Adenine nucleotide translocator
Adenine nucleotide translocator (ANT), also known as the ADP/ATP translocase (ANT), ADP/ATP carrier protein (AAC) or mitochondrial ADP/ATP carrier, exchanges free ATP with free ADP across the inner mitochondrial membrane.[1][2] ANT is the most abundant protein in the inner mitochondrial membrane and belongs to mitochondrial carrier family.[3]
| ADP/ATP translocases | |||||||||
|---|---|---|---|---|---|---|---|---|---|
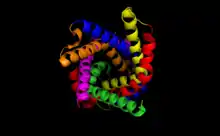 Cytoplasmic view of the binding pocket of ATP–ADP translocase 1, PDB: 1OKC. | |||||||||
| Identifiers | |||||||||
| Symbol | Aden_trnslctor | ||||||||
| Pfam | PF00153 | ||||||||
| InterPro | IPR002113 | ||||||||
| TCDB | 2.A.29.1.2 | ||||||||
| OPM superfamily | 21 | ||||||||
| OPM protein | 2c3e | ||||||||
| |||||||||
| solute carrier family 25 (mitochondrial carrier; adenine nucleotide translocator), member 4 | |||||||
|---|---|---|---|---|---|---|---|
| Identifiers | |||||||
| Symbol | SLC25A4 | ||||||
| Alt. symbols | PEO3, PEO2, ANT1 | ||||||
| NCBI gene | 291 | ||||||
| HGNC | 10990 | ||||||
| OMIM | 103220 | ||||||
| RefSeq | NM_001151 | ||||||
| UniProt | P12235 | ||||||
| Other data | |||||||
| Locus | Chr. 4 q35 | ||||||
| |||||||
| solute carrier family 25 (mitochondrial carrier; adenine nucleotide translocator), member 5 | |||||||
|---|---|---|---|---|---|---|---|
| Identifiers | |||||||
| Symbol | SLC25A5 | ||||||
| Alt. symbols | ANT2 | ||||||
| NCBI gene | 292 | ||||||
| HGNC | 10991 | ||||||
| OMIM | 300150 | ||||||
| RefSeq | NM_001152 | ||||||
| UniProt | P05141 | ||||||
| Other data | |||||||
| Locus | Chr. X q24-q26 | ||||||
| |||||||
| solute carrier family 25 (mitochondrial carrier; adenine nucleotide translocator), member 6 | |||||||
|---|---|---|---|---|---|---|---|
| Identifiers | |||||||
| Symbol | SLC25A6 | ||||||
| Alt. symbols | ANT3 | ||||||
| NCBI gene | 293 | ||||||
| HGNC | 10992 | ||||||
| OMIM | 403000 | ||||||
| RefSeq | NM_001636 | ||||||
| UniProt | P12236 | ||||||
| Other data | |||||||
| Locus | Chr. Y p | ||||||
| |||||||
Free ADP is transported from the cytoplasm to the mitochondrial matrix, while ATP produced from oxidative phosphorylation is transported from the mitochondrial matrix to the cytoplasm, thus providing the cells with its main energy currency.[4] ADP/ATP translocases are exclusive to eukaryotes and are thought to have evolved during eukaryogenesis.[5] Human cells express four ADP/ATP translocases: SLC25A4, SLC25A5, SLC25A6 and SLC25A31, which constitute more than 10% of the protein in the inner mitochondrial membrane.[6] These proteins are classified under the mitochondrial carrier superfamily.
Types
In humans, there exist three paraologous ANT isoforms:
- SLC25A4 – found primarily in heart and skeletal muscle
- SLC25A5 – primarily expressed in fibroblasts
- SLC25A6 – primarily express in liver
Structure
_drawn_over.png.webp)

ANT has long been thought to function as a homodimer, but this concept was challenged by the projection structure of the yeast Aac3p solved by electron crystallography, which showed that the protein was three-fold symmetric and monomeric, with the translocation pathway for the substrate through the centre.[7] The atomic structure of the bovine ANT confirmed this notion, and provided the first structural fold of a mitochondrial carrier.[8] Further work has demonstrated that ANT is a monomer in detergents [9] and functions as a monomer in mitochondrial membranes.[10][11]
ADP/ATP translocase 1 is the major AAC in human cells and the archetypal protein of this family. It has a mass of approximately 30 kDa, consisting of 297 residues.[12] It forms six transmembrane α-helices that form a barrel that results in a deep cone-shaped depression accessible from the outside where the substrate binds. The binding pocket, conserved throughout most isoforms, mostly consists of basic residues that allow for strong binding to ATP or ADP and has a maximal diameter of 20 Å and a depth of 30 Å.[8] Indeed, arginine residues 96, 204, 252, 253, and 294, as well as lysine 38, have been shown to be essential for transporter activity.[13]
Function
ADP/ATP translocase transports ATP synthesized from oxidative phosphorylation into the cytoplasm, where it can be used as the principal energy currency of the cell to power thermodynamically unfavorable reactions. After the consequent hydrolysis of ATP into ADP, ADP is transported back into the mitochondrial matrix, where it can be rephosphorylated to ATP. Because a human typically exchanges the equivalent of his/her own mass of ATP on a daily basis, ADP/ATP translocase is an important transporter protein with major metabolic implications.[4][8]
ANT transports the free, i.e. deprotonated, non-Magnesium, non-Calcium bound forms of ADP and ATP, in a 1:1 ratio.[1] Transport is fully reversible, and its directionality is governed by the concentrations of its substrates (ADP and ATP inside and outside mitochondria), the chelators of the adenine nucleotides, and the mitochondrial membrane potential. The relationship of these parameters can be expressed by an equation solving for the 'reversal potential of the ANT" (Erev_ANT), a value of the mitochondrial membrane potential at which no net transport of adenine nucleotides takes place by the ANT.[14][15][16] The ANT and the F0-F1 ATP synthase are not necessarily in directional synchrony.[14]
Apart from exchange of ADP and ATP across the inner mitochondrial membrane, the ANT also exhibits an intrinsic uncoupling activity[1][17]
ANT is an important modulatory[18] and possible structural component of the Mitochondrial Permeability Transition Pore, a channel involved in various pathologies whose function still remains elusive. Karch et al. propose a "multi-pore model" in which ANT is at least one of the molecular components of the pore.[19]
Translocase mechanism
Under normal conditions, ATP and ADP cannot cross the inner mitochondrial membrane due to their high negative charges, but ADP/ATP translocase, an antiporter, couples the transport of the two molecules. The depression in ADP/ATP translocase alternatively faces the matrix and the cytoplasmic sides of the membrane. ADP in the intermembrane space, coming from the cytoplasm, binds the translocase and induces its eversion, resulting in the release of ADP into the matrix. Binding of ATP from the matrix induces eversion and results in the release of ATP into the intermembrane space, subsequently diffusing to the cytoplasm, and concomitantly brings the translocase back to its original conformation.[4] ATP and ADP are the only natural nucleotides recognized by the translocase.[8]
The net process is denoted by:
- ADP3−cytoplasm + ATP4−matrix → ADP3−matrix + ATP4−cytoplasm
ADP/ATP exchange is energetically expensive: about 25% of the energy yielded from electron transfer by aerobic respiration, or one hydrogen ion, is consumed to regenerate the membrane potential that is tapped by ADP/ATP translocase.[4]
The translocator cycles between two states, called the cytoplasmic and matrix state, opening up to these compartments in an alternating way.[1][2] There are structures available that show the translocator locked in a cytoplasmic state by the inhibitor carboxyatractyloside,[8][20] or in the matrix state by the inhibitor bongkrekic acid.[21]
Alterations
Rare but severe diseases such as mitochondrial myopathies are associated with dysfunctional human ADP/ATP translocase. Mitochondrial myopathies (MM) refer to a group of clinically and biochemically heterogeneous disorders that share common features of major mitochondrial structural abnormalities in skeletal muscle. The major morphological hallmark of MM is ragged, red fibers containing peripheral and intermyofibrillar accumulations of abnormal mitochondria.[22][23] In particular, autosomal dominant progressive external ophthalmoplegia (adPEO) is a common disorder associated with dysfunctional ADP/ATP translocase and can induce paralysis of muscles responsible for eye movements. General symptoms are not limited to the eyes and can include exercise intolerance, muscle weakness, hearing deficit, and more. adPEO shows Mendelian inheritance patterns but is characterized by large-scale mitochondrial DNA (mtDNA) deletions. mtDNA contains few introns, or non-coding regions of DNA, which increases the likelihood of deleterious mutations. Thus, any modification of ADP/ATP translocase mtDNA can lead to a dysfunctional transporter,[24] particularly residues involved in the binding pocket which will compromise translocase efficacy.[13] MM is commonly associated with dysfunctional ADP/ATP translocase, but MM can be induced through many different mitochondrial abnormalities.
Inhibition

ADP/ATP translocase is very specifically inhibited by two families of compounds. The first family, which includes atractyloside (ATR) and carboxyatractyloside (CATR), binds to the ADP/ATP translocase from the cytoplasmic side, locking it in a cytoplasmic side open conformation. In contrast, the second family, which includes bongkrekic acid (BA) and isobongkrekic acid (isoBA), binds the translocase from the matrix, locking it in a matrix side open conformation.[7] The negatively charged groups of the inhibitors bind strongly to the positively charged residues deep within the binding pocket. The high affinity (Kd in the nanomolar range) makes each inhibitor a deadly poison by obstructing cellular respiration/energy transfer to the rest of the cell.[8] There are structures available that show the translocator locked in a cytoplasmic state by the inhibitor carboxyatractyloside,[8][20] or in the matrix state by the inhibitor bongkrekic acid.[21]
History
In 1955, Siekevitz and Potter demonstrated that adenine nucleotides were distributed in cells in two pools located in the mitochondrial and cytosolic compartments.[25] Shortly thereafter, Pressman hypothesized that the two pools could exchange nucleotides.[26] However, the existence of an ADP/ATP transporter was not postulated until 1964 when Bruni et al. uncovered an inhibitory effect of atractyloside on the energy-transfer system (oxidative phosphorylation) and ADP binding sites of rat liver mitochondria.[27] Soon after, an overwhelming amount of research was done in proving the existence and elucidating the link between ADP/ATP translocase and energy transport.[28][29][30] cDNA of ADP/ATP translocase was sequenced for bovine in 1982[31] and a yeast species Saccharomyces cerevisiae in 1986[32] before finally Battini et al. sequenced a cDNA clone of the human transporter in 1989. The homology in the coding sequences between human and yeast ADP/ATP translocase was 47% while bovine and human sequences extended remarkable to 266 out of 297 residues, or 89.6%. In both cases, the most conserved residues lie in the ADP/ATP substrate binding pocket.[12]
See also
- Mitochondrial carrier
- Cellular respiration
- Oxidative phosphorylation
References
- Klingenberg M (October 2008). "The ADP and ATP transport in mitochondria and its carrier". Biochimica et Biophysica Acta (BBA) - Biomembranes. 1778 (10): 1978–2021. doi:10.1016/j.bbamem.2008.04.011. PMID 18510943.
- Kunji ER, Aleksandrova A, King MS, Majd H, Ashton VL, Cerson E, Springett R, Kibalchenko M, Tavoulari S, Crichton PG, Ruprecht JJ (October 2016). "The transport mechanism of the mitochondrial ADP/ATP carrier". Biochimica et Biophysica Acta (BBA) - Molecular Cell Research. Channels and transporters in cell metabolism. 1863 (10): 2379–93. doi:10.1016/j.bbamcr.2016.03.015. PMID 27001633.
- Palmieri F, Monné M (October 2016). "Discoveries, metabolic roles and diseases of mitochondrial carriers: A review". Biochimica et Biophysica Acta (BBA) - Molecular Cell Research. Channels and transporters in cell metabolism. 1863 (10): 2362–78. doi:10.1016/j.bbamcr.2016.03.007. PMID 26968366.
- Stryer L, Berg JM, Tymoczko JL (2007). Biochemistry. San Francisco: W.H. Freeman. p. 553. ISBN 978-0-7167-8724-2.
- Radzvilavicius AL, Blackstone NW (October 2015). "Conflict and cooperation in eukaryogenesis: implications for the timing of endosymbiosis and the evolution of sex". Journal of the Royal Society, Interface. 12 (111): 20150584. doi:10.1098/rsif.2015.0584. PMC 4614496. PMID 26468067.
- Brandolin G, Dupont Y, Vignais PV (April 1985). "Substrate-induced modifications of the intrinsic fluorescence of the isolated adenine nucleotide carrier protein: demonstration of distinct conformational states". Biochemistry. 24 (8): 1991–7. doi:10.1021/bi00329a029. PMID 2990548.
- Kunji ER, Harding M (September 2003). "Projection structure of the atractyloside-inhibited mitochondrial ADP/ATP carrier of Saccharomyces cerevisiae". The Journal of Biological Chemistry. 278 (39): 36985–8. doi:10.1074/jbc.C300304200. PMID 12893834.
- Pebay-Peyroula E, Dahout-Gonzalez C, Kahn R, Trézéguet V, Lauquin GJ, Brandolin G (November 2003). "Structure of mitochondrial ADP/ATP carrier in complex with carboxyatractyloside". Nature. 426 (6962): 39–44. Bibcode:2003Natur.426...39P. doi:10.1038/nature02056. PMID 14603310. S2CID 4338748.
- Bamber L, Slotboom DJ, Kunji ER (August 2007). "Yeast mitochondrial ADP/ATP carriers are monomeric in detergents as demonstrated by differential affinity purification". Journal of Molecular Biology. 371 (2): 388–95. doi:10.1016/j.jmb.2007.05.072. PMID 17572439. S2CID 6153793.
- Bamber L, Harding M, Monné M, Slotboom DJ, Kunji ER (June 2007). "The yeast mitochondrial ADP/ATP carrier functions as a monomer in mitochondrial membranes". Proceedings of the National Academy of Sciences of the United States of America. 104 (26): 10830–4. Bibcode:2007PNAS..10410830B. doi:10.1073/pnas.0703969104. PMC 1891095. PMID 17566106.
- Kunji ER, Crichton PG (March 2010). "Mitochondrial carriers function as monomers". Biochimica et Biophysica Acta (BBA) - Bioenergetics. 1797 (6–7): 817–31. doi:10.1016/j.bbabio.2010.03.023. PMID 20362544.
- Battini R, Ferrari S, Kaczmarek L, Calabretta B, Chen ST, Baserga R (March 1987). "Molecular cloning of a cDNA for a human ADP/ATP carrier which is growth-regulated". The Journal of Biological Chemistry. 262 (9): 4355–9. doi:10.1016/S0021-9258(18)61355-9. PMID 3031073.
- Nelson DR, Lawson JE, Klingenberg M, Douglas MG (April 1993). "Site-directed mutagenesis of the yeast mitochondrial ADP/ATP translocator. Six arginines and one lysine are essential". Journal of Molecular Biology. 230 (4): 1159–70. doi:10.1006/jmbi.1993.1233. PMID 8487299.
- Chinopoulos C, Gerencser AA, Mandi M, Mathe K, Töröcsik B, Doczi J, Turiak L, Kiss G, Konràd C, Vajda S, Vereczki V, Oh RJ, Adam-Vizi V (July 2010). "Forward operation of adenine nucleotide translocase during F0F1-ATPase reversal: critical role of matrix substrate-level phosphorylation". FASEB Journal. 24 (7): 2405–16. doi:10.1096/fj.09-149898. PMC 2887268. PMID 20207940.
- Chinopoulos C (May 2011). "Mitochondrial consumption of cytosolic ATP: not so fast". FEBS Letters. 585 (9): 1255–9. doi:10.1016/j.febslet.2011.04.004. PMID 21486564. S2CID 24773903.
- Chinopoulos C (December 2011). "The "B space" of mitochondrial phosphorylation". Journal of Neuroscience Research. 89 (12): 1897–904. doi:10.1002/jnr.22659. PMID 21541983. S2CID 6721812.
- Brustovetsky N, Klingenberg M (November 1994). "The reconstituted ADP/ATP carrier can mediate H+ transport by free fatty acids, which is further stimulated by mersalyl". The Journal of Biological Chemistry. 269 (44): 27329–36. doi:10.1016/S0021-9258(18)46989-X. PMID 7961643.
- Doczi J, Torocsik B, Echaniz-Laguna A, Mousson de Camaret B, Starkov A, Starkova N, Gál A, Molnár MJ, Kawamata H, Manfredi G, Adam-Vizi V, Chinopoulos C (May 2016). "Alterations in voltage-sensing of the mitochondrial permeability transition pore in ANT1-deficient cells". Scientific Reports. 6: 26700. Bibcode:2016NatSR...626700D. doi:10.1038/srep26700. PMC 4879635. PMID 27221760.
- Karch J, Bround MJ, Khalil H, et al. Inhibition of mitochondrial permeability transition by deletion of the ANT family and CypD. Sci Adv. 2019;5(8):eaaw4597. Published 2019 Aug 28. doi:10.1126/sciadv.aaw4597
- Ruprecht JJ, Hellawell AM, Harding M, Crichton PG, McCoy AJ, Kunji ER (January 2014). "Structures of yeast mitochondrial ADP/ATP carriers support a domain-based alternating-access transport mechanism". Proceedings of the National Academy of Sciences of the United States of America. 111 (4): E426-34. Bibcode:2014PNAS..111E.426R. doi:10.1073/pnas.1320692111. PMC 3910652. PMID 24474793.
- Ruprecht JJ, King MS, Zögg T, Aleksandrova AA, Pardon E, Crichton PG, Steyaert J, Kunji ER (January 2019). "The Molecular Mechanism of Transport by the Mitochondrial ADP/ATP Carrier". Cell. 176 (3): 435–447.e15. doi:10.1016/j.cell.2018.11.025. PMC 6349463. PMID 30611538.
- Harding AE, Petty RK, Morgan-Hughes JA (August 1988). "Mitochondrial myopathy: a genetic study of 71 cases". Journal of Medical Genetics. 25 (8): 528–35. doi:10.1136/jmg.25.8.528. PMC 1080029. PMID 3050098.
- Rose MR (January 1998). "Mitochondrial myopathies: genetic mechanisms". Archives of Neurology. 55 (1): 17–24. doi:10.1001/archneur.55.1.17. PMID 9443707.
- Kaukonen J, Juselius JK, Tiranti V, Kyttälä A, Zeviani M, Comi GP, Keränen S, Peltonen L, Suomalainen A (August 2000). "Role of adenine nucleotide translocator 1 in mtDNA maintenance". Science. 289 (5480): 782–5. Bibcode:2000Sci...289..782K. doi:10.1126/science.289.5480.782. PMID 10926541.
- Siekevitz P, Potter VR (July 1955). "Biochemical structure of mitochondria. II. Radioactive labeling of intra-mitochondrial nucleotides during oxidative phosphorylation". The Journal of Biological Chemistry. 215 (1): 237–55. doi:10.1016/S0021-9258(18)66032-6. PMID 14392158.
- Pressman BC (June 1958). "Intramitochondrial nucleotides. I. Some factors affecting net interconversions of adenine nucleotides". The Journal of Biological Chemistry. 232 (2): 967–78. doi:10.1016/S0021-9258(19)77415-8. PMID 13549480.
- Bruni A, Luciani S, Contessa AR (March 1964). "Inhibition by atractyloside of the binding of adenine-nucleotides to rat-liver mitochondria". Nature. 201 (1): 1219–20. Bibcode:1964Natur.201.1219B. doi:10.1038/2011219a0. PMID 14151375. S2CID 4170544.
- Duee ED, Vignais PV (August 1965). "[Exchange between extra- and intramitochondrial adenine nucleotides]". Biochimica et Biophysica Acta. 107 (1): 184–8. doi:10.1016/0304-4165(65)90419-8. PMID 5857365.
- Pfaff E, Klingenberg M, Heldt HW (June 1965). "Unspecific permeation and specific exchange of adenine nucleotides in liver mitochondria". Biochimica et Biophysica Acta (BBA) - General Subjects. 104 (1): 312–5. doi:10.1016/0304-4165(65)90258-8. PMID 5840415.
- Saks VA, Lipina NV, Smirnov VN, Chazov EI (March 1976). "Studies of energy transport in heart cells. The functional coupling between mitochondrial creatine phosphokinase and ATP ADP translocase: kinetic evidence". Archives of Biochemistry and Biophysics. 173 (1): 34–41. doi:10.1016/0003-9861(76)90231-9. PMID 1259440.
- Aquila H, Misra D, Eulitz M, Klingenberg M (March 1982). "Complete amino acid sequence of the ADP/ATP carrier from beef heart mitochondria". Hoppe-Seyler's Zeitschrift für Physiologische Chemie. 363 (3): 345–9. doi:10.1515/bchm2.1982.363.1.345. PMID 7076130.
- Adrian GS, McCammon MT, Montgomery DL, Douglas MG (February 1986). "Sequences required for delivery and localization of the ADP/ATP translocator to the mitochondrial inner membrane". Molecular and Cellular Biology. 6 (2): 626–34. doi:10.1128/mcb.6.2.626. PMC 367554. PMID 3023860.
External links
- Adenine+Nucleotide+Translocator+1 at the US National Library of Medicine Medical Subject Headings (MeSH)
- Adenine+Nucleotide+Translocator+2 at the US National Library of Medicine Medical Subject Headings (MeSH)
- Adenine+Nucleotide+Translocator+3 at the US National Library of Medicine Medical Subject Headings (MeSH)