von Braun reaction
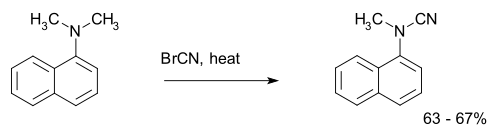
The von Braun reaction is an organic reaction in which a tertiary amine reacts with cyanogen bromide to an organocyanamide.[1] An example is the reaction of N,N-dimethyl-1-naphthylamine:[2]
| von Braun reaction | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Named after | Julius von Braun | ||||||||
| Reaction type | Substitution reaction | ||||||||
| Reaction | |||||||||
| |||||||||
These days, most chemist have replaced cyanogen bromide reagent with chloroethyl chloroformate reagent instead. It appears as though Olofson et al. was the first chemist to have reported this.[3]
Reaction mechanism
The reaction mechanism consists of two nucleophilic substitutions: the amine is the first nucleophile displacing the bromine atom which then acts as the second nucleophile.[4][5] In following the mechanism is described using trimethylamine as example:[6]

First, the trimethylamine reacts with the cyanogen bromide to form a quaternary ammonium salt, which in the next step reacts by splitting off bromomethane to give the dimethylcyanamide. This is a second-order nucleophilic substitution (SN2).
See also
References
- J. von Braun; K. Heider & E. Müller (1918). "Bromalkylierte aromatische Amine. II. Mitteilung". Chem. Ber. 51 (1): 273–282. doi:10.1002/cber.19180510132.
- Homer W. J. Cressman (1947). "N-Methyl-1-Naphthylcyanamide". Org. Synth. 27: 56. doi:10.15227/orgsyn.027.0056.
- J. Org. Chem. 1984, 49, 11, 2081–2082 Publication Date:June 1, 1984 https://doi.org/10.1021/jo00185a072
- March, Jerry (1985), Advanced Organic Chemistry: Reactions, Mechanisms, and Structure, 3rd edition, New York: Wiley, ISBN 9780471854722, OCLC 642506595
- Howard A. Hageman (1953). "The Von Braun Cyanogen Bromide Reaction". Organic Reactions. 7 (4): 198–262. doi:10.1002/0471264180.or007.04. ISBN 0471264180.
- Jie Jack Li (2014), Name reactions: A collection of detailed mechanisms and synthetic applications (in German) (5th ed.), Cham: Springer, p. 619, doi:10.1007/978-3-319-03979-4, ISBN 978-3-319-03979-4, S2CID 93616494