Tetrakis(3,5-bis(trifluoromethyl)phenyl)borate
Tetrakis[3,5-bis(trifluoromethyl)phenyl]borate is an anion with chemical formula [{3,5-(CF3)2C6H3}4B]−, which is commonly abbreviated as [BArF4]−, indicating the presence of fluorinated aryl (ArF) groups. It is sometimes referred to as Kobayashi's anion in honour of Hiroshi Kobayashi who led the team that first synthesised it.[1] More commonly it is affectionately nicknamed "BARF."[2] The BARF ion is also abbreviated BArF24−,[2] to distinguish it from the closely related BArF−
20, [(C6F5)4B]−.
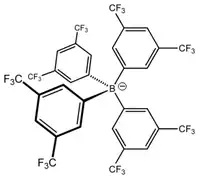
BARF has a tetrahedral geometry around the central boron atom but each of the four surrounding aryl groups is aromatic and planar. The motivation for its preparation was the search for an anion that coordinates more weakly than the then-available ions hexafluorophosphate, tetrafluoroborate, or perchlorate.[3] Salts of this anion are known as solids and in both aqueous and non-aqueous solutions. BARF can be used in catalytic systems where the active site requires an anion which will not coordinate to the metal centre and interfere with the catalytic cycle, such as in the preparation of polyketones.[4]
borate-2D-skeletal.png.webp)
Synthesis of BARF compounds
The sodium salt is the starting point for most BARF derivatives. It is prepared by treating Grignard reagents derived from XC6H3-3,5-(CF3)2 (X = Br, I) with NaBF4. A popular method is summarized in the following equation:[5]
- NaBF4 + 4 ArFMgBr → 4 MgBrF + NaBArF4
Brookhart's acid is the salt of the BARF anion with the diethyl ether oxonium cation, [(Et2O)2H]BArF4. It can be formed from the sodium salt in diethyl ether in the presence of hydrogen chloride as sodium chloride is insoluble in diethyl ether, facilitating cation exchange.[3]
- NaBArF4 + HCl + 2 Et2O → [(Et2O)2H]BArF4 + NaCl
BARF salts with hexakis(acetonitrile)metal(II) cations, [M(CH3CN)6]2+, are known for vanadium, chromium, manganese, iron, cobalt, and nickel. They are produced by salt metathesis reactions.[6]
Properties
Non-coordinating anions are anions that interact only weakly with cations, a useful property when studying highly electrophilic cations.[7] In coordination chemistry, the term can also be used to refer to anions which are unlikely to bind directly to the metal centre of a complex. Hexafluorophosphate is a non-coordinating anion in both senses of the term.[8][9] Three widely used non-coordinating anions are hexafluorophosphate, tetrafluoroborate BF−
4, and perchlorate ClO−
4; of these, the hexafluorophosphate ion has the least coordinating ability[10] and it is deliberately used for this property. BARF was developed as a new non-coordinating anion in the 1990s, and is far less coordinating than even the hexafluorophosphate anion.[3] Extremely Lewis acidic metal centers can, however, cleave the carbon-boron bond in BARF.[11]
NaBArF4 can be used in deprotection of acetal or ketal-protected carbonyl compounds.[12][13] For example, deprotection of 2-phenyl-1,3-dioxolane to benzaldehyde can be achieved in water in five minutes at 30 °C.[14]
- PhCH(OCH2)2 + H2O PhCHO + HOCH2CH2OH
References
- Nishida, H.; Takada, N.; Yoshimura, M.; Sonods, T.; Kobayshi, H. (1984). "Tetrakis(3,5-bis(trifluoromethyl)phenyl)borate. Highly Lipophilic Stable Anionic Agent for Solvent-Extraction of Cations". Bull. Chem. Soc. Jpn. 57 (9): 2600–2604. doi:10.1246/bcsj.57.2600.
- Yakelis, N. A.; Bergman, R. G. (2005). "Safe Preparation and Purification of Sodium Tetrakis[(3,5-trifluoromethyl)phenyl]borate (NaBArF24): Reliable and Sensitive Analysis of Water in Solutions of Fluorinated Tetraarylborates". Organometallics. 24 (14): 3579–3581. doi:10.1021/om0501428. PMC 2600718. PMID 19079785.
- Brookhart, M.; Grant, B.; Volpe, A. F. (1992). "[(3,5-(CF3)2C6H3)4B]−[H(OEt2)2]+: A Convenient Reagent for Generation and Stabilization of Cationic, Highly Electrophilic Organometallic Complexes". Organometallics. 11 (11): 3920–3922. doi:10.1021/om00059a071.
- Brookhart, M.; Rix, F. C.; DeSimone, J. M.; Barborak, J. C. (1992). "Palladium(II) catalysts for living alternating copolymerization of olefins and carbon monoxide". J. Am. Chem. Soc. 114 (14): 5894–5895. doi:10.1021/ja00040a082.
- Smith, C. R.; Zhang, A.; Mans, D. J.; RajanBabu, T. V. (2008). "(R)-3-Methyl-3-Phenyl-1-Pentene via Catalytic Asymmetric Hydrovinylation". Org. Synth. 85: 248–266. doi:10.15227/orgsyn.085.0248. PMC 2723857. PMID 19672483.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - Buschman, W. E.; Miller, J. S. (2002). "Synthesis of [MII(NCMe)6]2+ (M = V, Cr, Mn, Fe, Co, Ni) salts of tetra[3,5-bis(trifluoromethyl)phenyl]borate". Inorg. Synth. 33: 83. doi:10.1002/0471224502.ch2.
- Krossing, I.; Raabe, I. (2004). "Noncoordinating Anions - Fact or Fiction? A Survey of Likely Candidates". Angew. Chem. Int. Ed. 43 (16): 2066–2090. doi:10.1002/anie.200300620. PMID 15083452.
- Davies, J. A. (1996). Synthetic Coordination Chemistry: Principles and Practice. World Scientific. p. 165. ISBN 9810220847.
- Constant, S.; Lacour, J. (2005). Majoral, J.-P. (ed.). New Trends in Hexacoordinated Phosphorus Chemistry. New Aspects in Phosphorus Chemistry. Vol. 5. Springer. p. 3. ISBN 354022498X.
- Mayfield, H. G.; Bull, W. E. (1971). "Co-ordinating Tendencies of the Hexafluorophosphate Ion". J. Chem. Soc. A (14): 2279–2281. doi:10.1039/J19710002279.
- Salem, Hiyam; Shimon, Linda J. W.; Leitus, Gregory; Weiner, Lev; Milstein, David (2008-05-01). "B−C Bond Cleavage of BArF Anion Upon Oxidation of Rhodium(I) with AgBArF. Phosphinite Rhodium(I), Rhodium(II), and Rhodium(III) Pincer Complexes". Organometallics. 27 (10): 2293–2299. doi:10.1021/om800034t. ISSN 0276-7333.
- Greene, Theodora W.; Wuts, Peter G. M. (1999). "Dimethyl acetals". Greene's Protective Groups in Organic Synthesis (3rd ed.). Wiley-Interscience. pp. 297–304, 724–727. ISBN 9780471160199. Archived from the original on December 3, 2016. Retrieved June 20, 2017.
- Greene, Theodora W.; Wuts, Peter G. M. (1999). "1,3-Dioxanes, 1,3-Dioxolanes". Greene's Protective Groups in Organic Synthesis (3rd ed.). Wiley-Interscience. pp. 308–322, 724–727. ISBN 9780471160199. Archived from the original on December 7, 2016. Retrieved June 20, 2017.
- Chang, Chih-Ching; Liao, Bei-Sih; Liu, Shiuh-Tzung (2007). "Deprotection of Acetals and Ketals in a Colloidal Suspension Generated by Sodium Tetrakis(3,5-trifluoromethylphenyl)borate in Water". Synlett. 2007 (2): 283–287. doi:10.1055/s-2007-968009.