Tectitethya crypta
Tectitethya crypta is a species of demosponge belonging to the family Tethyidae.[1] Its classified family is characterized by fourteen different known genera, one of them being Tectitethya.[2] It is a massive, shallow-water sponge found in the Caribbean Sea.[3][4] This sponge was first discovered by Werner Bergmann in 1945 and later classified by de Laubenfels in 1949. It is located in reef areas situated on softer substrates such as sand or mud.[5][6] Oftentimes, it is covered in sand and algae.[3][4] This results in an appearance that is cream colored/ gray colored; however, when the animal is washed free of its sediment coverings, its body plan appears more green and gray. It's characterized with ostia peaking out of its body cavity, with the ability to abruptly open or close, changing its desired water flow rate through its mesohyl.
| Tectitethya crypta | |
|---|---|
 | |
| Scientific classification | |
| Domain: | Eukaryota |
| Kingdom: | Animalia |
| Phylum: | Porifera |
| Class: | Demospongiae |
| Order: | Tethyida |
| Family: | Tethyidae |
| Genus: | Tectitethya |
| Species: | T. crypta |
| Binomial name | |
| Tectitethya crypta (de Laubenfels, 1949) | |
| Synonyms[1] | |
| |
This sponge is widely known for its contributions to the field of medicine as a source for potent nucleoside analogues used in treating H.I.V, Acute Myeloid Leukemia, pancreatic cancer, Ebola, and others. The nucleosides spongothymidine and spongouridine were isolated from this sponge, providing the basis for anti-viral drugs and anti-cancer drugs.[3][4] Vidarabine, an antiviral drug, was derived from these compounds.[7] The discovery of these nucleosides also led to the development of cytarabine for clinical use in the treatment of leukemia and lymphoma.[8] Gemcitabine, a fluorinated derivative of cytarabine, is used to treat pancreatic, breast, bladder, and non-small-cell lung cancer.[8] Holding such valuable compounds, free-living within the animal, T. crypta has shaped the present and future world of medicine.
Anatomy & physiology
Body morphology
As described by Laubenfels, the body of this sponge is amorphous, bulky, and approximately the size of one's fist. Its dimensions are around 4 by 7 by 12 centimetres (1.6 in × 2.8 in × 4.7 in) and may be cylindrical, conical, or hemispherical in shape.[2] More recent studies have indicated a larger range of size within this species. The outermost, visible layer of the animal can be seen to have flat tubercules, approximately 3 to 5 millimeters in diameter and a thick layer of sediment. Its actual olive pigment isn't easily visible under this layer of sand/ sediment. In clustered bundles on the surface of the animal are structures called megascleres, radiating and branching outwards. Ray tips are rounded; micrasters are seen to be 8 to 12 micrometers in diameter. Star spicules makeup a layer beneath its exterior skeleton. T. crypta is not characterized with a cortex.[2]
Size
Three main developmental phases have been identified in conjunction with the sponges' localization of course sediment within its body.[3] The small sponges are characterized with a spherical shape and possess evenly spread sediment. The medium T. crypta sponges are seen to have a conical shape with their sediment concentrated near their bottom or base. The larger sponges are seen to be irregular in shape and also have evenly distributed sediments. With each body size are different habits that each acquire. Smaller sponges are unattached and are seen to rest and roll freely. The medium sponges are also unattached; however, they still have great stability with their shape and sediment concentration. Lastly, the larger sponges are attached on their bottom-end. Typically, 67% of their body is buried in sand.
Movement
T. crypta are capable of strong body contractions and allow oscula the ability to move (open/ close) at a quick rate. In fact, this sponge is capable of closing its osculum completely, which has been proven to be a useful adaptation for an animal living in sandy environments. Ostia are about 1 millimeter in size, occurring in clusters along the flank of the sponge.[5] The osculum, bearing a diameter of 20 to 25 millimeters, are seen near the top of the cone. These structures have the ability to be contracted. The ability to circulate water through bottom sediments possibly makes for a nutrient-rich and attractive environment for other organisms to live in or near the sponges.[5]
Sediment organization
The dirty exterior of the sponge smothered in layers of algae/ sediment/ sand serves a purpose to the animal and has been shown to hold structural organization across its species. Sand that is brought into the body will be organized in patterns determined by its granulometry and sponge size.[3] This sorting and distribution occur in the choanosome: sediments smaller than 500 micrometers gather in clusters (known as nuclei) while the larger particles are found to be distributed evenly through the sponge body. T. crypta sponges have been noted to favor the selection of fine sediment grains within the range of 40 to 60 micrometers.[3] Additional analysis through microscopic tools has revealed high selection for allocthonous sponge spicules, radiolarians, and diatoms.[3] Deeper analysis of incorporated sediment is needed to identify additional materials and cells that have not been identified as of now. The sand is eventually transported by a specific cell to a desired location through the use of a cellular track which facilitates sediment transport from the ectosome to the accumulated nuclei.[3] The ontogeny of the T. crypta sponge is largely affected by this process of sediment incorporation and organization. Differentiation between smaller and larger sediments and their corresponding location has proven useful in identifying possible functioning of positioning of these particles on the surface of the sponge. Smaller, fine sediments are packed in the nuclei within the body of the sponge while the more coarse grains are located towards the base of the sponge; this localization helps in anchoring and stabilizing the sponge with the help of gravity.[9] The sediments are involved in part with the morphogenesis of the animal. The forming of the nuclei clusters stabilizes the sponge's body, allowing the animal to alter its skeleton structure. A radial morphology is then able to change into a branched one, which further allows the animal to develop into its massive, irregular fully-formed shape.
Feeding
T. crypta are filter feeders, utilizing their choanocytes to generate an inward current and pulling in their nutrients. The course of action of filter feeding goes as follows: ostium, spongocoel, and osculum. In the middle of this route, nutrients may be absorbed and taken in by the sponge to utilize. T. crypta generally eat the following organisms: Chaetoceros, pinnulaira, striatella unipunctata, and skeleronema tropicum.[10]
Ecology
Tectitethya crypta can be found in shallow water, only about 1 to 20 meters in depth within the Caribbean.[6] It dwells on a soft substrate, typically substances such as muds, sands, or clays. It can geographically be located in a reef near the Florida Keys, Dry Tortugas, and north-west shores of Cuba, as well as the Florida west coast.[6] The larger of the sponges, sizing around 1.5-10 liters in volume are typically found attached to their substrate while the smaller sponges of this species, sizing around 0.5-1.5 liters in volume are typically found to be unattached and resting freely on their bottom.[3]
Human relations
Medicine
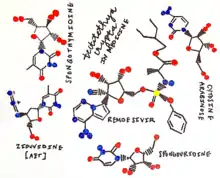
The discovery of T. crypta allowed for the discovery of the first sponge-derived pharmaceutical drugs. The two nucleosides, spongothymidine and spongouridine, are documented as the two nucleoside analogues used in the synthesis of life-saving drugs today. These are natural products - not artificially synthesized. Marine natural products (MNP's) have been shown to have stronger bioactive properties than those from terrestrial organisms, possessing cytotoxic and antiproliferative agents.[11] Understanding this has allowed scientists to recognize the role that these potent chemicals may play in chemical defense mechanisms and protection from prey. This may be the case for T. crypta, as it's a sessile organism not possessing an immune system.[12] The treatment of leukemia through the use of Ara-C (cytarabine) is the first documented anticancer agent that has come about from the sponge.[13] In fact, it was approved by the FDA in 1969 in treating non-Hodgkin's lymphoma and myeloid and myelocytic leukemia.[12] As of today, cytarabine is one of the greatest contributors towards anti-cancer therapies.[14] The drug disables Deoxyribonucleic Acid Polymerase, inhibiting DNA synthesis during the S phase of the cell cycle.[6] This discovery allowed for scientists to manipulate the replication of viral DNA within its host and put a complete halt in its division. This hallmark discovery led to the development of azidothymidine (AZT) through the use of Ara-A. Azidothymidine is utilized in the treatment of HIV-infected individuals. Vidarabine (Ara-A) alone is used in ophthalmologic applications today.[12] A fluorinated derivative of Ara-C has contributed towards the advancement of treatment for lung, pancreatic, breast, and bladder cancer.[15] This drug is known as gemcitabine — proven useful in its effectiveness against solid tumors such as these.[14] Manipulation of these two original nucleoside analogues provided by T. crypta has provided scientists and medical professionals the capability to offer humans potential cures to devastating diseases — and has inspired the future of medicine to search for "natural" cures in the sea.
References
- van Soest, R. (2008). Van Soest RW, Boury-Esnault N, Hooper JN, Rützler K, de Voogd NJ, de Glasby BA, Hajdu E, Pisera AB, Manconi R, Schoenberg C, Janussen D, Tabachnick KR, Klautau M, Picton B, Kelly M, Vacelet J (eds.). "Tectitethya crypta (de Laubenfels, 1949)". World Porifera database. World Register of Marine Species. Retrieved 8 April 2017.
- Sarà, Michele (2002), Hooper, John N. A.; Van Soest, Rob W. M.; Willenz, Philippe (eds.), "Family Tethyidae Gray, 1848", Systema Porifera, Boston, MA: Springer US, pp. 245–265, doi:10.1007/978-1-4615-0747-5_26, ISBN 978-0-306-47260-2, retrieved 2020-12-03
- Cerrano, Carlo; Pansini, Maurizio; Valisano, Laura; Calcinai, Barbara; Sarà, Michele; Bavestrello, Giorgio (2004). "Lagoon sponges from Carrie Bow Cay (Belize): Ecological benefits of selective sediment incorporation". Bollettino dei Musei e degli Istituti Biologici dell'Università di Genova. 68: 239–252. Retrieved 23 June 2012.
- Patricia R. Bergquist (1978). Sponges. University of California Press. p. 205. ISBN 978-0-520-03658-1. Retrieved 23 June 2012.
- Pérez, Thierry; Díaz, Maria-Cristina; Ruiz, César; Cóndor-Luján, Baslavi; Klautau, Michelle; Hajdu, Eduardo; Lobo-Hajdu, Gisele; Zea, Sven; Pomponi, Shirley A.; Thacker, Robert W.; Carteron, Sophie (2017-03-22). "How a collaborative integrated taxonomic effort has trained new spongiologists and improved knowledge of Martinique Island (French Antilles, eastern Caribbean Sea) marine biodiversity". PLOS ONE. 12 (3): e0173859. Bibcode:2017PLoSO..1273859P. doi:10.1371/journal.pone.0173859. ISSN 1932-6203. PMC 5362083. PMID 28329020.
- O’Donnell, Nicole (2012-06-01). "Book Review: Gulf of Mexico Origin, Waters, and Biota: Biodiversity (Volume 1)". Aquatic Mammals. 38 (2): 223. doi:10.1578/am.38.2.2012.223. ISSN 0167-5427.
- Sagar, Sunil; Kaur, Mandeep; Minneman, Kenneth P. (2010). "Antiviral lead compounds from marine sponges". Marine Drugs. 8 (10): 2619–2638. doi:10.3390/md8102619. PMC 2992996. PMID 21116410.
- Schwartsmann, G; Brondani da Rocha, A; Berlinck, RG; Jimeno, J (April 2001). "Marine organisms as a source of new anticancer agents". Lancet Oncology. 2 (4): 221–225. doi:10.1016/s1470-2045(00)00292-8. PMID 11905767.
- Calcinai, Barbara; Cerrano, Carlo; Sarà, Michele; Bavestrello, Giorgio (2000). "Boring sponges (Porifera, Demospongiae) from the Indian Ocean". Italian Journal of Zoology. 67 (2): 203–219. doi:10.1080/11250000009356314. ISSN 1125-0003. S2CID 84082407.
- "Tectitethya crypta (de Laubenfels 1949) data - Encyclopedia of Life". eol.org. Retrieved 2020-12-03.
- Altmann, Karl-Heinz (2017-10-25). "Drugs from the Oceans: Marine Natural Products as Leads for Drug Discovery". CHIMIA International Journal for Chemistry. 71 (10): 646–652. doi:10.2533/chimia.2017.646. ISSN 0009-4293. PMID 29070409.
- Hansen KØ, Isaksson J, Bayer A, Johansen JA, Andersen JH, Hansen E (2017). "Securamine Derivatives from the Arctic Bryozoan Securiflustra securifrons". Journal of Natural Products. 80 (12): 3276–3283. doi:10.1021/acs.jnatprod.7b00703.s001.
- Essack, Magbubah; Bajic, Vladimir B.; Archer, John A. C. (September 20, 2011). "Recently Confirmed Apoptosis-Inducing Lead Compounds Isolated from Marine Sponge of Potential Relevance in Cancer Treatment". Marine Drugs. 9 (9): 1580–1606. doi:10.3390/md9091580. PMC 3225937. PMID 22131960.
- Schwartsmann, Gilberto; da Rocha, Adriana Brondani; Berlinck, Roberto GS; Jimeno, Jose (April 2001). "Marine organisms as a source of new anticancer agents". The Lancet Oncology. 2 (4): 221–225. doi:10.1016/s1470-2045(00)00292-8. ISSN 1470-2045. PMID 11905767.
- Anjum, Komal; Abbas, Syed Qamar; Shah, Sayed Asmat Ali; Akhter, Najeeb; Batool, Sundas; Hassan, Syed Shams ul (July 2016). "Marine Sponges as a Drug Treasure". Biomolecules & Therapeutics. 24 (4): 347–362. doi:10.4062/biomolther.2016.067. ISSN 1976-9148. PMC 4930278. PMID 27350338.