Side chain
In organic chemistry and biochemistry, a side chain is a chemical group that is attached to a core part of the molecule called the "main chain" or backbone. The side chain is a hydrocarbon branching element of a molecule that is attached to a larger hydrocarbon backbone. It is one factor in determining a molecule's properties and reactivity.[2] A side chain is also known as a pendant chain, but a pendant group (side group) has a different definition.
Branch
Side-chain
Pendant chain
An oligomeric or polymeric offshoot from a macromolecular chain.Notes
- An oligomeric branch may be termed a short-chain branch.
- A polymeric branch may be termed a long-chain branch.[1]
Conventions
The placeholder R is often used as a generic placeholder for alkyl (saturated hydrocarbon) group side chains in chemical structure diagrams. To indicate other non-carbon groups in structure diagrams, X, Y, or Z are often used.
History
The R symbol was introduced by 19th-century French chemist Charles Frédéric Gerhardt, who advocated its adoption on the grounds that it would be widely recognizable and intelligible given its correspondence in multiple European languages to the initial letter of "root" or "residue": French racine ("root") and résidu ("residue"), these terms' respective English translations along with radical (itself derived from Latin radix below), Latin radix ("root") and residuum ("residue"), and German Rest ("remnant" and, in the context of chemistry, both "residue" and "radical").[3]
Usage
Organic chemistry
In polymer science, the side chain of an oligomeric or polymeric offshoot extends from the backbone chain of a polymer. Side chains have noteworthy influence on a polymer's properties, mainly its crystallinity and density. An oligomeric branch may be termed a short-chain branch, and a polymeric branch may be termed a long-chain branch. Side groups are different from side chains; they are neither oligomeric nor polymeric.[4]
Biochemistry
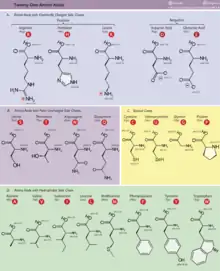
In proteins, which are composed of amino acid residues, the side chains are attached to the alpha-carbon atoms of the amide backbone. The side chain connected to the alpha-carbon is specific for each amino acid and is responsible for determining charge and polarity of the amino acid. The amino acid side chains are also responsible for many of the interactions that lead to proper protein folding and function.[5] Amino acids with similar polarity are usually attracted to each other, while nonpolar and polar side chains usually repel each other. Nonpolar/polar interactions can still play an important part in stabilizing the secondary structure due to the relatively large amount of them occurring throughout the protein.[6] Spatial positions of side-chain atoms can be predicted based on protein backbone geometry using computational tools for side-chain reconstruction.[7]
See also
References
- Jenkins, A. D.; Kratochvíl, P.; Stepto, R. F. T.; Suter, U. W. (1996). "Glossary of basic terms in polymer science (IUPAC Recommendations 1996)" (PDF). Pure and Applied Chemistry. 68 (12): 2287–2311. doi:10.1351/pac199668122287. S2CID 98774337.
- Wade, L.G. (2010). Organic Chemistry, 7th Edition. Upper Saddle River, NJ: Pearson Prentice Hall. pp. 70–78. ISBN 978-0-321-59231-6.
- Jensen W.B., Journal of Chemical Education 87, 360 (2010)
- Chemistry, International Union of Pure and Applied. IUPAC Compendium of Chemical Terminology. doi:10.1351/goldbook.B00720.
{{cite book}}:|website=ignored (help) - Voet, Donald; Voet, Judith; Pratt, Charlotte (2013). Fundamentals of Biochemistry: Life at the Molecular Level (Fourth ed.). Hoboken, NJ: John Wiley & Sons, Inc. ISBN 9781118129180.
- Andrew, C. D.; Penel, S.; Jones, G. R.; Doig, A. J. (2001-12-01). "Stabilizing nonpolar/polar side-chain interactions in the alpha-helix". Proteins. 45 (4): 449–455. doi:10.1002/prot.1161. ISSN 0887-3585. PMID 11746692. S2CID 25739520.
- Badaczewska-Dawid, Aleksandra E.; Kolinski, Andrzej; Kmiecik, Sebastian (2019-12-26). "Computational reconstruction of atomistic protein structures from coarse-grained models". Computational and Structural Biotechnology Journal. 18: 162–176. doi:10.1016/j.csbj.2019.12.007. ISSN 2001-0370. PMC 6961067. PMID 31969975.