Building. She retired in 1993 with Emeritus Reader status.[1]
Epigenetics and the eye

Epigenetics in its very broadest sense (and that originally intended by Waddington) is the study of the processes by which the information encoded in genes (the genotype) becomes manifest (is expressed) in the observable characteristics (the phenotype) of an individual during development. The eye in general and in particular the abundant proteins of the eye lens, which are termed crystallins provided Clayton and her colleague DES Truman with an ideal system in which to investigate the link between gene expression and cell differentiation during embryonic development. The complexity of crystallin protein composition and expression were explored by early adoption of a wide range of innovative immunological,[7][8][9] biochemical,[10][11][12][13] molecular, and cellular[14][15][16][17][18] techniques then being developed in Edinburgh and elsewhere. On the basis of these and other studies she developed and advocated the view that the differences in gene expression that underlie cell differentiation are quantitative in nature and combinatorial in effect.[9][19][20] This nuanced view contrasted markedly with the prevailing ideas at that time of a strict demarcation between “house-keeping" and “luxury" gene products, strictly tissue-specific expression of luxury gene products and a single unitary function for individual proteins.[21] Crystallin gene expression was further explored in a detailed series of studies using long-term cell culture models in which progenitor chick lens cells continuously differentiated into lens fibre cells in controlled laboratory conditions. The intrinsic programme of crystallin gene expression during this process was found to be non-coordinate within and between crystallin protein classes, tightly controlled and resembled the developmental programme seen in the animal itself,[22][23] but it could be modified by age, genotype, cellular growth rate, and soluble factors.[24][25][26][27][28] All of this indicated that the epigenetic programme was intrinsic to the cells, but it could be modified by external and internal variables.
Differentiation and transdifferentiation of ocular tissues

The epigenetic landscape model of Waddington's proposed that development proceeds by progressive restriction of cell fate, commitment and subsequent differentiation to a range of well-defined cell types. Surprisingly, embryonic chick retinal cells can also differentiate into lens cells under certain conditions when grown isolated in culture; a rare example of the transdifferentiation of one differentiated cell type into another, and a phenomenon that is thought to relate to the capacity of certain species to undergo lens regeneration. Clayton's work showed that although transdifferentiation potential diminishes with age,[29][30] it involves the expression of authentic lens crystallins[17][31][32][33] according to a precise programme that differs in sequence from that seen during lens differentiation.[33][34] The potential for transdifferentiation itself being proposed to be related to the prior low-level expression of lens crystallins.[35][36][37] From a contemporary perspective, what once appeared to some as an experimental curiosity (ocular transdifferentiation) now seems to represent an early recognised example of the kind of cellular reprogramming that underlies contemporary induced pluripotent stem cell technology and its hoped-for application as truly regenerative medicine.
Extralenticular crystallins
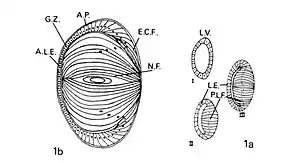
The contention that extralenticular (non-lens) crystallin expression was biologically significant initially attracted the criticisms that it was either an experimental artefact (perhaps resulting from inadvertent lens tissue contamination) or that it represented an example of non-functional (“leaky") gene expression. Definitive demonstration of expression of individual crystallin genes in tissues other than lens, and indeed outside of the eye in several vertebrate species by Clayton's group[15][17][18][33][38][39] and other international research groups led to a reversal of this view. A slew of papers ensued on the expression and putative functions of extralenticular crystallins, and specifically their roles in nervous system diseases. Crystallin sequence similarities with, or in some cases their identity to particular enzymes or heat shock proteins[40][41] led to the concept of gene sharing (or protein moonlighting) as fully developed by Joram Piatigorsky.[42] The abundant water-soluble proteins of the vertebrate lens (by definition, crystallins) are therefore now generally considered to be examples of multifunctional proteins with their precise functions being context- and concentration-dependant, consistent with a combinatorial and quantitative role of gene expression in cell differentiation.
Risk factors for cataract
Clayton's lens research encompassed both studies of normal and abnormal development, and studies of aging, in particular research on the most common cause of blindness world-wide, namely cataract. A direct approach was employed in which candidate risk factors for cataract were investigated in a large-scale case-controlled study correlating clinical, pathological and biochemical data from cataract patients in South East Scotland. Specific risk factors were identified included the levels of particular blood plasma constituents,[43] coexisting clinical (and sub-clinical) disease and medication[43][44][45] and pattern of alcohol use.[46] These studies showed that risk factors were most likely cumulative[47] and that some risk factors correlated with particular cataract types.[48] All of which indicates that so called senile cataract is not a simple function of aging, but more likely has a complex epidemiology, and one in which lens transparency is affected by a life-long accumulation of quantitative and potentially interacting risk factors, both medical and socio-economic.[47]
Behavioural teratogenesis
Later in her career, Clayton pursued a series of studies in collaboration with University of Edinburgh zoologist Aubrey Manning. Rodent models and cell cultures were used to assess the potentially deleterious effects on foetal neural development of low-level exposure to pharmaceuticals and environmental contaminants during pregnancy. They found that common anticonvulsant drugs and aluminium sulfate had measurable neurochemical and behavioural effects on pups, dams or pup-dam interactions following low-level pre-natal exposure, and that some of these changes were modified by genetics.[49][50][51][52]
Personal life
Clayton was married to geneticist George Clayton. She had four children; Anthony (a professor of sustainable development), Christopher (a physicist), Kate (a translator), and Paul (a nutrition scientist). Latterly, Clayton worked as a councillor and wrote on the ethical implications of genetics.[53] Throughout her life Clayton pursued an active interest in progressive politics and the arts, including speculative fiction and was a talented painter.[54]
References
- Truman, DES. Obituaries: Ruth Clayton. Bulletin, The University of Edinburgh News. April/May 2002/03. No.4, p14. Staff news pre-2009. eBulletin Archive. February 2003. Obituaries. Ruth Clayton.
- Martynoga, B. Molecular Tinkering: The Edinburgh scientists who changed the face of modern biology. 2018. Matador Publishing Limited, Kibworth Beauchamp, UK. ISBN 978 1789014 273
- Towards Dolly: Edinburgh, Roslin and the birth of modern genetics. .
- Simon, E. The House of Strangers. 1953. G.P. Putnam's Sons, New York, USA.
- A sense of place. Towards Dolly: Edinburgh, Roslin and the birth of modern genetics. 2013.
- Falconer D. Quantitative genetics in Edinburgh: 1947–1980. Genetics. 1993 Feb;133(2):137-42. PMID 8436263 PMCID: PMC1205304
- Clayton RM. Localization of embryonic antigens by antisera labelled with fluorescent dyes. Nature. 1954 Dec 4;174(4440):1059. PubMed PMID 13214077
- Clayton RM, Truman DE. Molecular structure and antigenicity of lens proteins. Nature. 1967 Jun 17;214(5094):1201-4. PubMed PMID 4965206.
- Clayton RM, Campbell JC, Truman DE. A re-examination of the organ specificity of lens antigens. Exp Eye Res. 1968 Jan;7(1):11-29. PubMed PMID 4966535
- Clayton RM, Truman DE, Campbell JC. A method for direct assay of messenger RNA turnover for different crystallins in the chick lens. Cell Differ. 1972 Apr;1(1):25-35. PubMed PMID 4670874
- Clayton RM, Truman DE. The antigenic structure of chick beta-crystallin subunits. Exp Eye Res. 1974 May;18(5):495-506. PubMed PMID 4834401.
- Truman DE, Clayton RM. The subunit structure of chick beta-crystallins. Exp Eye Res. 1974 May;18(5):485-94. PubMed PMID 4834400.
- Clayton RM, Truman DE, Hannah AI. RNA turnover and translational regulation of specific crystallin synthesis. Cell Differ. 1974 Sep;3(3):135-45. PubMed PMID 4415666.
- Jackson JF, Clayton RM, Williamson R, Thomson I, Truman DE, de Pomerai DI. Sequence complexity and tissue distribution of chick lens crystallin mRNAs. Dev Biol. 1978 Aug;65(2):383-95. PubMed PMID 680368.
- Bower DJ, Errington LH, Wainwright NR, Sime C, Morris S, Clayton RM. Cytoplasmic RNA sequences complementary to cloned chick delta-crystallin cDNA show size heterogeneity. Biochem J. 1982 Feb 1;201(2):339-44. PubMed PMID 6282266; PubMed Central PMCID: PMC1163648.
- Errington LH, Cooper DN, Clayton RM. The pattern of DNA methylation in the delta-crystallin genes in transdifferentiating neural retina cultures. Differentiation. 1983;24(1):33-8. PubMed PMID 6307794
- Bower DJ, Errington LH, Pollock BJ, Morris S, Clayton RM. The pattern of expression of chick delta-crystallin genes in lens differentiation and in trans-differentiating cultured tissues. EMBO J. 1983;2(3):333-8. PubMed PMID 11894946; PubMed Central PMCID: PMC555137.
- Jeanny JC, Bower DJ, Errington LH, Morris S, Clayton RM. Cellular heterogeneity in the expression of the delta-crystallin gene in non-lens tissue. Dev Biol. 1985 Nov;112(1):94-9. PubMed PMID 4074468.
- Clayton RM, Truman DE, Bird AP. Quantitative regulation. Adv Exp Med Biol. 1982;158:327-9. PubMed PMID 7158545
- Bird AP, Truman DE, Clayton RM. The molecular basis of differentiation and competence. Adv Exp Med Biol. 1982;158:61-4. PubMed PMID 7158553.
- Truman DES. The Biochemistry of Cytodifferentiation. 1974. Blackwell Scientific Publications, Oxford, London, Edinburgh, Melbourne. ISBN 0 632 09110 X
- Patek CE, Clayton RM. A comparison of the changing patterns of crystallin expression in vivo, in long-term primary cultures in vitro and in response to a carcinogen. Exp Eye Res. 1985 Mar;40(3):357-78. PubMed PMID 4065232.
- Patek CE, Clayton RM. Alterations in crystallin gene expression during subculture of chick lens cells. Exp Eye Res. 1986 Oct;43(4):595-606. PubMed PMID 3792462.
- Patek CE, Clayton RM. Patterns of crystallin expression during differentiation in vitro of several chick genotypes with different effects on lens cell growth rate. Exp Eye Res. 1986 Dec;43(6):1111-26. PubMed PMID 3817027.
- Patek CE, Clayton RM. The influence of the genotype on the process of ageing of chick lens cells in vitro. Exp Cell Res. 1988 Feb;174(2):330-43. PubMed PMID 3338493.
- Patek CE, Clayton RM. Age-related changes in the response of chick lens cells during long-term culture to insulin, cyclic AMP, retinoic acid and a bovine retinal extract. Exp Eye Res. 1990 Apr;50(4):345-54. PubMed PMID 2159887.
- Clayton RM, Patek CE, Head MW, Cuthbert J. Ageing in the chick lens: in vitro studies. Mutat Res. 1991 Mar-Nov;256(2-6):203-20. Review. PubMed PMID 1722011.
- Patek C, Head M, Clayton R. Effects of age and genetic growth rate on the crystallin composition of the chick lens. Int J Dev Biol. 1994 Dec;38(4):717-24. PubMed PMID 7779693.
- De Pomerai DI, Pritchard DJ, Clayton RM. Biochemical and immunological studies of lentoid formation in cultures of embryonic chick neural retina and day-old chick lens epithelium. Dev Biol. 1977 Oct 15;60(2):416-27. PubMed PMID 336440.
- De Pomerai DI, Clayton RM. Influence of embryonic stage on the transdifferentiation of chick neural retina cells in culture. J Embryol Exp Morphol. 1978 Oct;47:179-93. PubMed PMID 722231.
- Thomson I, Wilkinson CE, Jackson JF, de Pomerai DI, Clayton RM, Truman DE, Williamson R. Isolation and cell-free translation of chick lens crystallin mRNA during normal development and transdifferentiation of neural retina. Dev Biol. 1978 Aug;65(2):372-82. PubMed PMID 680367.
- Errington LH, Bower J, Cuthbert J, Clayton RM. The expression of chick alpha A2-crystallin RNA during lens development and transdifferentiation. Biol Cell. 1985;54(2):101-8. PubMed PMID 2933101.
- Head MW, Peter A, Clayton RM. Evidence for the extralenticular expression of members of the beta-crystallin gene family in the chick and a comparison with delta-crystallin during differentiation and transdifferentiation. Differentiation. 1991 Dec;48(3):147-56. PubMed PMID 1725161.
- Patek CE, Jeanny JC, Clayton RM. Changes in crystallin expression during transdifferentiation and subsequent ageing of embryonic chick neural retina in vitro: comparison with lens epithelium. Exp Eye Res. 1993 Nov;57(5):527-37. PubMed PMID 8282039.
- Clayton RM, Thomson I, de Pomerai DI. Relationship between crystallin mRNA expression in retina cells and their capacity to re-differentiate into lens cells. Nature. 1979 Dec 6;282(5739):628-9. PubMed PMID 551295.
- Clayton RM. The molecular basis for competence, determination and transdifferentiation: a hypothesis. Adv Exp Med Biol. 1982;158:23-38. PubMed PMID 6186130.
- Clayton RM, Jeanny JC, Bower DJ, Errington LH. The presence of extralenticular crystallins and its relationship with transdifferentiation to lens. Curr Top Dev Biol. 1986;20:137-51. Review. PubMed PMID 2420533.
- Head MW, Triplett EL, Clayton RM. Independent regulation of two coexpressed delta-crystallin genes in chick lens and nonlens tissues. Exp Cell Res. 1991 Apr;193(2):370-4. PubMed PMID 2004651.
- Head MW, Sedowofia K, Clayton RM. Beta B2-crystallin in the mammalian retina. Exp Eye Res. 1995 Oct;61(4):423-8. PubMed PMID 8549683.
- Wistow G, Piatigorsky J. Recruitment of enzymes as lens structural proteins. Science. 1987 Jun 19;236(4808):1554-6. PMID 3589669.
- de Jong WW, Hendriks W, Mulders JW, Bloemendal H. Evolution of eye lens crystallins: the stress connection. Trends Biochem Sci. 1989 Sep;14(9):365-8. Review. PMID 2688200.
- Piatigorsky, J. Gene Sharing and Evolution: The diversity of protein function. 2007. Harvard University Press, Cambridge, Massachusetts, USA. ISBN 978-0-674-02341-3.
- Donnelly CA, Seth J, Clayton RM, Phillips CI, Cuthbert J, Prescott RJ. Some blood plasma constituents correlate with human cataract. Br J Ophthalmol. 1995 Nov;79(11):1036-41. PMID 8534650 PMCID: PMC505324 DOI: 10.1136/bjo.79.11.1036.
- Cuthbert J, Clayton RM, Phillips CI, Seth J. Diuretic drugs as risk factors in cataractogenesis. Metab Pediatr Syst Ophthalmol (1985). 1987;10(2):48-54. PMID 2908208.
- Phillips CI, Donnelly CA, Clayton RM, Cuthbert J. Skin disease and age-related cataract. Acta Derm Venereol. 1996 Jul;76(4):314-8. PMID 8869693 DOI: 10.2340/0001555576314318.
- Phillips CI, Clayton RM, Cuthbert J, Qian W, Donnelly CA, Prescott RJ. Human cataract risk factors: significance of abstention from, and high consumption of, ethanol (U-curve) and non-significance of smoking. Ophthalmic Res. 1996;28(4):237-47. PMID 8878187 DOI: 10.1159/000267909.
- Clayton RM, Cuthbert J, Seth J, Phillips CI, Bartholomew RS, Reid JM. Epidemiological and other studies in the assessment of factors contributing to cataractogenesis. Ciba Found Symp. 1984;106:25-47. PMID 6568978.
- Donnelly CA, Seth J, Clayton RM, Phillips CI, Cuthbert J. Some plasma constituents correlate with human cataract location and nuclear colour. Ophthalmic Res. 1997;29(4):207-17. PMID 9261844 DOI: 10.1159/000268015.
- Sedowofia SK, Clayton RM. Effects of anticonvulsant drugs on brain cultures from chick embryos: a comparison with cultures from embryos treated in ovo. Teratog Carcinog Mutagen. 1985;5(3):205-17.PMID 2866603.
- Sedowofia SK, Innes J, Peter A, Alleva E, Manning A, Clayton RM. Differential effects of prenatal exposure to phenobarbital on the behaviour and neurochemistry of CBA and C57BL/6J mice. Psychopharmacology (Berl). 1989;97(1):123-30. PubMed PMID 2496418.
- Clayton RM, Sedowofia SK, Rankin JM, Manning A. Long-term effects of aluminium on the fetal mouse brain. Life Sci. 1992;51(25):1921-8. PubMed PMID 1453876.
- Laviola G, Terranova ML, Sedowofia K, Clayton R, Manning A. A mouse model of early social interactions after prenatal drug exposure: a genetic investigation. Psychopharmacology (Berl). 1994 Jan;113(3-4):388-94. PMID 7862850
- Clayton R. Genetic Intervention in human subjects. Journal of Medical Ethics. 1997 23 (6):385-386.
- Clare Button Pecha Kucha. University of Edinburgh Innovative Learning Week. 2014.