Strobilurin
Strobilurins are a group of natural products and their synthetic analogs. A number of strobilurins are used in agriculture as fungicides. They are part of the larger group of QoIs (Quinone outside Inhibitors), which act to inhibit the respiratory chain at the level of Complex III.
The first parent natural products, strobilurins A and B, were extracted from the fungus Strobilurus tenacellus.[1] Commercial strobilurin fungicides[2] were developed through optimization of photostability and activity.[3] Strobilurins represented a major development in fungus-based fungicides. First released in 1996, there are now ten major strobilurin fungicides on the market, which account for 23-25 % of the global fungicide sales.[4] Examples of commercialized strobilurin derivatives are azoxystrobin, kresoxim-methyl, picoxystrobin, fluoxastrobin, oryzastrobin, dimoxystrobin, pyraclostrobin and trifloxystrobin.
Strobilurins are mostly contact fungicides with a long half time as they are absorbed into the cuticle and not transported any further. They have a suppressive effect on other fungi, reducing competition for nutrients; they inhibit electron transfer in mitochondria, disrupting metabolism and preventing growth of the target fungi.[5]
Natural strobilurins
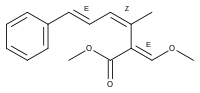
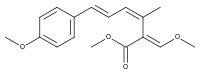
Strobilurin A
Strobilurin A (also known as mucidin) is produced by Oudemansiella mucida, Strobilurus tenacellus, Bolinea lutea, and others.[6][7][8] When first isolated it was incorrectly assigned as the E E E geometric isomer but was later identified by total synthesis as being the E Z E isomer, as shown.[5]: 694
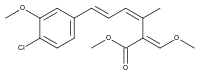
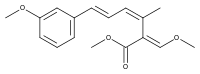
9-Methoxystrobilurin A
9-Methoxystrobilurin A is produced by Favolaschia spp.[7]
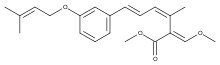
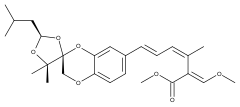
Strobilurin D and G
Strobilurin D is produced by Cyphellopsis anomala.[8] Its structure was originally incorrectly assigned and is now considered to be identical to that of strobilurin G, produced by B. lutea.[7][8] A related material, hydroxystrobilurin D, with an additional hydroxyl group attached to the methyl of the main chain is produced by Mycena sanguinolenta.[7]
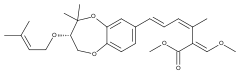
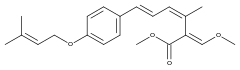
Strobilurin H
Strobilurin H is produced by B. lutea.[7] The natural product with a phenolic hydroxy group in place of the aromatic methoxy group of strobilurin H is called strobilurin F1 and is found in C. anomala[8] and Agaricus spp.[6]
Oudemansins
The oudemansins are closely related to the strobilurins and are also quinone outside inhibitors.[7]
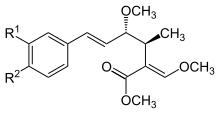
Oudemansin A with R1 = R2 = H was first described in 1979, after being isolated from mycelial fermentations of the basidiomycete fungus Oudemansiella mucida.[9] Later it was found in cultures of the basidiomycete fungi Mycena polygramma and Xerula melanotricha. The latter fungus also produces oudemansin B, with R1 = MeO and R2 = Cl. Oudemansin X, with R1 = H and R2 = MeO was isolated from Oudemansiella radicata.[6]
Synthetic strobilurins
The discovery of the strobilurin class of fungicides led to the development of a group of commercial fungicides used in agriculture. Examples are shown below.[5]
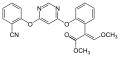
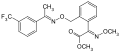 Trifloxystrobin
Trifloxystrobin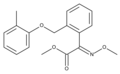 Kresoxim methyl
Kresoxim methyl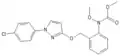
See also
References
- Schramm, Georg; Steglich, Wolfgang; Anke, Timm; Oberwinkler, Franz (1978). "Antibiotika aus Basidiomyceten, III. Strobilurin a und B, antifungische Stoffwechselprodukte aus Strobilurus tenacellus". Chemische Berichte. 111 (8): 2779–2784. doi:10.1002/cber.19781110806.
- Peter Jeschke, Matthias Witschel, Wolfgang Krämer, Ulrich Schirmer (eds.): Modern Crop Protection Compounds, 3rd edition, Wiley-VCH, 2019, ISBN 978-3-527-34089-7.
- Sauter, Hubert; Steglich, Wolfgang; Anke, Timm (1999). "Strobilurins: Evolution of a New Class of Active Substances". Angewandte Chemie International Edition. 38 (10): 1328–1349. doi:10.1002/(SICI)1521-3773(19990517)38:10<1328::AID-ANIE1328>3.0.CO;2-1. PMID 29711574.
-
Juliet D. Tang, Tina Ciaramitaro, Maria Tomaso-Peterson, Susan V. Diehl (2017). "Activity of Two Strobilurin Fungicides Against Three Species of Decay Fungi in Agar Plate Tests". Proc. IRG Annual Meeting: IRG/WP 17-30704.
{{cite journal}}: CS1 maint: multiple names: authors list (link) (pdf link) - Schaefer, Bernd (2014). "Agrochemicals: 8.2 Strobilurins". Natural Products in the Chemical Industry. pp. 688–704. doi:10.1007/978-3-642-54461-3_8. ISBN 978-3-642-54460-6.
- Lorenzen, K.; Anke, T. (1998). Mori, Kenji (ed.). "Basidiomycetes as a Source for New Bioactive Natural Products". Current Organic Chemistry. Bentham Science Publishers. 2 (4): 329–364. doi:10.2174/1385272802666220128213627. ISSN 1385-2728. S2CID 246934805.
- Anke, T.; Erkel, G. (2002). "Non-β-Lactam Antibiotics". Industrial Applications. pp. 101–104. doi:10.1007/978-3-662-10378-4_5. ISBN 978-3-642-07481-3.
- Zakharychev, Vladimir V; Kovalenko, Leonid V (1998-06-30). "Natural compounds of the strobilurin series and their synthetic analogues as cell respiration inhibitors". [Russian Chemical Reviews] (Успехи химии). IOP Publishing. 67 (6): 535–544. Bibcode:1998RuCRv..67..535Z. doi:10.1070/rc1998v067n06abeh000426. ISSN 0036-021X. S2CID 95676421.
- Anke, Timm; Hecht, Hans Jürgen; Chramm, Georgs; Steglich, Wolfgang (1979). "Antibiotics from basidiomycetes. IX. Oudemansin, an antifungal antibiotic from Oudemansiella mucida (Schrader ex Fr.) hoehnel (Agaricales)". The Journal of Antibiotics. 32 (11): 1112–1117. doi:10.7164/antibiotics.32.1112. PMID 528381.
External links
- Resistance to strobilurins in the southern USA, archived from the original on 2019-09-08, retrieved 2019-09-08
- Agricultural mycocides for the 21st century: strobilurins, archived from the original on 2019-09-08, retrieved 2019-09-08, from: David Moore, Geoffrey D. Robson, Anthony P. J. Trinci, 21st Century Guidebook to Fungi, 2nd edition.