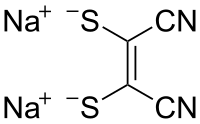
Sodium maleonitriledithiolate
Sodium maleonitriledithiolate is the chemical compound described by the formula Na2S2C2(CN)2. The name refers to the cis compound, structurally related to maleonitrile ((CHCN)2). Maleonitriledithiolate is often abbreviated mnt. It is a "dithiolene", i.e. a chelating alkene-1,2-dithiolate. It is a prototypical non-innocent ligand in coordination chemistry. Several complexes are known, such as [Ni(mnt)2]2−.[2]: 143–146
2Ni(mnt)2.jpg.webp)
Sample of (Et4N)2Ni(mnt)2.
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Disodium (Z)-1,2-dicyanoethene-1,2-bis(thiolate) | |
| Other names
sodium mnt sodium maleonitriledithiolate | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
| |
| |
| Properties | |
| C4N2Na2S2 | |
| Molar mass | 186.17 g/mol |
| Appearance | yellow solid |
| Solubility in ethanol, DMF | Soluble |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
The salt is synthesized by treating carbon disulfide with sodium cyanide to give the cyanodithioformate salt, which eliminates elemental sulfur in aqueous solution:[3]
- 2 NaCN + 2 CS2 → Na2S2C2(CN)2 + 1/4 S8
The compound was first described by Bähr and Schleitzer 1958.[4]
References
- Chem Sources U.S.A. Directories Publishing Company, Incorporated. 2001. p. 535. ISBN 978-0-937020-34-0.
- Day, P. and Coronado, E. (2004) Molecular Materials Combining Magnetic and Conducting Properties, in Magnetism: Molecules to Materials V (eds J. S. Miller and M. Drillon), Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, FRG. doi: 10.1002/3527604383.ch4
- R. H. Holm, A. Davison "Metal Complexes Derived from cis-1,2-Dicyano-1,2-Ethylenedithiolate and Bis(trifluoromethyl)-1,2-Dithiete" Inorganic Syntheses 1967, volume X, pp.8-26.
- G. Bähr and G. Schleitzer (1957). "Beiträge zur Chemie des Schwefelkohlenstoffs und Selenkohlenstoffs, II. Die Kondensierende Spontan-Entschwefelung von Salzen und Estern der Cyan-Dithioameisensäure. Freie Cyan-Dithioameisensäure". Chemische Berichte. 90 (3): 438–443. doi:10.1002/cber.19570900322.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.