Ventricular zone
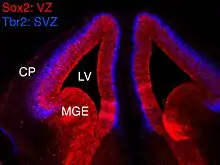
In vertebrates, the ventricular zone (VZ) is a transient embryonic layer of tissue containing neural stem cells, principally radial glial cells, of the central nervous system (CNS).[1][2] The VZ is so named because it lines the ventricular system, which contains cerebrospinal fluid (CSF). The embryonic ventricular system contains growth factors and other nutrients needed for the proper function of neural stem cells.[3] Neurogenesis, or the generation of neurons, occurs in the VZ during embryonic and fetal development as a function of the Notch pathway,[4][5] and the newborn neurons must migrate substantial distances to their final destination in the developing brain or spinal cord where they will establish neural circuits.[6][7] A secondary proliferative zone, the subventricular zone (SVZ), lies adjacent to the VZ. In the embryonic cerebral cortex, the SVZ contains intermediate neuronal progenitors that continue to divide into post-mitotic neurons.[8][9] Through the process of neurogenesis, the parent neural stem cell pool is depleted and the VZ disappears.[10] The balance between the rates of stem cell proliferation and neurogenesis changes during development,[11] and species from mouse to human show large differences in the number of cell cycles, cell cycle length, and other parameters, which is thought to give rise to the large diversity in brain size and structure.
Epigenetic DNA modifications appear to have a central role in regulating gene expression during differentiation of neural stem cells. One type of epigenetic modification occurring in the VZ is the formation of DNA 5-Methylcytosine from cytosine by DNA methyltransferases.[12] Another important type of epigenetic modification is the demethylation of 5mC, catalyzed in several steps by TET enzymes and enzymes of the base excision repair pathway.[12]
See also
References
- Rakic, P (October 2009). "Evolution of the neocortex: a perspective from developmental biology". Nature Reviews. Neuroscience. 10 (10): 724–35. doi:10.1038/nrn2719. PMC 2913577. PMID 19763105.
- Noctor, SC; Flint, AC; Weissman, TA; Dammerman, RS; Kriegstein, AR (8 February 2001). "Neurons derived from radial glial cells establish radial units in neocortex". Nature. 409 (6821): 714–20. doi:10.1038/35055553. PMID 11217860.
- Lehtinen, MK; Zappaterra, MW; Chen, X; Yang, YJ; Hill, AD; Lun, M; Maynard, T; Gonzalez, D; Kim, S; Ye, P; D'Ercole, AJ; Wong, ET; LaMantia, AS; Walsh, CA (10 March 2011). "The cerebrospinal fluid provides a proliferative niche for neural progenitor cells". Neuron. 69 (5): 893–905. doi:10.1016/j.neuron.2011.01.023. PMC 3085909. PMID 21382550.
- Kageyama, R; Ohtsuka, T; Shimojo, H; Imayoshi, I (November 2008). "Dynamic Notch signaling in neural progenitor cells and a revised view of lateral inhibition". Nature Neuroscience. 11 (11): 1247–51. doi:10.1038/nn.2208. PMID 18956012.
- Rash, BG; Lim, HD; Breunig, JJ; Vaccarino, FM (26 October 2011). "FGF signaling expands embryonic cortical surface area by regulating Notch-dependent neurogenesis". The Journal of Neuroscience. 31 (43): 15604–17. doi:10.1523/jneurosci.4439-11.2011. PMC 3235689. PMID 22031906.
- Rakic, P (March 1971). "Neuron-glia relationship during granule cell migration in developing cerebellar cortex. A Golgi and electronmicroscopic study in Macacus Rhesus". The Journal of Comparative Neurology. 141 (3): 283–312. doi:10.1002/cne.901410303. PMID 4101340.
- Rakic, P (May 1972). "Mode of cell migration to the superficial layers of fetal monkey neocortex". The Journal of Comparative Neurology. 145 (1): 61–83. doi:10.1002/cne.901450105. PMID 4624784.
- Noctor, SC; Martínez-Cerdeño, V; Ivic, L; Kriegstein, AR (February 2004). "Cortical neurons arise in symmetric and asymmetric division zones and migrate through specific phases". Nature Neuroscience. 7 (2): 136–44. doi:10.1038/nn1172. PMID 14703572.
- Hevner, RF; Haydar, TF (February 2012). "The (not necessarily) convoluted role of basal radial glia in cortical neurogenesis". Cerebral Cortex. 22 (2): 465–8. doi:10.1093/cercor/bhr336. PMC 3256413. PMID 22116731.
- Dehay, C; Kennedy, H (June 2007). "Cell-cycle control and cortical development". Nature Reviews. Neuroscience. 8 (6): 438–50. doi:10.1038/nrn2097. PMID 17514197.
- Takahashi, T; Nowakowski, RS; Caviness VS, Jr (1 October 1996). "The leaving or Q fraction of the murine cerebral proliferative epithelium: a general model of neocortical neuronogenesis". The Journal of Neuroscience. 16 (19): 6183–96. PMID 8815900.
- Wang Z, Tang B, He Y, Jin P. DNA methylation dynamics in neurogenesis. Epigenomics. 2016 Mar;8(3):401-14. doi:10.2217/epi.15.119. Epub 2016 Mar 7. Review. PMID 26950681