Concept
Version 8
Created by Boundless
Dehydration Synthesis
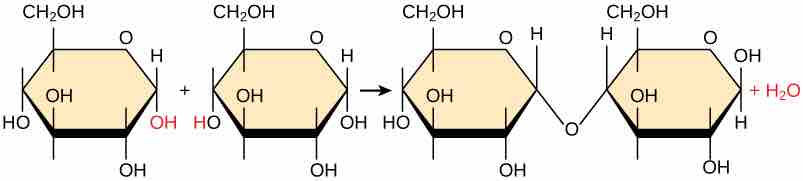
A dehydration synthesis reaction involving un-ionized moners. .
In the dehydration synthesis reaction between two molecules of glucose, a hydroxyl group from the first glucose is combined with a hydrogen from the second glucose, creating a covalent bond that links the two monomeric sugars (monosaccharides) together to form the dissacharide maltose. In the process, a water molecule is formed.
Source
Boundless vets and curates high-quality, openly licensed content from around the Internet. This particular resource used the following sources:
"OpenStax College, Synthesis of Biological Macromolecules. October 16, 2013."
http://cnx.org/content/m44397/latest/Figure_03_01_01.jpg
OpenStax CNX
CC BY 3.0.