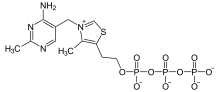
Thiamine triphosphate
Thiamine triphosphate (ThTP) is a biomolecule found in most organisms including bacteria, fungi, plants and animals.[1] Chemically, it is the triphosphate derivative of the vitamin thiamine.
 | |
| Identifiers | |
|---|---|
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| MeSH | Thiamine+triphosphate |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C12H19N4O10P3S | |
| Molar mass | 504.288 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Function
It has been proposed that ThTP has a specific role in nerve excitability,[2] but this has never been confirmed and recent results suggest that ThTP probably plays a role in cell energy metabolism.[1][3] Low or absent levels of thiamine triphosphate have been found in Leighs disease.[4] [5]
In E. coli, ThTP is accumulated in the presence of glucose during amino acid starvation.[1][3] On the other hand, suppression of the carbon source leads to the accumulation, of adenosine thiamine triphosphate (AThTP).
Metabolism
It has been shown that in brain ThTP is synthesized in mitochondria by a chemiosmotic mechanism, perhaps similar to ATP synthase.[6] In mammals, ThTP is hydrolyzed to thiamine pyrophosphate (ThDP) by a specific thiamine-triphosphatase.[3][7] It can also be converted into ThDP by thiamine-diphosphate kinase.
History
Thiamine triphosphate (ThTP) was chemically synthesized in 1948 at a time when the only organic triphosphate known was ATP.[8] The first claim of the existence of ThTP in living organisms was made in rat liver,[9] followed by baker’s yeast.[10] Its presence was later confirmed in rat tissues[11] and in plants germs, but not in seeds, where thiamine was essentially unphosphorylated.[12] In all those studies, ThTP was separated from other thiamine derivatives using a paper chromatographic method, followed by oxidation in fluorescent thiochrome compounds with ferricyanide in alkaline solution. This method is at best semi-quantitative, and the development of liquid chromatographic methods suggested that ThTP represents far less than 10% of total thiamine in animal tissues.[13]
References
- Makarchikov AF, Lakaye B, Gulyai IE, Czerniecki J, Coumans B, Wins P, Grisar T, Bettendorff L (2003). "Thiamine triphosphate and thiamine triphosphatase activities: from bacteria to mammals". Cell. Mol. Life Sci. 60 (7): 1477–88. doi:10.1007/s00018-003-3098-4. PMID 12943234. S2CID 25400487.
- Matsuda, T; Cooper, JR (1981). "Thiamine as an integral component of brain synaptosomal membranes". Proceedings of the National Academy of Sciences of the United States of America. 78 (9): 5886–9. Bibcode:1981PNAS...78.5886M. doi:10.1073/pnas.78.9.5886. PMC 348897. PMID 6272323.
- Lakaye, B.; Makarchikov, AF; Antunes, AF; Zorzi, W; Coumans, B; De Pauw, E; Wins, P; Grisar, T; Bettendorff, L (2002). "Molecular Characterization of a Specific Thiamine Triphosphatase Widely Expressed in Mammalian Tissues". Journal of Biological Chemistry. 277 (16): 13771–7. doi:10.1074/jbc.M111241200. PMID 11827967.
- Murphy, J. V.; Craig, L. (1975). "Leigh's disease: Significance of the biochemical changes in brain". Journal of Neurology, Neurosurgery, and Psychiatry. 38 (11): 1100–1103. doi:10.1136/jnnp.38.11.1100. PMC 492163. PMID 1206418.
- Murphy, Jerome V. (1974). "Leigh Disease". Archives of Neurology. 31 (4): 220. doi:10.1001/archneur.1974.00490400034002.
- Gangolf, M.; Wins, P.; Thiry, M.; El Moualij, B.; Bettendorff, L. (2009). "Thiamine Triphosphate Synthesis in Rat Brain Occurs in Mitochondria and is Coupled to the Respiratory Chain". Journal of Biological Chemistry. 285 (1): 583–94. doi:10.1074/jbc.M109.054379. PMC 2804207. PMID 19906644.
- Song, J.; Bettendorff, L.; Tonelli, M.; Markley, J. L. (2008). "Structural Basis for the Catalytic Mechanism of Mammalian 25-kDa Thiamine Triphosphatase". Journal of Biological Chemistry. 283 (16): 10939–48. doi:10.1074/jbc.M709675200. PMC 2447667. PMID 18276586.
- Velluz L., Amiard G. & Bartos J. (1948). "Un nouveau derive de phosphorylation de la vitamine=B1, lacide thiamine triphosphorique". J. Bull. Soc. Chim. Fr. 15: 871.
- Rossi-Fanelli A., Siliprandi N. & Fasella P. (1952). "On the presence of triphosphothiamine (TPT) in the liver". Science. 116 (3026): 711–713. Bibcode:1952Sci...116..711R. doi:10.1126/science.116.3026.711. PMID 13028255.
- Kiessling K.-H. (1953). "Thiamine triphosphate in baker's yeast". Nature. 172 (4391): 1187–1188. Bibcode:1953Natur.172.1187K. doi:10.1038/1721187a0. PMID 13111284. S2CID 4289038.
- Greiling H. & Kiesow L. (1958). "Zur Biochemie der Thiamintriphosphorsäure IV. Das Vorkommen von Thiamintriphosphat im tierischen Organismus". Zeitschrift für Naturforschung B. 13: 251–252. doi:10.1515/znb-1958-0412.
- Yusa T. (1962). "Studies on thiamine triphosphate II. Thiamine triphosphate as phosphate donor". Plant Cell. 3: 95–103.
- Rindi G. & de Giuseppe L. (1961). "A new chromatographic method for the determination of thiamine and its mono-, di- and triphosphates in animal tissues". Biochem. J. 78 (3): 602–606. doi:10.1042/bj0780602. PMC 1205381. PMID 13741738.