Milvexian
Milvexian is a factor XIa inhibitor which acts as an anticoagulant. It is taken by mouth. As of late 2021, it was under study for the prevention of blood clots in patients undergoing surgery.[2]
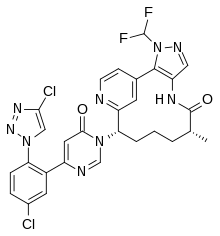 | |
| Clinical data | |
|---|---|
| Other names | BMS-986177, JNJ-70033093 |
| Routes of administration | By mouth |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Elimination half-life | 11.4–18.1 h[1] |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C28H23Cl2F2N9O2 |
| Molar mass | 626.45 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
References
- Perera V, Wang Z, Luettgen J, Li D, DeSouza M, Cerra M, Seiffert D (September 2021). "First-in-human study of milvexian, an oral, direct, small molecule factor XIa inhibitor". Clinical and Translational Science. doi:10.1111/cts.13148. PMID 34558200. S2CID 237617815.
- Weitz JI, Strony J, Ageno W, Gailani D, Hylek EM, Lassen MR, et al. (November 2021). "Milvexian for the Prevention of Venous Thromboembolism". The New England Journal of Medicine. doi:10.1056/NEJMoa2113194. PMID 34780683. S2CID 244132392.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.