Hentriacontanonaene
Hentriacontanonaene is a long-chain polyunsaturated hydrocarbon produced by numerous gamma-proteobacteria primarily from the marine environment. Hentriacontanonaene was originally isolated from bacterial isolates from Antarctic sea ice cores.[1] All isolated bacteria that produced hentriacontanonaene also produced the polyunsaturated fatty acids eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA).[1][2] Given its polyunsaturated nature it has been proposed that this molecule is produced as part of a response to maintain optimal membrane fluidity.[1][3]
| Names | |
|---|---|
| IUPAC name
(3Z,6Z,9Z,12Z,15Z,19Z,22Z,25Z,28Z)-Hentriaconta-3,6,9,12,15,19,22,25,28-nonaene | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula |
C31H46 |
| Molar mass | 418.709 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Biosynthesis
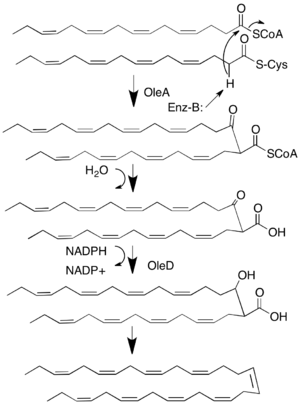
The biosynthesis of this compound was initially identified by its similarity to other known pathways found in bacteria that produce similar long-chain hydrocarbons.[4] Production of monounsaturated and tri-unsaturated long-chain hydrocarbons in various microbial lineages has been attributed to the oleABCD gene cluster.[3] In this pathway two acyl-CoA or acyl-ACP are condensed using a non-decarboxylative Claisen condensation to yield a β-keto-thioester.[5] Hydrolysis from the enzyme is followed by reduction of the β-keto group to an alcohol catalyzed by an NADPH dependent reductase OleD.[6] The remaining steps include decarboxylation and dehydration, which might be combined as a single decarboxylation elimination step.[6] The exact roles of OleB and OleC in this pathway are unknown, however deletion of oleC yielded a strain that produced a mono-ketone product without the completed olefin.[3]
The overall unsaturation of the compound is determined by the acyl precursors and it has been hypothesized that condensation of two 16:4(n-3) acyl chains by OleABCD yields hentriacontanonaene.[2][3][4] A polyketide-like pathway responsible for the production of eicosapentaenoic acid provides the polyunsaturated precursor for hentriacontanonaene.[3]
References
- Nichols, David S.; Nichols, Peter D.; McMeekin, Tom A. (1995). "A new n-C31:9polyene hydrocarbon from Antarctic bacteria". FEMS Microbiology Letters. 125 (2–3): 281–285. doi:10.1111/j.1574-6968.1995.tb07369.x. ISSN 0378-1097.
- Sugihara, Shinji; Hori, Ryuji; Nakanowatari, Hitomi; Takada, Yasuhiro; Yumoto, Isao; Morita, Naoki; Yano, Yutaka; Watanabe, Kazuo; Okuyama, Hidetoshi (2009). "Possible Biosynthetic Pathways for all cis-3,6,9,12,15,19,22,25,28-Hentriacontanonaene in Bacteria". Lipids. 45 (2): 167–177. doi:10.1007/s11745-009-3380-9. hdl:2115/43824. ISSN 0024-4201. PMID 20037794. S2CID 4036770.
- Sukovich, D. J.; Seffernick, J. L.; Richman, J. E.; Hunt, K. A.; Gralnick, J. A.; Wackett, L. P. (2010). "Structure, Function, and Insights into the Biosynthesis of a Head-to-Head Hydrocarbon in Shewanella oneidensis Strain MR-1". Applied and Environmental Microbiology. 76 (12): 3842–3849. Bibcode:2010ApEnM..76.3842S. doi:10.1128/AEM.00433-10. ISSN 0099-2240. PMC 2893466. PMID 20418444.
- Beller, H. R.; Goh, E.-B.; Keasling, J. D. (2009). "Genes Involved in Long-Chain Alkene Biosynthesis in Micrococcus luteus". Applied and Environmental Microbiology. 76 (4): 1212–1223. doi:10.1128/AEM.02312-09. ISSN 0099-2240. PMC 2820947. PMID 20038703.
- Frias, J. A.; Richman, J. E.; Erickson, J. S.; Wackett, L. P. (2011). "Purification and Characterization of OleA from Xanthomonas campestris and Demonstration of a Non-decarboxylative Claisen Condensation Reaction". Journal of Biological Chemistry. 286 (13): 10930–10938. doi:10.1074/jbc.M110.216127. ISSN 0021-9258. PMC 3064148. PMID 21266575.
- Bonnett, Shilah A.; Papireddy, Kancharla; Higgins, Samuel; del Cardayre, Stephen; Reynolds, Kevin A. (2011). "Functional Characterization of an NADPH Dependent 2-Alkyl-3-ketoalkanoic Acid Reductase Involved in Olefin Biosynthesis inStenotrophomonas maltophilia". Biochemistry. 50 (44): 9633–9640. doi:10.1021/bi201096w. ISSN 0006-2960. PMID 21958090.