Sodium chloride (data page)
This page provides supplementary chemical data on sodium chloride.
Material safety data sheet
The handling of this chemical may incur notable safety precautions. It is highly recommended that you seek the material safety data sheet (MSDS) for this chemical from a reliable source such as eChemPortal, and follow its direction.
Structure and properties
| Structure and properties | |
|---|---|
| Index of refraction, nD | 1.5442 |
| Abbe number | ? |
| Dielectric constant, | 6.12 at 17–22 °C |
| Bond strength | ? |
| Bond length | ? |
| Bond angle | ? |
| Magnetic susceptibility | −30.3×10−6 cgs |
Thermodynamic properties
| Phase behavior | |
|---|---|
| Triple point | 1074 K (801 °C), 30 Pa |
| Critical point | 3900 K (3600 °C), 26×106 Pa |
| Std enthalpy change of fusion, ΔfusH |
27.95 kJ/mol (0.52 kJ/g)[1][2] |
| Std entropy change of fusion, ΔfusS |
26.02 J/(mol·K) |
| Std enthalpy change of vaporization, ΔvapH |
? kJ/mol |
| Std entropy change of vaporization, ΔvapS |
? J/(mol·K) |
| Solid properties | |
| Std enthalpy change of formation, ΔfH |
−411.12 kJ/mol[2] |
| Standard molar entropy, S |
72 J/(mol·K) |
| Heat capacity, cp | 50 J/(mol·K) ; 0.853 J/(g·k)[2] |
| Liquid properties | |
| Std enthalpy change of formation, ΔfH |
−385.92 kJ/mol |
| Standard molar entropy, S |
95.06 J/(mol·K) |
| Density | 1.549 g/cm3,[2] at 850 °C |
| Heat capacity, cp | ? J/(mol·K) |
| Boiling point | 1465 °C[2] |
| Gas properties | |
| Std enthalpy change of formation, ΔfH |
−181.42 kJ/mol |
| Standard molar entropy, S |
229.79 J/(mol·K) |
| Heat capacity, cp | ? J/(mol·K) |
Density data of aqueous solutions
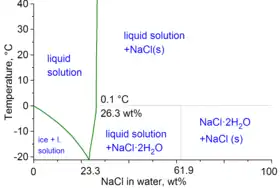
Water–NaCl phase diagram
| NaCl, wt% | Teq, °C | ρ, g/cm3 | n | η, mPa·s |
|---|---|---|---|---|
| 0 | 0 | 0.99984 | 1.333 | 1.002 |
| 0.5 | −0.3 | 1.0018 | 1.3339 | 1.011 |
| 1 | −0.59 | 1.0053 | 1.3347 | 1.02 |
| 2 | −1.19 | 1.0125 | 1.3365 | 1.036 |
| 3 | −1.79 | 1.0196 | 1.3383 | 1.052 |
| 4 | −2.41 | 1.0268 | 1.34 | 1.068 |
| 5 | −3.05 | 1.034 | 1.3418 | 1.085 |
| 6 | −3.7 | 1.0413 | 1.3435 | 1.104 |
| 7 | −4.38 | 1.0486 | 1.3453 | 1.124 |
| 8 | −5.08 | 1.0559 | 1.347 | 1.145 |
| 9 | −5.81 | 1.0633 | 1.3488 | 1.168 |
| 10 | −6.56 | 1.0707 | 1.3505 | 1.193 |
| 12 | −8.18 | 1.0857 | 1.3541 | 1.25 |
| 14 | −9.94 | 1.1008 | 1.3576 | 1.317 |
| 16 | −11.89 | 1.1162 | 1.3612 | 1.388 |
| 18 | −14.04 | 1.1319 | 1.3648 | 1.463 |
| 20 | −16.46 | 1.1478 | 1.3684 | 1.557 |
| 22 | −19.18 | 1.164 | 1.3721 | 1.676 |
| 23.3 | −21.1 | |||
| 23.7 | −17.3 | |||
| 24.9 | −11.1 | |||
| 26.1 | −2.7 | |||
| 26.28 | 0 | |||
| 26.32 | 10 | |||
| 26.41 | 20 | |||
| 26.45 | 25 | |||
| 26.52 | 30 | |||
| 26.67 | 40 | |||
| 26.84 | 50 | |||
| 27.03 | 60 | |||
| 27.25 | 70 | |||
| 27.5 | 80 | |||
| 27.78 | 90 | |||
| 28.05 | 100 |
Note: ρ is density, n is refractive index at 589 nm, and η is viscosity, all at 20 °C; Teq is the equilibrium temperature between two phases: ice/liquid solution for Teq < 0–0.1 °C and NaCl/liquid solution for Teq above 0.1 °C.
Spectral data
| UV-Vis | |
|---|---|
| λmax | ? nm |
| Extinction coefficient, ε | ? |
| IR | |
| Major absorption bands | ? cm−1 |
| NMR | |
| Proton NMR | |
| Carbon-13 NMR | |
| Other NMR data | |
| MS | |
| Masses of main fragments |
|
This box:
- Except where noted otherwise, data relate to Standard temperature and pressure.
- Reliability of data general note.
References
- "NaCl CID 5234, 4.2.14 Other Experimental Properties"
- "NaCl Other Chemical/Physical Properties"
- Lide, D. R., ed. (2005). CRC Handbook of Chemistry and Physics (86th ed.). Boca Raton (FL): CRC Press. pp. 8–71, 8–116. ISBN 0-8493-0486-5.
External links
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.