Sperm chemotaxis
Sperm chemotaxis is a form of sperm guidance, in which sperm cells (spermatozoa) follow a concentration gradient of a chemoattractant secreted from the oocyte and thereby reach the oocyte.
Background
Since the discovery of sperm attraction to the female gametes in ferns over a century ago,[1] sperm guidance in the form of sperm chemotaxis has been established in a large variety of species[2] Although sperm chemotaxis is prevalent throughout the Metazoa kingdom, from marine species with external fertilization such as sea urchins and corals, to humans,[2][3][4] much of the current information on sperm chemotaxis is derived from studies of marine invertebrates, primarily sea urchin and starfish.[5] As a matter of fact, until not too long ago, the dogma was that, in mammals, guidance of spermatozoa to the oocyte was unnecessary. This was due to the common belief that, following ejaculation into the female genital tract, large numbers of spermatozoa 'race' towards the oocyte and compete to fertilize it.
Research during the 1980s[6] caused this belief to be taken apart when it became clear that only few of the ejaculated spermatozoa — in humans, only ~1 of every million spermatozoa — succeed in entering the oviducts (Fallopian tubes)[4][7] and when more recent studies showed that mammalian spermatozoa do respond chemotactically.[8]
Sperm chemotaxis in non-mammalian species
In sperm chemotaxis, the oocyte secretes a chemoattractant, which, as it diffuses away, forms a concentration gradient: a high concentration close to the egg, and a gradually lower concentration as the distance from the oocyte increases. Spermatozoa can sense this chemoattractant and orient their swimming direction up the concentration gradient towards the oocyte. Sperm chemotaxis was demonstrated in a large number of non-mammalian species, from marine invertebrates[2][3] to frogs.[9]
Chemoattractants
The sperm chemoattractants in non-mammalian species vary to a large extent. Some examples are shown in Table 1. So far, most sperm chemoattractants that have been identified in non-mammalian species are peptides or low-molecular-weight proteins (1–20 kDa), which are heat stable and sensitive to proteases.[2][3] Exceptions to this rule are the sperm chemoattractants of corals, ascidians, plants such as ferns, and algae (Table 1).
Table 1. Some sperm chemoattractants in non-mammalian species*
| Species | Chemoattractant | References |
|---|---|---|
| Algae | Low-molecular-weight unsaturated pheromones of cyclic or linear structure (for example 532 Da pentosylated hydroquinone in the case of Chlamydomonas allensworthii) | [3][10][11] |
| Amphibians | Allurin — a 21 kDa protein (for Xenopus) | [9][12] |
| Ascidians | SAAF — a sulfated steroid: 3,4,7,26-tetrahydroxycholestane-3,26-disulfate (for Ciona savignyi and intestinalis) | [13][14][15] |
| Corals | A lipid-like long chain fatty alcohol CH3-(CH2)8-CH=CH-CH=CH-CH2OH (for Montipora digitata) | [16] |
| Ferns | Dicarboxylic acids, for example malic acid in its partially ionized form (for Pteridium aquilinum) | [17] |
| Mollusks | SepSAP — a 6-residue peptide-amide with the sequence PIDPGV-CONH2 (for Sepia officinalis) | [18] |
| Sea urchins | Resact — a 14-residue peptide with the sequence CVTGAPGCVGGGRL-NH2 (for Arbacia punctulata) | [19] |
| Starfish | Startrak — a 13 kDa heat-stable protein (for Pycnopodia helianthoides) | [20] |
- Taken from reference.[21]
Species specificity
The variety of chemoattractants raises the question of species specificity with respect to the chemoattractant identity. There is no single rule for chemoattractant-related specificity. Thus, in some groups of marine invertebrates (e.g., hydromedusae and certain ophiuroids), the specificity is very high; in others (e.g., starfish), the specificity is at the family level and, within the family, there is no specificity.[2][3][22] In mollusks, there appears to be no specificity at all. Likewise, in plants, a unique simple compound [e.g., fucoserratene — a linear, unsaturated alkene (1,3-trans 5-cis-octatriene)] might be a chemoattractant for various species.[10]
Behavioral mechanism
Here, too, there is no single rule. In some species (for example, in hydroids like Campanularia or tunicate like Ciona), the swimming direction of the spermatozoa changes abruptly towards the chemoattractant source. In others (for example, in sea urchin, hydromedusa, fern, or fish such as Japanese bitterlings), the approach to the chemoattractant source is indirect and the movement is by repetitive loops of small radii. In some species (for example, herring or the ascidian Ciona) activation of motility precedes chemotaxis.[2][3][23][24] In chemotaxis, cells may either sense a temporal gradient of the chemoattractant, comparing the occupancy of its receptors at different time points (as do bacteria[25]), or they may detect a spatial gradient, comparing the occupancy of receptors at different locations along the cell (as do leukocytes[26]). In the best-studied species, sea urchin, the spermatozoa sense a temporal gradient and respond to it with a transient increase in flagellar asymmetry. The outcome is a turn in the swimming path, followed by a period of straight swimming,[27] leading to the observed epicycloid-like movements directed towards the chemoattractant source.[28] The particular mechanism by which sea urchin sperm cells sense the temporal gradient has been recently identified as a natural implementation of the well-known adaptive controller known as extremum seeking. [29]
Molecular mechanism
The molecular mechanism of sperm chemotaxis is still not fully known. The current knowledge is mainly based on studies in the sea urchin Arbacia punctulata, where binding of the chemoattractant resact (Table 1) to its receptor, a guanylyl cyclase, activates cGMP synthesis (Figure 1). The resulting rise of cGMP possibly activates K+-selective ion channels. The consequential hyperpolarization activates hyperpolarization-activated and cyclic nucleotide-gated (HCN) channels. The depolarizing inward current through HCN channels possibly activates voltage-activated Ca2+ channels, resulting in elevation of intracellular Ca2+. This rise leads to flagellar asymmetry and, consequently, a turn of the sperm cell.[23]
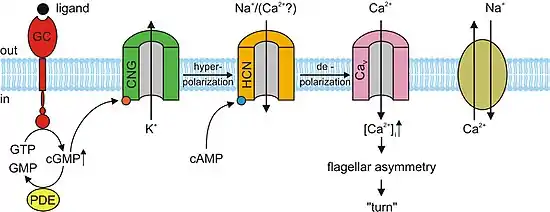
A model of the signal-transduction pathway during sperm chemotaxis of the sea urchin Arbacia punctulata. Binding of a chemoattractant (ligand) to the receptor — a membrane-bound guanylyl cyclase (GC) — activates the synthesis of cGMP from GTP. Cyclic GMP possibly opens cyclic nucleotide-gated (CNG) K+-selective channels, thereby causing hyperpolarization of the membrane. The cGMP signal is terminated by the hydrolysis of cGMP through phosphodiesterase (PDE) activity and inactivation of GC. On hyperpolarization, hyperpolarization-activated and cyclic nucleotide-gated (HCN) channels allow the influx of Na+ that leads to depolarization and thereby causes a rapid Ca2+ entry through voltage-activated Ca2+ channels (Cav), Ca2+ ions interact by unknown mechanisms with the axoneme of the flagellum and cause an increase of the asymmetry of flagellar beat and eventually a turn or bend in the swimming trajectory. Ca2+ is removed from the flagellum by a Na+/Ca2+ exchange mechanism. (Taken from ref.[23])
Sperm chemotaxis in mammals

Following the findings that human spermatozoa accumulate in follicular fluid[30][31] and that there is a remarkable correlation between this in vitro accumulation and oocyte fertilization,[30] chemotaxis was substantiated as the cause of this accumulation.[8] Sperm chemotaxis was later also demonstrated in mice[32] and rabbits.[33] In addition, sperm accumulation in follicular fluid (but without substantiating that it truly reflects chemotaxis) was demonstrated in horses[34] and pigs.[35] A key feature of sperm chemotaxis in humans is that this process is restricted to capacitated cells[36][37] — the only cells that possess the ability to penetrate the oocyte and fertilize it.[38] This raised the possibility that, in mammals, chemotaxis is not solely a guidance mechanism but it is also a mechanism of sperm selection.[36][37] Importantly, the fraction of capacitated (and, hence, chemotactically responsive) spermatozoa is low (~10% in humans), the life span of the capacitated/chemotactic state is short (1–4 hours in humans), a spermatozoon can be at this state only once in its lifetime, and sperm individuals become capacitated/chemotactic at different time points, resulting in continuous replacement of capacitated/chemotactic cells within the sperm population, i.e., prolonged availability of capacitated cells.[36][39] These sperm features raised the possibility that prolonging the time period, during which capacitated spermatozoa can be found in the female genital tract, is a mechanism, evolved in humans, to compensate for the lack of coordination between insemination and ovulation.[7][36][37][40]
Chemoattractants
In humans, there are at least two different origins of sperm chemoattractants. One is the cumulus cells that surround the oocyte, and the other is the mature oocyte itself.[41] The chemoattractant secreted from the cumulus cells is the steroid progesterone, shown to be effective at the picomolar range.[42][43][44] The chemoattractant secreted from the oocyte is even more potent.[41] It is a hydrophobic non-peptide molecule which, when secreted from the oocyte, is in complex with a carrier protein[45] Additional compounds have been shown to act as chemoattractants for mammalian spermatozoa. They include the chemokine CCL20,[46] atrial natriuretic peptide (ANP),[47] specific odorants,[48] natriuretic peptide type C (NPPC),[49] and allurin,[50] to mention a few. It is reasonable to assume that not all of them are physiologically relevant.
Species specificity
Species specificity was not detected in experiments that compared the chemotactic responsiveness of human and rabbit spermatozoa to follicular fluids or egg-conditioned media obtained from human, bovine, and rabbit.[51] The subsequent findings that cumulus cells of both human and rabbit (and, probably, of other mammals as well) secrete the chemoattractant progesterone[42][43][44] is sufficient to account for the lack of specificity in the chemotactic response of mammalian spermatozoa.
Behavioral mechanism
Mammalian spermatozoa, like sea-urchin spermatozoa, appear to sense the chemoattractant gradient temporally (comparing receptor occupancy over time) rather than spatially (comparing receptor occupancy over space). This is because the establishment of a temporal gradient in the absence of spatial gradient, achieved by mixing human spermatozoa with a chemoattractant[52] or by photorelease of a chemoattractant from its caged compound,[53] results in delayed transient changes in swimming behavior that involve increased frequency of turns and hyperactivation events. On the basis of these observations and the finding that the level of hyperactivation events is reduced when chemotactically responsive spermatozoa swim in a spatial chemoattractant gradient[53] it was proposed that turns and hyperactivation events are suppressed when capacitated spermatozoa swim up a chemoattractant gradient, and vice versa when they swim down a gradient.[52][53] In other words, human spermatozoa approach chemoattractants by modulating the frequency of turns and hyperactivation events, similarly to Escherichia coli bacteria.[25]
Molecular mechanism
As in non-mammalian species, the end signal in chemotaxis for changing the direction of swimming is Ca2+.[54] The discovery of progesterone as a chemoattractant[42][43][44] led to the identification of its receptor on the sperm surface – CatSper, a Ca2+ channel present exclusively in the tail of mammalian spermatozoa.[55][56] (Note, though, that progesterone only stimulates human CatSper but not mouse CatSper.[56] Consistently, sperm chemotaxis to progesterone was not found in mice.[57]) However, the molecular steps subsequent to CatSper activation by progesterone are obscure, though the involvement of trans-membrane adenylyl cyclase, cAMP and protein kinase A as well as soluble guanylyl cyclase, cGMP, inositol trisphosphate receptor and store-operated Ca2+ channel was proposed.[58]
Physiology
Chemotaxis is a short-range guidance mechanism. As such, it can guide spermatozoa for short distances only, estimated at the order of millimeters.[59] It is, therefore, believed that, in mammals, sperm chemotaxis occurs in the oviduct, close to the oocyte. First spermatozoa may be chemotactically guided to the oocyte-cumulus complex by the gradient of progesterone, secreted from the cumulus cells.[42][43][44] In addition, progesterone may inwardly guide spermatozoa, already present within the periphery of the cumulus oophorus.[42] Spermatozoa that are already deep within the cumulus oophorus may sense the more potent chemoattractant that is secreted from the oocyte[41][45] and chemotactically guide themselves to the oocyte according to the gradient of this chemoattractant. It should be borne in mind, however, that this scenario may be an oversimplification. In view of the increasing number of different chemoattractants that are being discovered, the physiology of chemotaxis in vivo might be much more complex.
References
- Pfeffer, W. (1884) Lokomotorische richtungsbewegungen durch chemische reize. Untersuch. aus d. Botan. Inst. Tübingen 1, 363–482.
- Miller, R.L. (1985) Sperm chemo-orientation in the metazoa. In: Biology of Fertilization (Metz, C.B. and Monroy, A., eds.), pp. 275–337. Academic Press, New York.
- Cosson, M.P. (1990) Sperm chemotaxis. In: Controls of Sperm Motility: Biological and Clinical Aspects (Gagnon, C., ed.) pp. 103–135. CRC Press, Boca Raton, FL.
- Eisenbach, M. and Tur-Kaspa, I. (1994) Human sperm chemotaxis is not enigmatic anymore. Fertil. Steril. 62, 233–235.
- Kaupp, U.B., Kashikar, N.D. and Weyand, I. (2008) Mechanisms of sperm chemotaxis Annu. Rev. Physiol. 70, 93-117.
- "Professor Michael Eisenbach of the Weizmann Membrane Reearch Department was inspired to initiate this research several years ago when ..." "Egg to Sperm: Don't Call Us, We'll Call You". Weizmann News (Fall 1991, p. 3). 1991.
- Eisenbach, M. and Giojalas, L.C. (2006) Sperm guidance in mammals - an unpaved road to the egg. Nat. Rev. Mol. Cell Biol. 7, 276–285.
- Ralt, D., Manor, M., Cohen-Dayag, A., Tur-Kaspa, I., Makler, A., Yuli, I., Dor, J., Blumberg, S., Mashiach, S. and Eisenbach, M. (1994) Chemotaxis and chemokinesis of human spermatozoa to follicular factors. Biol. Reprod. 50, 774–785.
- Al-Anzi, B. and Chandler, D.E. (1998) A sperm chemoattractant is released from Xenopus egg jelly during spawning. Dev. Biol. 198, 366–375.
- Maier, I. and Müller, D.G. (1986) Sexual pheromones in algae. Biol. Bull. 170, 145–175.
- Starr, R.C., Marner, F.J. and Jaenicke, L. (1995) Chemoattraction of male gametes by a pheromone produced by female gametes of chlamydomonas. Proc. Natl. Acad. Sci. U.S.A. 92, 641–645.
- Olson, J.H., Xiang, X.Y., Ziegert, T., Kittelson, A., Rawls, A., Bieber, A.L. and Chandler, D.E. (2001) Allurin, a 21-kDa sperm chemoattractant from Xenopus egg jelly, is related to mammalian sperm-binding proteins. Proc. Natl. Acad. Sci. U.S.A. 98, 11205–11210.
- Yoshida, M., Inaba, K. and Morisawa, M. (1993) Sperm chemotaxis during the process of fertilization in the ascidians Ciona-Savignyi and Ciona-Intestinalis. Dev. Biol. 157, 497–506.
- Yoshida, M., Inaba, K., Ishida, K. and Morisawa, M. (1994) Calcium and cyclic AMP mediate sperm activation, but Ca2+ alone contributes sperm chemotaxis in the ascidian, Ciona savignyi. Dev. Growth Dif. 36, 589–595.
- Yoshida, M., Murata, M., Inaba, K. and Morisawa, M. (2002) A chemoattractant for ascidian spermatozoa is a sulfated steroid. Proc. Natl. Acad. Sci. U.S.A. 99, 14831–14836.
- Coll, J.C. and Miller, R.L. (1992) The nature of sperm chemo-attractants in coral and starfis. In: Comparative Spermatology: 20 Years After (Baccetti, B., ed.) pp. 129–134. Raven Press, New York.
- Brokaw, C.J. (1958) Chemotaxis of bracken spermatozoids. The role of bimalate ions. J. Exp. Zool. 35, 192–196.
- Zatylny, C., Marvin, L., Gagnon, J. and Henry, J.L. (2002) Fertilization in Sepia officinalis: the first mollusk sperm-attracting peptide. Biochem. Biophys. Res. Commun. 296, 1186–1193.
- Ward, G.E., Brokaw, C.J., Garbers, D.L. and Vacquier, V.D. (1985) Chemotaxis of Arbacia punctulata spermatozoa to resact, a peptide from the egg jelly layer. J. Cell Biol. 101, 2324–2329.
- Miller, R.D. and Vogt, R. (1996) An N-terminal partial sequence of the 13kDa Pycnopodia helianthoides sperm chemoattractant 'startrak' possesses sperm-attracting activity. J. Exp. Biol. 199, 311–318.
- Eisenbach, M. (2004) Chemotaxis. Imperial College Press, London.
- Miller, R.L. (1997) Specificity of sperm chemotaxis among great barrier reef shallow-water holothurians and ophiuroids. J. Exp. Zool. 279, 189–200.
- Kaupp, U.B., Hildebrand, E. and Weyand, I. (2006) Sperm chemotaxis in marine invertebrates - molecules and mechanism. J. Cell. Physiol. 208, 487–494.
- Morisawa, M. (1994). Cell signaling mechanisms for sperm motility. Zool. Sci. 11, 647–662.
- Macnab, R.M. and Koshland, D.E. (1972) The gradient-sensing mechanism in bacterial chemotaxis. Proc. Natl. Acad. Sci. U.S.A. 69, 2509–2512.
- Devreotes, P.N. and Zigmond, S.H. (1988) Chemotaxis in eukaryotic cells: a focus on leukocytes and Dictyostelium. Annu Rev Cell Biol 4, 649–86.
- Kaupp, U.B., Solzin, J., Hildebrand, E., Brown, J.E., Helbig, A., Hagen, V., Beyermann, M., Pampaloni, F. and Weyand, I. (2003) The signal flow and motor response controlling chemotaxis of sea urchin sperm. Nature Cell Biol. 5, 109–117.
- Böhmer, M., Van, Q., Weyand, I., Hagen, V., Beyermann, M., Matsumoto, M., Hoshi, M., Hildebrand, E. and Kaupp, U.B. (2005) Ca2+ spikes in the flagellum control chemotactic behavior of sperm. EMBO J. 24, 2741–2752.
- Abdelgalil, M., Aboelkassem, Y., Taha, H. (2022) Sea urchin sperm exploit extremum seeking control to find the egg. Phys. Rev. E 106, L062401
- Ralt, D., Goldenberg, M., Fetterolf, P., Thompson, D., Dor, J., Mashiach, S., Garbers, D.L. and Eisenbach, M. (1991) Sperm attraction to a follicular factor(s) correlates with human egg fertilizability. Proc. Natl. Acad. Sci. U.S.A. 88, 2840–2844.
- Villanueva-Díaz, C., Vadillo-Ortega, F., Kably-Ambe, A., Diaz-Perez, M.A. and Krivitzky, S.K. (1990) Evidence that human follicular fluid contains a chemoattractant for spermatozoa. Fertil. Steril. 54, 1180–1182.
- Oliveira, R.G., Tomasi, L., Rovasio, R.A. and Giojalas, L.C. (1999) Increased velocity and induction of chemotactic response in mouse spermatozoa by follicular and oviductal fluids. J. Reprod. Fertil. 115, 23–27.
- Fabro, G., Rovasio, R.A., Civalero, S., Frenkel, A., Caplan, S.R., Eisenbach, M. and Giojalas, L.C. (2002) Chemotaxis of capacitated rabbit spermatozoa to follicular fluid revealed by a novel directionality-based assay. Biol. Reprod. 67, 1565–1571.
- Navarro, M.C., Valencia, J., Vazquez, C., Cozar, E. and Villanueva, C. (1998) Crude mare follicular fluid exerts chemotactic effects on stallion spermatozoa. Reprod. Domest. Anim. 33, 321–324.
- Serrano, H., Canchola, E. and García-Suárez, M.D. (2001) Sperm-attracting activity in follicular fluid associated to an 8.6-kDa protein. Biochem. Biophys. Res. Commun. 283, 782–784.
- Cohen-Dayag, A., Tur-Kaspa, I., Dor, J., Mashiach, S. and Eisenbach, M. (1995) Sperm capacitation in humans is transient and correlates with chemotactic responsiveness to follicular factors. Proc. Natl. Acad. Sci. U.S.A. 92, 11039–11043.
- Eisenbach, M. (1999) Mammalian sperm chemotaxis and its association with capacitation. Dev. Genet. 25, 87–94.
- Jaiswal, B.S. and Eisenbach, M. (2002) Capacitation. In: Fertilization (Hardy, D.M., ed.) pp. 57–117. Academic Press, San Diego.
- Cohen-Dayag, A., Ralt, D., Tur-Kaspa, I., Manor, M., Makler, A., Dor, J., Mashiach, S. and Eisenbach, M. (1994) Sequential acquisition of chemotactic responsiveness by human spermatozoa. Biol. Reprod. 50, 786–790.
- Giojalas, L.C., Rovasio, R.A., Fabro, G., Gakamsky, A. and Eisenbach, M. (2004) Timing of sperm capacitation appears to be programmed according to egg availability in the female genital tract. Fertil. Steril. 82, 247–249.
- Sun, F., Bahat, A., Gakamsky, A., Girsh, E., Katz, N., Giojalas, L.C., Tur-Kaspa, I. and Eisenbach, M. (2005) Human sperm chemotaxis: both the oocyte and its surrounding cumulus cells secrete sperm chemoattractants. Hum. Reprod. 20, 761–767.
- Teves, M.E., Barbano, F., Guidobaldi, H.A., Sanchez, R., Miska, W. and Giojalas, L.C. (2006) Progesterone at the picomolar range is a chemoattractant for mammalian spermatozoa. Fertil. Steril. 86, 745–749.
- Oren-Benaroya, R., Orvieto, R., Gakamsky, A., Pinchasov, M. and Eisenbach, M. (2008) The sperm chemoattractant secreted from human cumulus cells is progesterone. Hum. Reprod. 23, 2339–2345.
- Guidobaldi, H.A., Teves, M.E., Unates, D.R., Anastasia, A. and Giojalas, L.C. (2008) Progesterone from the cumulus cells is the sperm chemoattractant secreted by the rabbit oocyte cumulus complex. PLOS One 3, e3040.
- Armon, L., Ben-Ami, I., Ron-El, R. and Eisenbach, M. (2014) Human oocyte-derived sperm chemoattractant is a hydrophobic molecule associated with a carrier protein. Fertil. Steril. 102, 885–890.
- Caballero-Campo, P., Buffone, M.G., Benencia, F., Conejo-García, J.R., Rinaudo, P.F and Gerton, G.L. (2014) A role for the chemokine receptor CCR6 in mammalian sperm motility and chemotaxis. J. Cell. Physiol. 229, 68–78.
- Zamir, N., Riven-Kreitman, R., Manor, M., Makler, A., Blumberg, S., Ralt, D. and Eisenbach, M. (1993) Atrial natriuretic peptide attracts human spermatozoa in vitro. Biochem. Biophys. Res. Commun. 197, 116–122.
- Spehr, M., Gisselmann, G., Poplawski, A., Riffell, J.A., Wetzel, C.H., Zimmer, R.K. and Hatt, H. (2003) Identification of a testicular odorant receptor mediating human sperm chemotaxis'. Science 299, 2054–2058.
- Kong, N., Xu, X., Zhang, Y., Wang, Y., Hao, X., Zhao, Y., Qiao, J., Xia, G. and Zhang, M. (2017) Natriuretic peptide type C induces sperm attraction for fertilization in mouse. Sci. Rep. 7, 39711.
- Burnett, L.A., Anderson, D.M., Rawls, A., Bieber, A.L. and Chandler, D.E. (2011) Mouse sperm exhibit chemotaxis to allurin, a truncated member of the cysteine-rich secretory protein family. Dev. Biol. 360, 318–328.
- Sun, F., Giojalas, L.C., Rovasio, R.A., Tur-Kaspa, I., Sanchez, R. and Eisenbach, M. (2003) Lack of species-specificity in mammalian sperm chemotaxis. Dev. Biol. 255, 423–427.
- Gakamsky, A., Armon, L. and Eisenbach, M. (2009) Behavioral response of human spermatozoa to a concentration jump of chemoattractants or intracellular cyclic nucleotides. Hum. Reprod. 24, 1152-1163.
- Armon, L. and Eisenbach, M. (2011) Behavioral mechanism during human sperm chemotaxis: Involvement of hyperactivation. PLOS One 6, e28359.
- Sugiyama, H. and Chandler, D.E. (2014) Sperm guidance to the egg finds calcium at the helm. Protoplasma 251, 461-475.
- Strünker, T., Goodwin, N., Brenker, C., Kashikar, N.D., Weyand, I., Seifert, R. and Kaupp, U.B. (2011) The CatSper channel mediates progesterone-induced Ca2+ influx in human sperm. Nature 471, 382–386.
- Lishko, P.V., Botchkina, I.L. and Kirichok, Y. (2011) Progesterone activates the principal Ca2+ channel of human sperm. Nature 471, 387–391.
- Chang, H., Kim, B. J., Kim, Y. S., Suarez, S. S., and Wu, M. (2013) Different migration patterns of sea urchin and mouse sperm revealed by a microfluidic chemotaxis device. PLOS One 8, e60587.
- Teves, M.E., Guidobaldi, H.A., Unates, D.R., Sanchez, R., Miska, W., Publicover, S.J., Morales Garcia, A.A. and Giojalas, L. (2009) Molecular mechanism for human sperm chemotaxis mediated by progesterone. PLOS One 4, e8211.
- Pérez-Cerezales, S., Boryshpolets, S. and Eisenbach, M. (2015) Behavioral mechanisms of mammalian sperm guidance. Asian J. Androl. 17, 628-632
Further reading
- Michael Eisenbach (with contribution from Joseph W Lengeler) (2004). "Chemotaxis." Imperial College Press, London.
- Michael Eisenbach and Laura Cecilia Giojalas (2006). "Sperm guidance in mammals - an unpaved road to the egg". Nat. Rev. Mol. Cell Biol. 7, 276–285.
- Alvarez, L., Friedrich, B.M., Gompper, G., Kaupp. U.B. (2013). "The computational sperm cell". Trends in Cell Biol.