Squalamine
Squalamine is a steroid-polyamine conjugate compound with broad spectrum antimicrobial activity and anti-angiogenic activity. It was studied as a potential cancer drug and as a potential treatment for wet macular degeneration but as of 2018 had not succeeded in Phase III trials for any use.
 | |
| Names | |
|---|---|
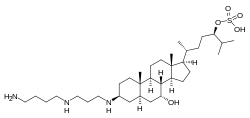
| IUPAC name
(24R)-3β-({3-[(3-Aminopropyl)amino]propyl}amino)-7α-hydroxycholestan-24-yl hydrogen sulfate | |
| Systematic IUPAC name
(3R,6R)-6-[(1R,3aS,3bR,4R,5aR,7S,9aS,9bS,11aR)-7-({3-[(3-Aminopropyl)amino]propyl}amino)-4-hydroxy-9a,11a-dimethylhexadecahydro-1H-cyclopenta[a]phenanthren-1-yl]-2-methylheptan-3-yl hydrogen sulfate | |
| Identifiers | |
3D model (JSmol) |
|
| ChEMBL | |
| ChemSpider | |
| KEGG | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C34H65N3O5S | |
| Molar mass | 628 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Pharmacology
Squalamine disrupts microbial membranes; the exact mechanism is not known.[1][2]
Squalamine appears to have an anti-angiogenic effect in mammalian cells by binding to calmodulin once it is taken up by the cell; this prevents signal transduction downstream of pro-angiogenic factors like VEGF.[3]
Chemistry
Squalamine is a steroid-polyamine conjugate.[4]
It was first isolated from the liver of sharks of the genus Squalus but methods to make it synthetically have been subsequently developed.[4]
Research
Squalamine was studied as a potential drug to treat several forms of cancer and wet macular degeneration by scientists at Magainin Pharmaceuticals (subsequently called Genaera), but the company ran out of money and closed in 2009.[5][6] Squalamine and some other drug assets were sold to Ohr Pharmaceutical for $200,000 by Genaera's liquidator.[7]
It was studied in an eye drop formulation as a potential way to treat wet macular degeneration in combination with ranibizumab by Ohr Pharmaceuticals.[3] A Phase II trial failed in 2015, but Ohr said a subpopulation with choroidal neovascularization responded well enough to justify a Phase III trial.[8] Ohr announced that the Phase III trial had failed in January 2018.[9]
References
- Khelaifia, S; Drancourt, M (September 2012). "Susceptibility of archaea to antimicrobial agents: applications to clinical microbiology". Clinical Microbiology and Infection. 18 (9): 841–8. doi:10.1111/j.1469-0691.2012.03913.x. PMID 22748132.

- Cushnie, TP; Cushnie, B; Lamb, AJ (November 2014). "Alkaloids: an overview of their antibacterial, antibiotic-enhancing and antivirulence activities". International Journal of Antimicrobial Agents. 44 (5): 377–86. doi:10.1016/j.ijantimicag.2014.06.001. PMID 25130096.
- Schlottmann, PG; Alezzandrini, AA; Zas, M; Rodriguez, FJ; Luna, JD; Wu, L (2017). "New Treatment Modalities for Neovascular Age-Related Macular Degeneration". Asia-Pacific Journal of Ophthalmology. 6 (6): 514–519. doi:10.22608/APO.2017258. PMID 28933517.

- Brycki, B; Koenig, H; Pospieszny, T (23 November 2015). "Quaternary Alkylammonium Conjugates of Steroids: Synthesis, Molecular Structure, and Biological Studies". Molecules (Basel, Switzerland). 20 (11): 20887–900. doi:10.3390/molecules201119735. PMC 6331993. PMID 26610455.

- George, John (April 29, 2009). "Biotech Genaera shutting down: Never brought drug to market". Philadelphia Business Journal.
- "Squalamine". AdisInsight. Retrieved 16 January 2018.
- "Ohr Pharmaceutical 10-K for the fiscal year ended September 30, 2009". Ohr via SEC Edgar. January 8, 2010. p. 11.
- Garde, Damian (March 27, 2015). "Ohr Pharma's eye drop fails a Phase II AMD trial". FierceBiotech.
- "Press release: Ohr Pharmaceutical Announces Efficacy Results from the MAKO Study in Wet-AMD". Ohr via GlobeNewswire. January 5, 2018.