Spin–lattice relaxation
During nuclear magnetic resonance observations, spin–lattice relaxation is the mechanism by which the longitudinal component of the total nuclear magnetic moment vector (parallel to the constant magnetic field) exponentially relaxes from a higher energy, non-equilibrium state to thermodynamic equilibrium with its surroundings (the "lattice"). It is characterized by the spin–lattice relaxation time, a time constant known as T1.
There is a different parameter, T2, the spin-spin relaxation time, which concerns the exponential relaxation of the transverse component of the nuclear magnetization vector (perpendicular to the external magnetic field). Measuring the variation of T1 and T2 in different materials is the basis for some magnetic resonance imaging techniques.[1]
Nuclear physics
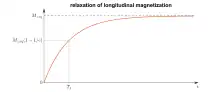
T1 characterizes the rate at which the longitudinal Mz component of the magnetization vector recovers exponentially towards its thermodynamic equilibrium, according to equation
Or, for the specific case that
It is thus the time it takes for the longitudinal magnetization to recover approximately 63% [1-(1/e)] of its initial value after being flipped into the magnetic transverse plane by a 90° radiofrequency pulse.
Nuclei are contained within a molecular structure, and are in constant vibrational and rotational motion, creating a complex magnetic field. The magnetic field caused by thermal motion of nuclei within the lattice is called the lattice field. The lattice field of a nucleus in a lower energy state can interact with nuclei in a higher energy state, causing the energy of the higher energy state to distribute itself between the two nuclei. Therefore, the energy gained by nuclei from the RF pulse is dissipated as increased vibration and rotation within the lattice, which can slightly increase the temperature of the sample. The name spin-lattice relaxation refers to the process in which the spins give the energy they obtained from the RF pulse back to the surrounding lattice, thereby restoring their equilibrium state. The same process occurs after the spin energy has been altered by a change of the surrounding static magnetic field (e.g. pre-polarization by or insertion into high magnetic field) or if the nonequilibrium state has been achieved by other means (e.g., hyperpolarization by optical pumping).
The relaxation time, T1 (the average lifetime of nuclei in the higher energy state) is dependent on the gyromagnetic ratio of the nucleus and the mobility of the lattice. As mobility increases, the vibrational and rotational frequencies increase, making it more likely for a component of the lattice field to be able to stimulate the transition from high to low energy states. However, at extremely high mobilities, the probability decreases as the vibrational and rotational frequencies no longer correspond to the energy gap between states.
Different tissues have different T1 values. For example, fluids have long T1s (1500-2000 ms), and water-based tissues are in the 400-1200 ms range, while fat based tissues are in the shorter 100-150 ms range. The presence of strongly magnetic ions or particles (e.g., ferromagnetic or paramagnetic) also strongly alter T1 values and are widely used as MRI contrast agents.
T1 weighted images
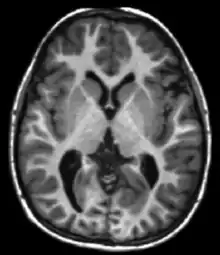
Magnetic resonance imaging uses the resonance of the protons to generate images. Protons are excited by a radio frequency pulse at an appropriate frequency (Larmor frequency) and then the excess energy is released in the form of a minuscule amount of heat to the surroundings as the spins return to their thermal equilibrium. The magnetization of the proton ensemble goes back to its equilibrium value with an exponential curve characterized by a time constant T1 (see Relaxation (NMR)).
T1 weighted images can be obtained by setting short repetition time (TR) such as < 750 ms and echo time (TE) such as < 40 ms in conventional spin echo sequences, while in Gradient Echo Sequences they can be obtained by using flip angles of larger than 50o while setting TE values to less than 15 ms.
T1 is significantly different between grey matter and white matter and is used when undertaking brain scans. A strong T1 contrast is present between fluid and more solid anatomical structures, making T1 contrast suitable for morphological assessment of the normal or pathological anatomy, e.g., for musculoskeletal applications.
In the rotating frame
Spin–lattice relaxation in the rotating frame is the mechanism by which Mxy, the transverse component of the magnetization vector, exponentially decays towards its equilibrium value of zero, under the influence of a radio frequency (RF) field in nuclear magnetic resonance (NMR) and magnetic resonance imaging (MRI). It is characterized by the spin–lattice relaxation time constant in the rotating frame, T1ρ. It is named in contrast to T1, the spin-lattice relaxation time.[2]
T1ρ MRI is an alternative to conventional T1 and T2 MRI by its use of a long-duration, low-power radio frequency referred to as spin-lock (SL) pulse applied to the magnetization in the transverse plane. The magnetization is effectively spin-locked around an effective B1 field created by the vector sum of the applied B1 and any off-resonant component. The spin-locked magnetization will relax with a time constant T1ρ, which is the time it takes for the magnetic resonance signal to reach 37% (1/e) of its initial value, . Hence the relation: , where tSL is the duration of the RF field.
Measurement
T1ρ can be quantified (relaxometry) by curve fitting the signal expression above as a function of the duration of the spin-lock pulse while the amplitude of spin-lock pulse (γB1~0.1-few kHz) is fixed. Quantitative T1ρ MRI relaxation maps reflect the biochemical composition of tissues.[3]
References
- Rinck, Peter A. (2022). Relaxation Times and Basic Pulse Sequences in MR Imaging. in: Magnetic Resonance in Medicine. A Critical Introduction. 12th edition. pp. 65-92. Offprint to download: TRTF - The Round Table Foundation / EMRF - European Magnetic Resonance Forum. ISBN 978-3-7460-9518-9.
- Levitt, Malcolm H. (2016). Spin Dynamics: Basics of Nuclear Magnetic Resonance 2nd Edition. Wiley. ISBN 978-0470511176.
- Borthakur, A; Mellon, E; Niyogi, S; Witschey, W; Kneeland, JB; Reddy, R (Nov 2006). "Sodium and T1rho MRI for molecular and diagnostic imaging of articular cartilage". NMR in Biomedicine. 19 (7): 781–821. doi:10.1002/nbm.1102. PMC 2896046. PMID 17075961.
- Li, X; Benjamin Ma, C; Link, TM; Castillo, DD; Blumenkrantz, G; Lozano, J; Carballido-Gamio, J; Ries, M; Majumdar, S (Jul 2007). "In vivo T(1rho) and T(2) mapping of articular cartilage in osteoarthritis of the knee using 3 T MRI". Osteoarthritis and Cartilage. 15 (7): 789–97. doi:10.1016/j.joca.2007.01.011. PMC 2040334. PMID 17307365.
- Witschey, WR; Borthakur, A; Fenty, M; Kneeland, BJ; Lonner, JH; McArdle, EL; Sochor, M; Reddy, R (May 2010). "T1rho MRI quantification of arthroscopically confirmed cartilage degeneration". Magnetic Resonance in Medicine. 63 (5): 1376–82. doi:10.1002/mrm.22272. PMC 2933515. PMID 20432308.
- Borthakur, A; Maurer, PM; Fenty, M; Wang, C; Berger, R; Yoder, J; Balderston, RA; Elliott, DM (Dec 1, 2011). "T1ρ magnetic resonance imaging and discography pressure as novel biomarkers for disc degeneration and low back pain". Spine. 36 (25): 2190–6. doi:10.1097/BRS.0b013e31820287bf. PMC 4002043. PMID 21358489.
- Borthakur, A; Sochor, M; Davatzikos, C; Trojanowski, JQ; Clark, CM (Jul 15, 2008). "T1rho MRI of Alzheimer's disease". NeuroImage. 41 (4): 1199–205. doi:10.1016/j.neuroimage.2008.03.030. PMC 2473861. PMID 18479942.
- Cai, K; Haris, M; Singh, A; Kogan, F; Greenberg, JH; Hariharan, H; Detre, JA; Reddy, R (Jan 22, 2012). "Magnetic resonance imaging of glutamate". Nature Medicine. 18 (2): 302–6. doi:10.1038/nm.2615. PMC 3274604. PMID 22270722.
- Witschey, WR; Pilla, JJ; Ferrari, G; Koomalsingh, K; Haris, M; Hinmon, R; Zsido, G; Gorman JH, 3rd; Gorman, RC; Reddy, R (Nov 2010). "Rotating frame spin lattice relaxation in a swine model of chronic, left ventricular myocardial infarction". Magnetic Resonance in Medicine. 64 (5): 1453–60. doi:10.1002/mrm.22543. PMC 2965811. PMID 20677236.
- Li, LZ; Zhou, R; Xu, HN; Moon, L; Zhong, T; Kim, EJ; Qiao, H; Reddy, R; Leeper, D; Chance, B; Glickson, JD (Apr 21, 2009). "Quantitative magnetic resonance and optical imaging biomarkers of melanoma metastatic potential". Proceedings of the National Academy of Sciences of the United States of America. 106 (16): 6608–13. Bibcode:2009PNAS..106.6608L. doi:10.1073/pnas.0901807106. PMC 2672511. PMID 19366661.
- Cai, K; Shore, A; Singh, A; Haris, M; Hiraki, T; Waghray, P; Reddy, D; Greenberg, JH; Reddy, R (Feb 2, 2012). "Blood oxygen level dependent angiography (BOLDangio) and its potential applications in cancer research". NMR in Biomedicine. 25 (10): 1125–1132. doi:10.1002/nbm.2780. PMC 3390450. PMID 22302557.
- McRobbie D., et al. MRI, From picture to proton. 2003
- Hashemi Ray, et al. MRI, The Basics 2ED. 2004.