Properties of Isotopes of Hydrogen
Hydrogen has three naturally occurring isotopes: 1H (protium), 2H (deuterium), and 3H (tritium). Other highly unstable nuclei (4H to 7H) have been synthesized in the laboratory, but do not occur in nature. The most stable radioisotope of hydrogen is tritium, with a half-life of 12.32 years. All heavier isotopes are synthetic and have a half-life less than a zeptosecond (10-21 sec). Of these, 5H is the most stable, and the least stable isotope is 7H .

Protium
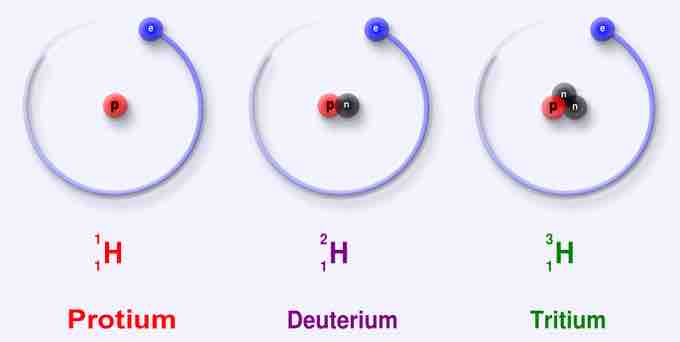
Protium, the most common isotope of hydrogen, consists of one proton and one electron. Unique among all stable isotopes, it has no neutrons.
Protium
1H is the most common hydrogen isotope with an abundance of more than 99.98%. The nucleus of this isotope consists of only a single proton (atomic number = mass number = 1) and its mass is 1.007825 amu. Hydrogen is generally found as diatomic hydrogen gas H2, or it combines with other atoms in compounds—monoatomic hydrogen is rare. The H–H bond is one of the strongest bonds in nature, with a bond dissociation enthalpy of 435.88 kJ/mol at 298 K. As a consequence, H2 dissociates to only a minor extent until higher temperatures are reached. At 3000K, the degree of dissociation is only 7.85%. Hydrogen atoms are so reactive that they combine with almost all elements.
Deuterium
2H, or deuterium (D), is the other stable isotope of hydrogen. It has a natural abundance of ~156.25 ppm in the oceans, and accounts for approximately 0.0156% of all hydrogen found on earth. The nucleus of deuterium, called a deuteron, contains one proton and one neutron (mass number = 2), whereas the far more common hydrogen isotope, protium, has no neutrons in the nucleus. Because of the extra neutron present in the nucleus, deuterium is roughly twice the mass of protium (deuterium has a mass of 2.014102 amu, compared to the mean hydrogen atomic mass of 1.007947 amu). Deuterium occurs in trace amounts naturally as deuterium gas, written 2H2 or D2, but is most commonly found in the universe bonded with a protium 1H atom, forming a gas called hydrogen deuteride (HD or 1H2H).
Chemically, deuterium behaves similarly to ordinary hydrogen (protium), but there are differences in bond energy and length for compounds of heavy hydrogen isotopes, which are larger than the isotopic differences in any other element. Bonds involving deuterium and tritium are somewhat stronger than the corresponding bonds in protium, and these differences are enough to make significant changes in biological reactions. Deuterium can replace the normal hydrogen in water molecules to form heavy water (D2O), which is about 10.6% denser than normal water. Heavy water is slightly toxic in eukaryotic animals, with 25% substitution of the body water causing cell division problems and sterility, and 50% substitution causing death by cytotoxic syndrome (bone marrow failure and gastrointestinal lining failure). Consumption of heavy water does not pose a health threat to humans. It is estimated that a 70 kg person might drink 4.8 liters of heavy water without serious consequences.
The most common use for deuterium is in nuclear resonance spectroscopy. As nuclear magnetic resonance (NMR) requires compounds of interest to be dissolved in solution, the solution signal should not register in the analysis. As NMR analyzes the nuclear spins of hydrogen atoms, the different nuclear spin property of deuterium is not 'seen' by the NMR instrument, making deuterated solvents highly desirable due to the lack of solvent-signal interference.
Isotopes of Hydrogen
The three naturally occurring isotopes of hydrogen.
Tritium
3H is known as tritium and contains one proton and two neutrons in its nucleus (mass number = 3). It is radioactive, decaying into helium-3 through beta-decay accompanied by a release of 18.6 keV of energy. It has a half-life of 12.32 years. Naturally occurring tritium is extremely rare on Earth, where trace amounts are formed by the interaction of the atmosphere with cosmic rays.
Heavier Synthetic Isotopes
4H contains one proton and three neutrons in its nucleus. It is a highly unstable isotope of hydrogen. It has been synthesized in the laboratory by bombarding tritium with fast-moving deuterium nuclei. In this experiment, the tritium nuclei captured neutrons from the fast-moving deuterium nucleus. The presence of the hydrogen-4 was deduced by detecting the emitted protons. Its atomic mass is 4.02781 ± 0.00011 amu. It decays through neutron emission with a half-life of 1.39 ×10−22 seconds.
5H is another highly unstable heavy isotope of hydrogen. The nucleus consists of a proton and four neutrons. It has been synthesized in a laboratory by bombarding tritium with fast-moving tritium nuclei. One tritium nucleus captures two neutrons from the other, becoming a nucleus with one proton and four neutrons. The remaining proton may be detected and the existence of hydrogen-5 deduced. It decays through double neutron emission and has a half-life of at least 9.1 × 10−22 seconds.
6H decays through triple neutron emission and has a half-life of 2.90×10−22 seconds. It consists of one proton and five neutrons.
7H consists of one proton and six neutrons. It was first synthesized in 2003 by a group of Russian, Japanese and French scientists at RIKEN's RI Beam Science Laboratory, by bombarding hydrogen with helium-8 atoms. The helium-8's neutrons were donated to the hydrogen's nucleus. The two remaining protons were detected by the "RIKEN telescope", a device composed of several layers of sensors, positioned behind the target of the RI Beam cyclotron.